This study aimed to determine if accelerated BEP is superior to standard BEP as first-line chemotherapy for intermediate and poor risk metastatic germ cell tumors.
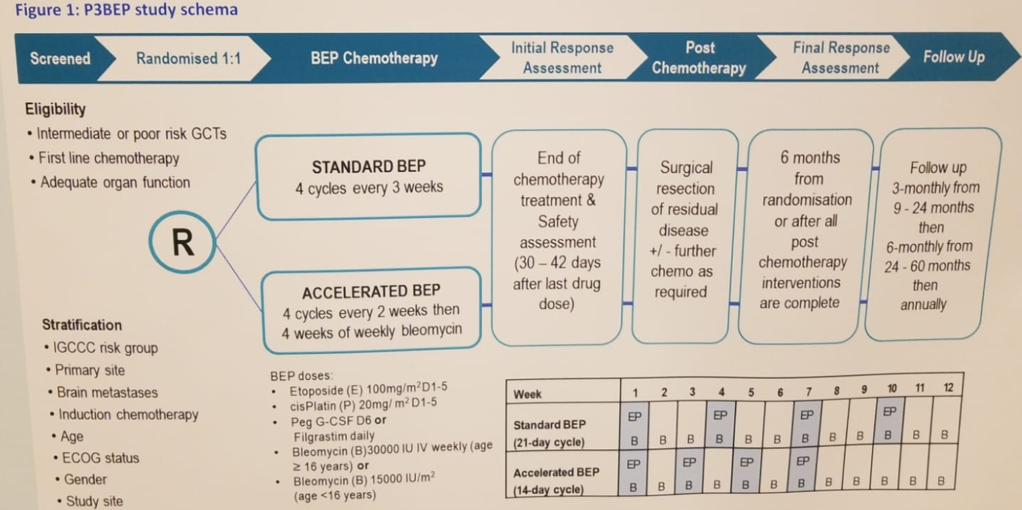
This is an open-label, randomized, stratified, 2-arm, multicenter, 2-stage phase 3 trial (Clinical trial information: NCT02582697). Patients will be aged between 11 and 45, with intermediate or poor risk germ cell tumor, arising in testis, ovary, retroperitoneum, or mediastinum, who are considering first-line chemotherapy. A total of 150 patients are planned for stage 1, and 500 are planned for stage 2. The study scheme is shown in figure 1.
The primary endpoint will be progression-free survival, and the secondary endpoints include response following treatment completion, adverse events, health-related quality of life, treatment preference, delivered dose intensity, and overall survival.
The enrolment opened in February 2014 in 4 countries (USA, UK, Australia and New Zealand), with 67 sites involved, and 76 patients randomized so far.
Figure 1 – Trial scheme:
Presented by: Alison Zhang, MBBS, FRACP, MMedSc, Department of Medical Oncology, Peter MacCallum Cancer Centre, Australia
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, @GoldbergHanan at the 2019 American Society of Clinical Oncology Genitourinary Cancers Symposium, (ASCO GU) #GU19, February 14-16, 2019 - San Francisco, CA


