San Francisco, CA (UroToday.com) In prostate cancer, therapeutic pressure may lead to small cell or neuroendocrine prostate cancer transformation from prostate adenocarcinoma. This evolution can occur through epigenetic reprogramming (Figure 1).
Figure 1 – Epigenetic reprogramming: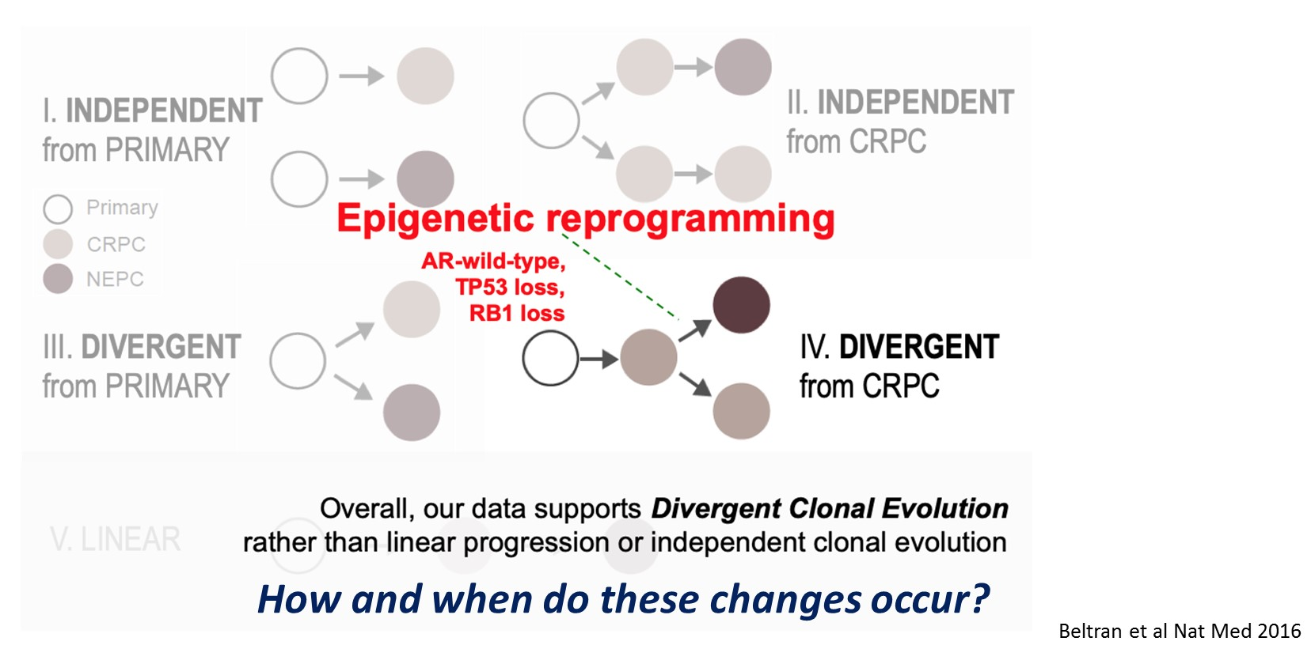
Himisha Beltran, MD, began by elaborating on the tumor dynamics towards the development of neuroendocrine prostate cancer. The timing of genomic, epigenomic, and molecular events may have biomarker and or therapeutic implications. It is very challenging to perform serial metastatic biopsies in an attempt to follow these dynamic changes. However, circulating tumor DNA may overcome some of these challenges. This allows serial sampling in a non-invasive and feasible approach, and it could potentially avoid serial metastatic tumor biopsy looking for neuroendocrine development, as is the current standard.
Dr. Beltran presented a study where 62 patients across the metastatic disease spectrum were evaluated. The patients had matched metastatic tumor biopsy and plasma samples including serial samples with overall 73 plasma and 116 metastatic tissues obtained. A comprehensive approach was undertaken with CTDNA whole-exome sequencing plus biopsies plus germline DNA analysis performed.
This study’s questions where to analyze whether circulating tumor DNA are reflective of what we see in tumor biopsies, possibly paving the way for non-invasive detection of the development of castrate-resistant prostate cancer with neuroendocrine features. Additionally, the authors wanted to know what degree of heterogeneity underlies castrate-resistant prostate cancer with-neuroendocrine features, compared with castrate-resistant prostate adenocarcinoma. The last question was whether CTDNA can distinguish clonal evolution versus rare clonal selection.
