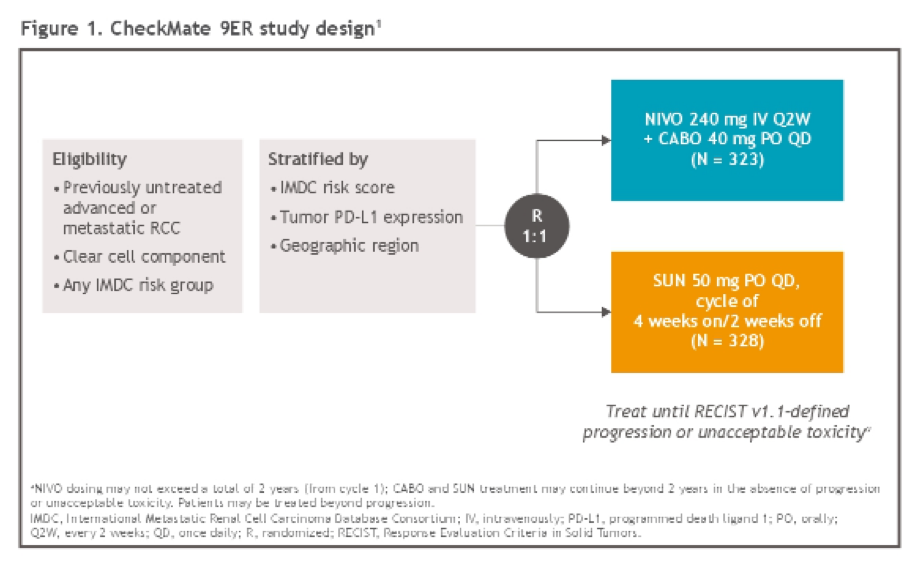
While previously reported, to briefly summarize, the phase III, open-label CheckMate 9ER trial (NCT03141177) randomized patients with advanced RCC in a 1:1 fashion to receive nivolumab 240 mg IV Q2W + cabozantinib 40 mg PO QD (N+C; n = 323) or sunitinib (S) 50 mg PO (4 weeks of 6-week cycles; n = 328) in the first-line setting, with randomization stratified by International Metastatic Renal Cell Carcinoma Database Consortium risk score, tumor programmed death ligand 1 expression, geographic region. Treatment was continued until disease progression or unacceptable toxicity.
In this analysis, the authors examined health-related quality of life (HRQoL) patient-reported outcome results, including overall between-group comparisons of treatment groups and time to confirmed deterioration (TCD). Patient reported outcomes were an exploratory endpoint among all randomized patients, as assessed using the Functional Assessment of Cancer Therapy Kidney Symptom Index-19 (FKSI-19) and EQ-5D-3L instruments. These assessments were performed at baseline, at common on-treatment scheduled visits, and at common follow-up visits.
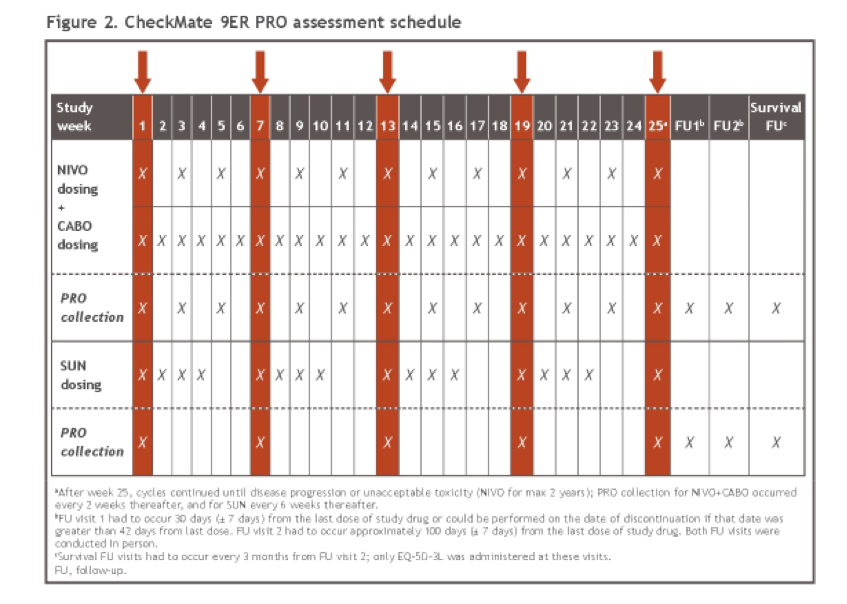
Changes from baseline were assessed using mixed-model repeated measures (MMRM), adjusting for baseline scores and stratification factors. TCD was calculated from Kaplan–Meier estimates and Cox proportional hazards models.
