(UroToday.com) Prostate cancer diagnosis is predicated on needle biopsy. Histopathologic examination of prostate biopsy specimens is both necessary for diagnosis and to risk-stratify patients with prostate cancer, based on assessment of tumor grade and volume. Despite the key role in assessment of prostate biopsies to guide treatment decision making, there are considerable limitations in the accuracy of grading on the basis of intra-tumoral heterogeneity, inter-observer variability, and other factors.
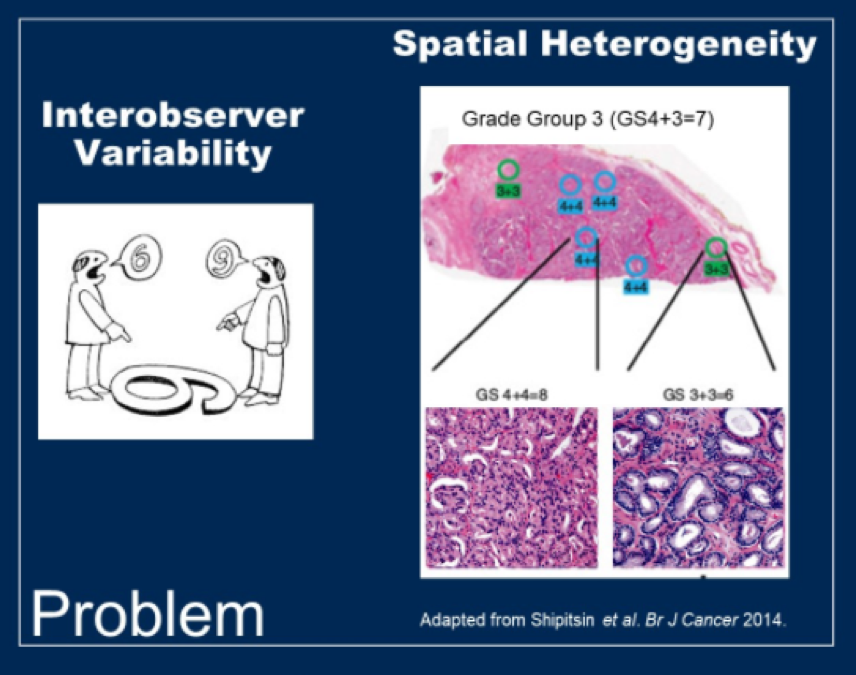
In a plenary abstract presentation in the Poster Highlights Session: Prostate Cancer - Localized Disease session at the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (ASCO GU), Dr. Berman and colleagues present their work which sought to create objective molecular biomarkers that would be resistant to sampling error and should be useful when applied to biopsy tissue, on the basis of radical prostatectomy grade.
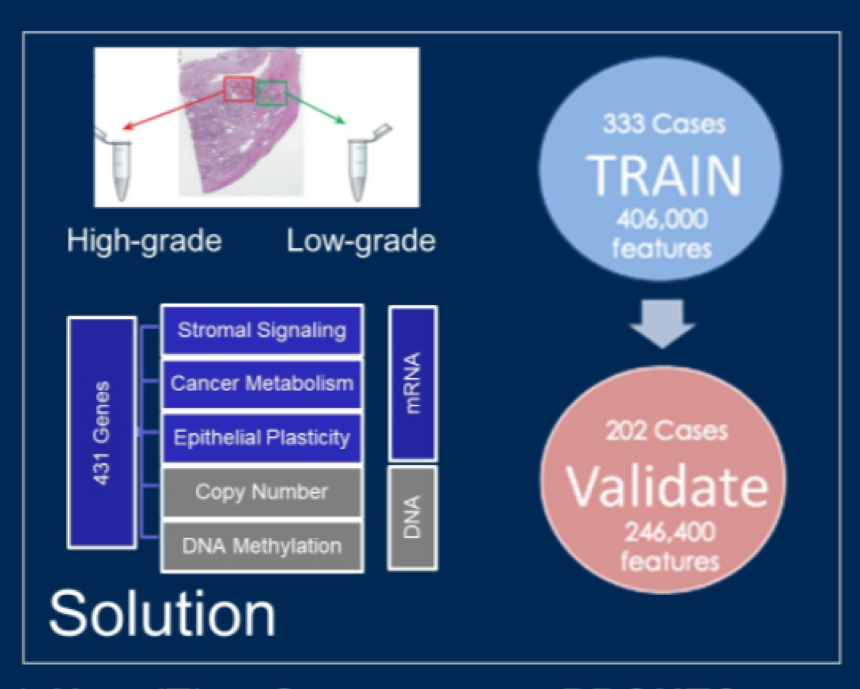
The authors used pathologic assessment of radical prostatectomy specimens as the gold standard to derive a novel objective biomarker of prostate cancer grade. They utilized training (333 patients) and validation (202 patients) cohorts of patients with low- and intermediate-risk prostate cancer on the basis of Cancer of the Prostate Risk Assessment (CAPRA) score. To account for intra-tumoral heterogeneity, each tumor was sampled at two locations. To derive their novel biomarker, the authors profiled the abundance of 342 mRNAs complemented by 100 canonical DNA copy number aberration loci (CNAs) and 14 hypermethylation events. Using the training cohort with cross-validation, we evaluated models for training classifiers of clinically significant prostate cancer (as defined as pathologic Grade Group ≥2). Restricting to strategies resulting in true negative rates ≥0.5, true positive (TP) rates ≥0.8, we selected two strategies to train classifiers, PRONTO-e and PRONTO-m.

The PRONTO-e classifier comprises 353 mRNA and CNA features, while the PRONTO-m classifier comprises 94 mRNA, CNA, methylation, and clinical features. Both classifiers (PRONTO-e, PRONTO-m) validated in the independent cohort, with respective true positive rates of 0.809 and 0.760, false-positive rates of 0.429 and 0.262, F1 scores of 0.709 and 0.724, and AUCs of 0.792 and 0.818.
In summary, the authors developed and validated two classifiers, each of which achieved excellent performance by integrating different types of molecular data to predict prostate tumor grade, on the basis of prostate biopsy. The use of such an approach may allow for more refined inclusion of patients for active surveillance.
Presented by: David Monty Berman, MD, Queen’s University in Kingston Ontario
Co-Authors: Anna YW Lee, Robert Lesurf, Palak Patel, Walead Ebrahimizadeh, Jane Bayani, Nadia Boufaied, Tamara Jamaspishvili, Karl-Philippe Guerard, Dan Dion, Laura A Lee, Vasundara Venkateswaran, Simone Chevalier, Fadi Brimo, Ralph Buttyan, Axel A Thomson, Paul C Park, Jacques Lapointe, Paul Christopher Boutros, John Bartlett
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center, @WallisCJD on Twitter during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021