The study showed the patients had 84% metastatic bone lesions, 52% metastatic lymph node lesions, 30% liver metastatic lesions, 16% lung lesions an 6% brain lesions. The cfDNA tumor content did not associate with pathological subtype (adenocarcinoma or neuroendocrine) or the sites or the number of metastasis. Moreover, the cfDNA content did not associate with serum PSA or serum neuroendocrine markers. The prostate cancer genomic alteration in plasma and tissues are shown in figure 2.
Figure 2 - Prostate cancer genomic alteration in plasma and tissues:
The results also showed that androgen receptor gain, RB1 and TP53 were associated with overall survival in prostate adenocarcinoma, but not in the neuroendocrine cancer (Figure 3 and 4). The concordance of genomic alterations between tissue and plasma were reflective of less intra-patient heterogeneity in neuroendocrine prostate cancer (Figure 5).
Figure 3: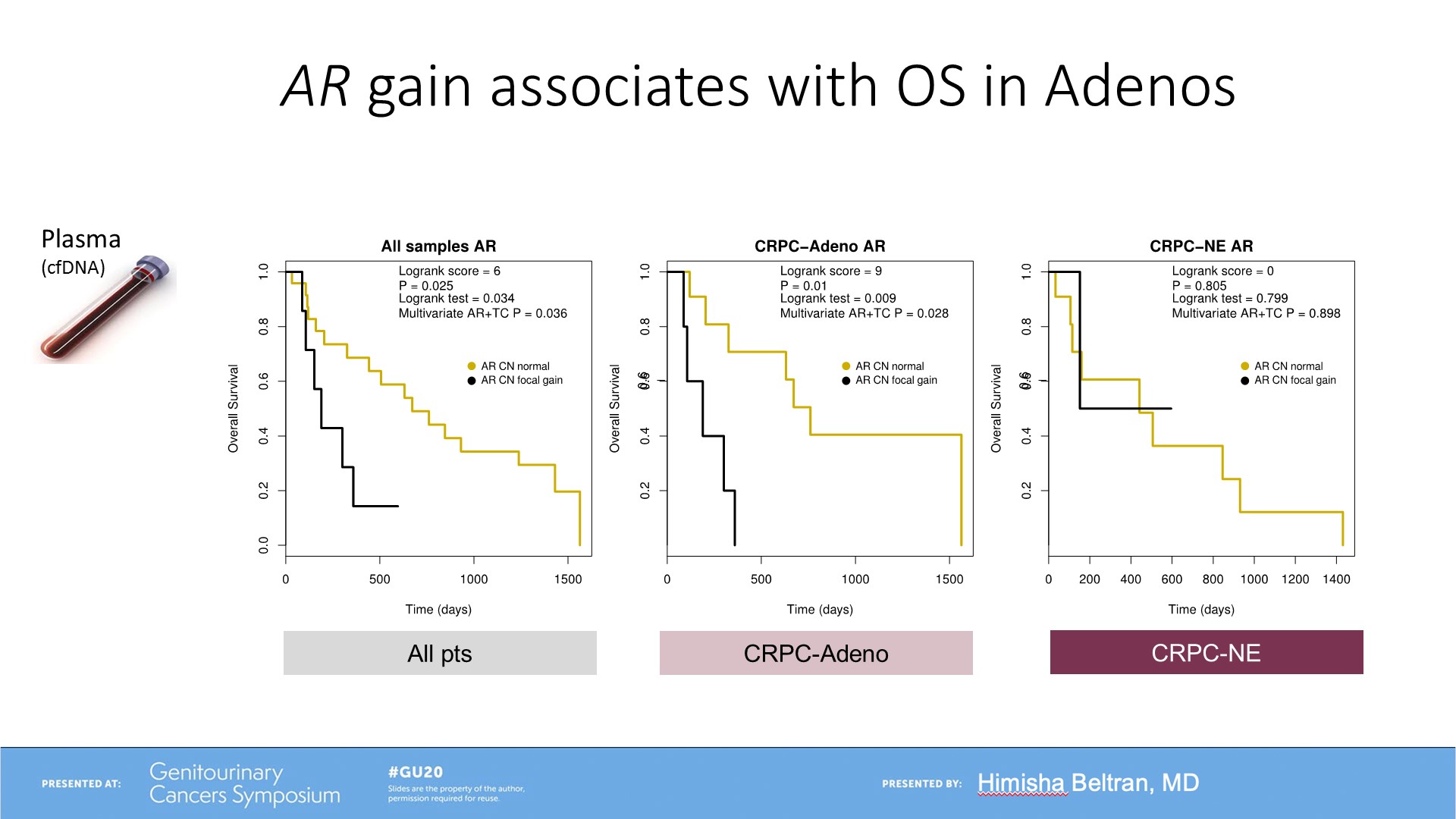
Figure 4: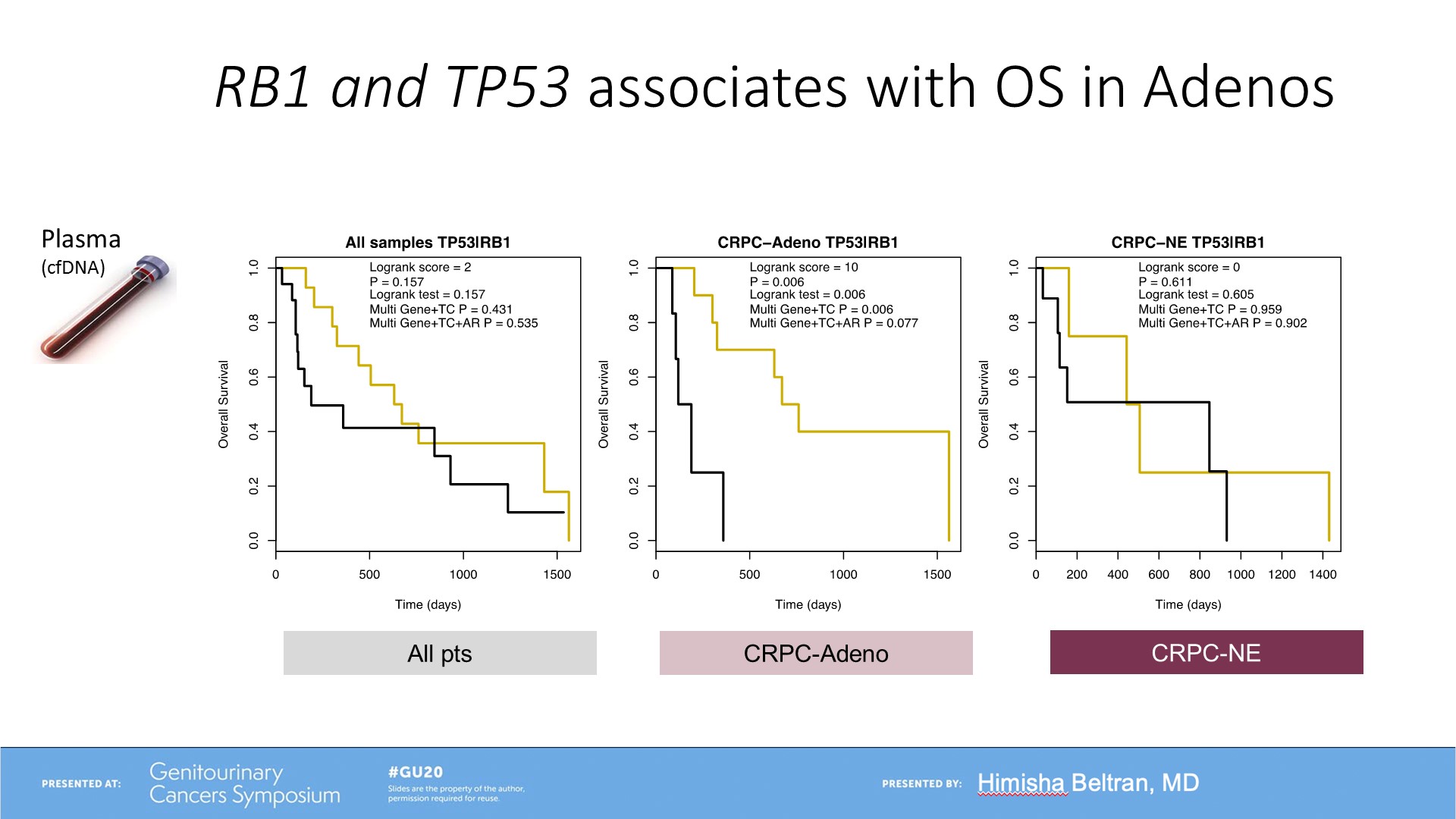
Figure 5 – Intra-patient heterogeneity in prostate adenocarcinoma and neuroendocrine cancer:
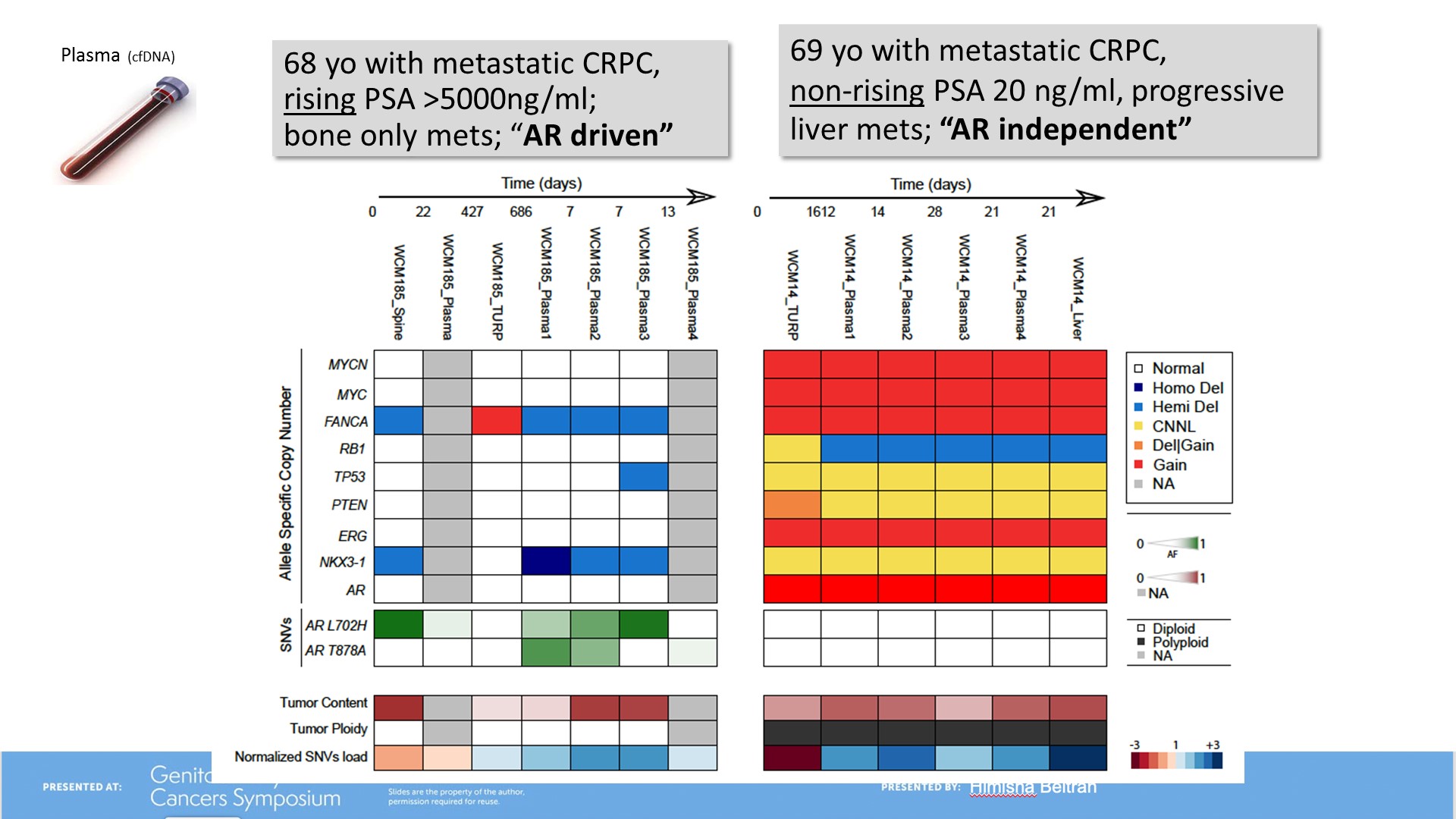
Dr. Beltran continued to suggest a proposed model of clonal evolution to neuroendocrine prostate cancer (Figure 6).