At the time of data analysis, the median follow-up for overall survival was 18.1 months. Patient reported outcome measures were completed among more than 90% of patients at baseline, and at least 80% of patients at all on-treatment assessments through week 91 in both arms.
The overall least squares mean difference in change from baseline favored patients treated with nivolumab and cabozantinib, as compared to those receiving sunitinib, as measured with the FKSI-19 (all domains) and with the EQ-5D-3L. 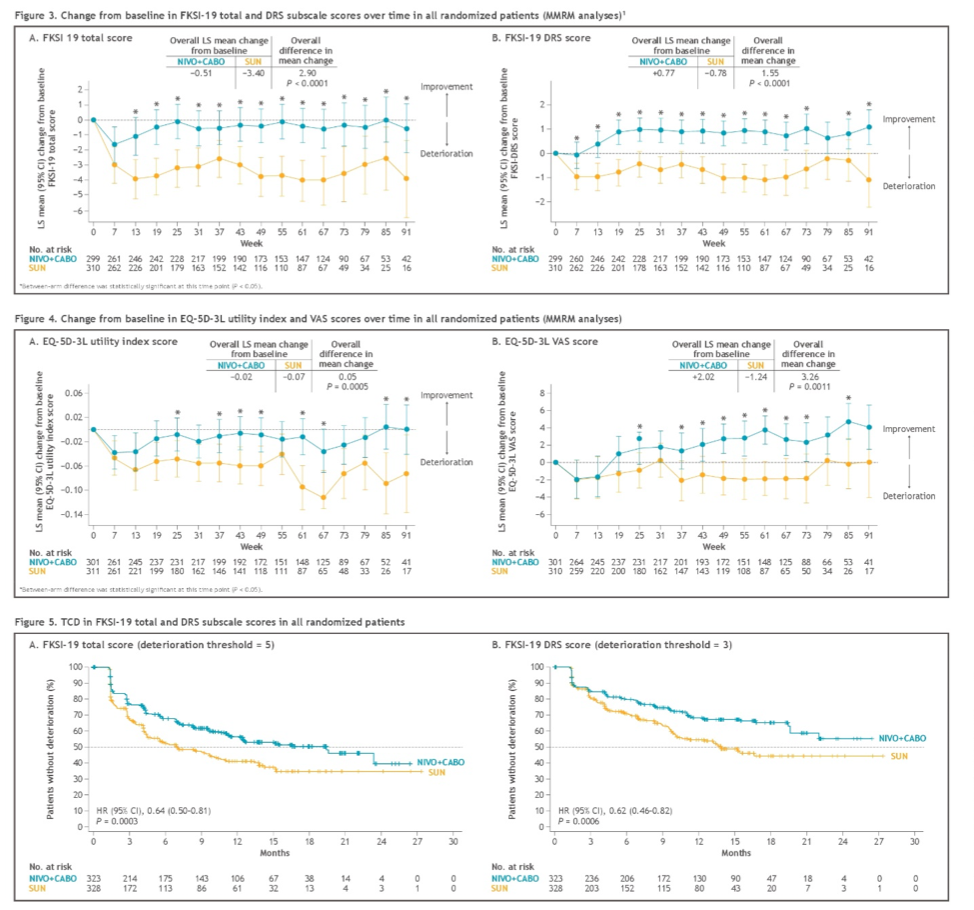
Patients treated with nivolumab and cabozantinib experienced a lower treatment burden, with decreased risk of confirmed deterioration across most measurements versus sunitinib, including FKSI-19 total, disease-related symptoms (DRS), DRS-physical (DRS-P), DRS-emotional (DRS-E), functional well-being (FWB), and EQ-5D-3L visual analog scale (VAS) scores.
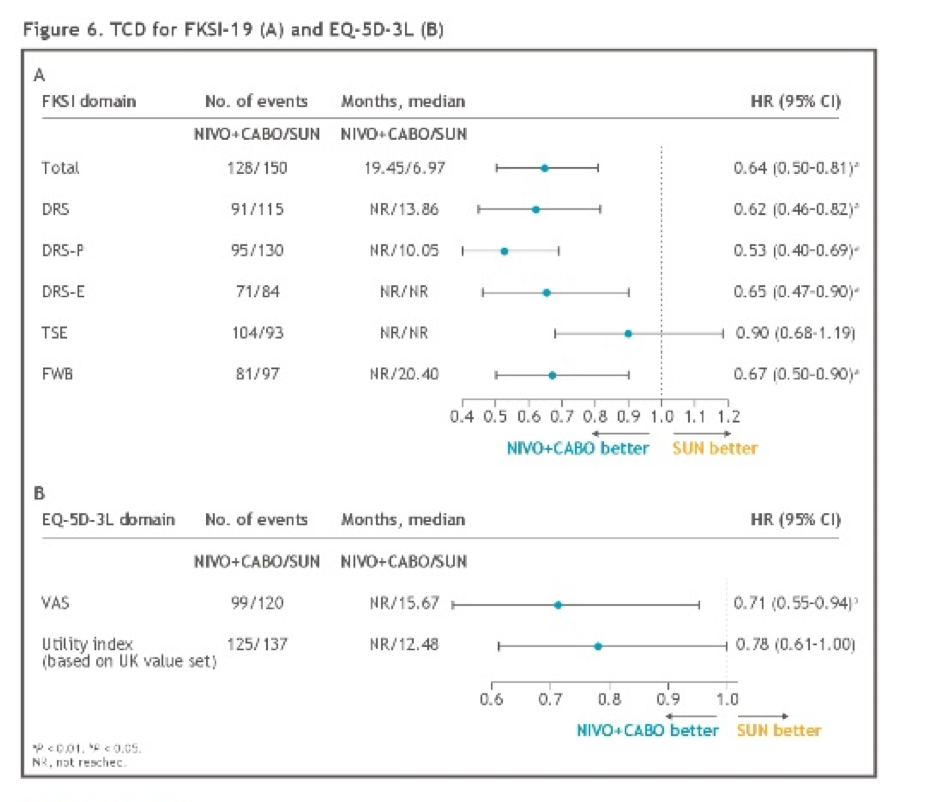
The authors conclude that, in addition to previously presented data demonstrating superiority with respect to progression-free survival and overall survival, patients treated with nivolumab and cabozantinib had statistically significant HRQoL benefits, compared to patients treated with sunitinib.
Presented by: David Cella, PhD, Professor, Medical Social Sciences; Feinberg School of Medicine
Co-Authors: Toni K. Choueiri, Steven I. Blum, Flavia Ejzykowicz, Melissa Hamilton, Joshua Zhang, Cristina Ivanescu, Robert J. Motzer
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center, @WallisCJD on Twitter during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021
Related Content:
ESMO 2020: CheckMate-9ER Study of Nivolumab Combined With Cabozantinib Vs. Sunitinib in Participants With Previously Untreated mRCC - Toni Choueiri
ESMO Virtual Congress 2020: Invited Discussant: First Results From the Randomized Phase 3 CheckMate 9ER Trial