Figure 6 – Proposed model of clonal evolution: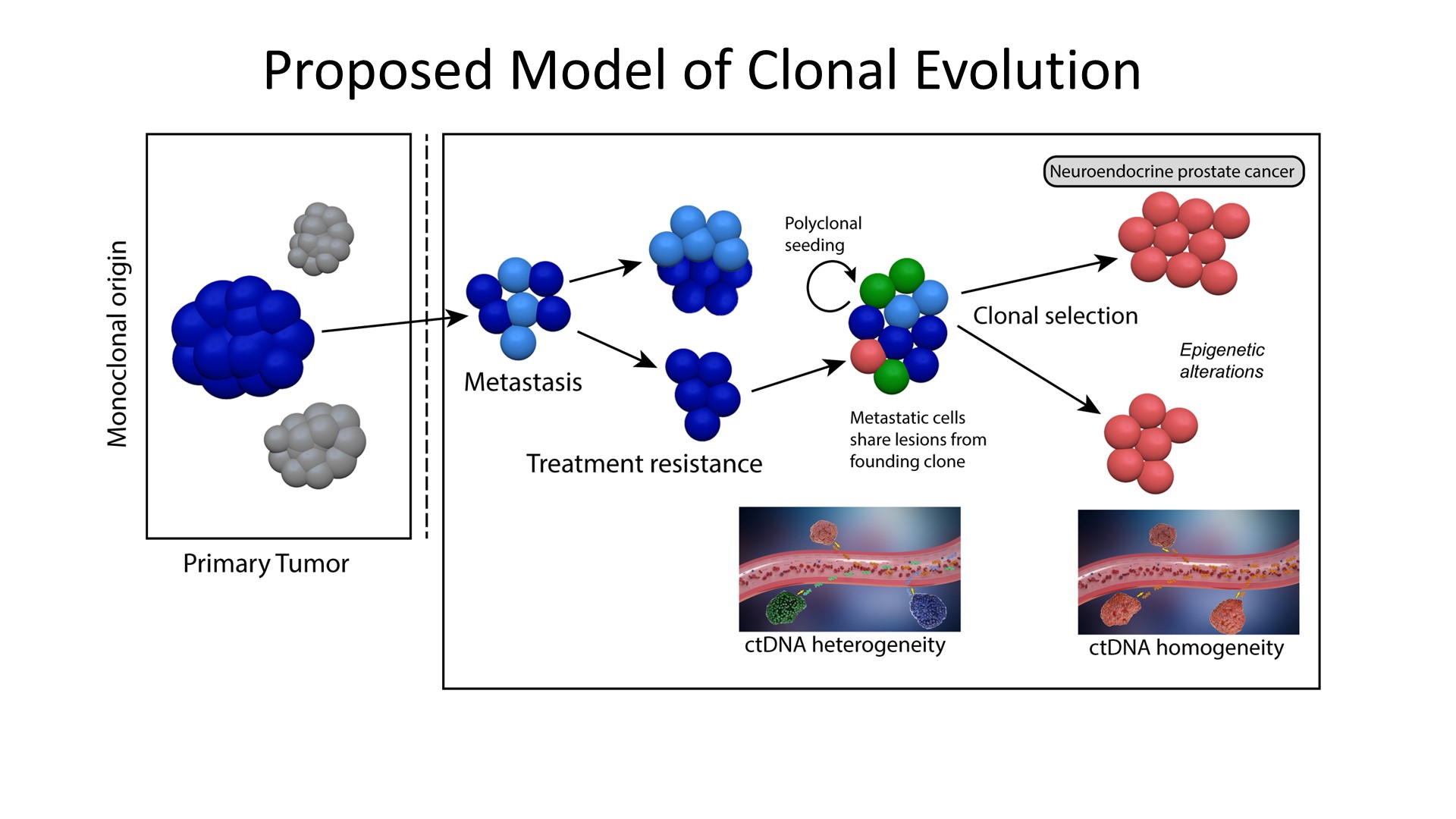
Finalizing her talk, Dr. Beltran raised some open questions that still need to be addressed. How often and when do these changes emerge after potent androgen receptor therapy? Are there high risk localized metastatic patients with castrate-resistant prostate cancer with neuroendocrine feature alterations? What features predispose patients to future neuroendocrine transformation? Is there anything else that is needed to develop this histologic transformation? And lastly, how can we develop liquid biopsies for trial selection?
In summary, according to Dr. Beltran, circulating tumor DNA captures genomic and epigenomic features of neuroendocrine prostate cancer in a non-invasive manner. Castrate resistant prostate cancer with neuroendocrine features is characterized by less intra-patient and less inter-patient tumor heterogeneity versus castrate-resistant prostate cancer with adenocarcinoma features. The known clinical data supports the fact that castrate-resistant prostate cancer with neuroendocrine features arises from a prostate adenocarcinoma precursor, but other data also suggests that there may be early dominance in the emergence of a resistant clone (clonal selection) - not mutually exclusive clonal evolution.
Presented by: Himisha Beltran, MD, Associate Professor of Medicine, Lank Center for Genitourinary Oncology and the Division of Molecular and Cellular Oncology, Dana Farber Cancer Institution, Boston, MA
Written By: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, NY, USA, Twitter: @GoldbergHanan, at the 2020 Genitourinary Cancers Symposium, ASCO GU #GU20, February 13-15, 2020, San Francisco, California.


