
Huang KL et al (Cell 2018) looked at 10,000+ adult cancers in the TCGA dataset and confirmed historical rates of 1-9% of RCC’s having a “strong genetic component.” Indeed, in patients with clear cell, papillary and chromophobe RCC, 6%, 9%, and 6% (respectively) had pathogenic germline mutations. While some of these may be incidental, known driver mutations were identified in these subsets.
What is the importance of genetic testing?
Genetic testing has implications to the patient other than for just RCC management. It may reveal risk of pathology in other organs, allow for early intervention to prevent later complications, and may help inform management choice (or alter from standard of care). It also has implications for patient families, as cascade testing has become more common –screening at risk family members may result in earlier intervention. Some patients also may utilize this to inform prenatal genetic testing.
Who should get genetic testing?
There are numerous pitfalls to genetic testing in the RCC patient population. While patients with classic syndromes are often taught in urology training programs, patients don’t often present with “classic” symptoms. Additionally, physicians may be unfamiliar with many of the rarer syndromes, unfamiliar with all the non-renal manifestations of these syndromes, patients may not be the best historians, and there is variable penetrance of certain manifestations. Also, it is important to remember that ~10% of patients with syndromes present with de novo mutations (founder mutations).
Unfortunately, the current guidelines from different institutions are vague –and while they share some common features (younger age, bilateral tumors, etc), there needs to be a better consensus on the details.
However, age is clearly the strongest predictor of syndromic RCC. The median age of RCC is 64. RCC in younger patients deserves special attention. In this work by Dr. Shuch, comparing patients with syndromic RCC managed at the NIH vs. RCC’s in the SEER dataset, you can see a distinct population presenting itself:

In a separate study, in a validation of a gene panel (Ambry RenalNext panel of 19 genes), Dr. Shuch found that age was the strongest predictor of a positive panel result –more than ethnicity, race and family history. (Nguyen et al. Cancer 2017)
His take-home current recommendations on who to refer, in the absence of uniform guidelines:
- Age < 46-Bilateral or multifocal disease
- Family history of RCC
- Personal history of RCC or benign tumors
- Suspicious RCC subtypes (non-ccRCC)
- Suspicious somatic tumor profiling
There are many options for genetic testing and where to get genetic testing can be confusing.
- Home genetic testing –while he wouldn’t recommend this clinically, many patients now have this already done. You are obligated to review their results from these organizations, such as color.com, 23andme.com, ancestry.com, etc.
- Cancer genetics clinic –this is ideal, but resources are limited! They involve a genetic counselor, geneticist, and associated clinical providers
- Clinicians office –should be a clinician with appropriate expertise, NP with advanced genetics certification
When to test and when to treat?
Localized kidney cancer in at risk individuals –test first if:
- High suspicion AND safe to delay intervention AND
- Management may change
- It's better to wait for correct treatment AND delay is safe
He notes that testing must be thorough and requires clinical expertise. Often times insurance companies will require 3-generation pedigrees and will only cover genetic testing once –so you have to make sure it counts!
Again, counseling and evaluation does not mean mandatory testing –multiple factors go into the decision to test:

ASCO actually provides an extensive pre-testing guideline (can be found on their website).
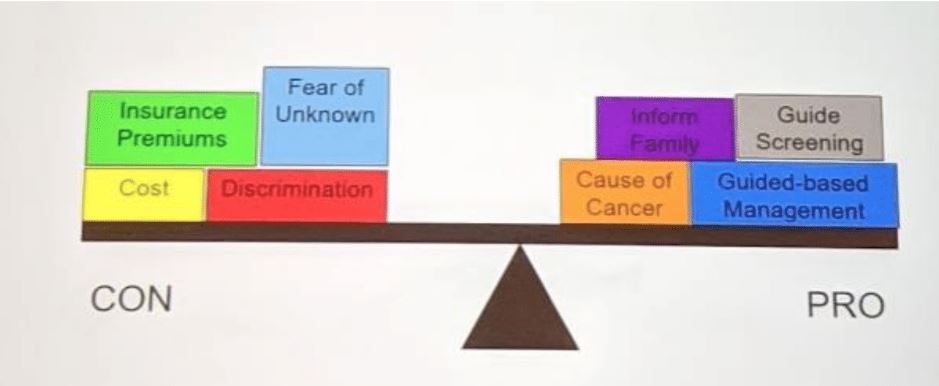
The decision to test is also a shared decision –and don’t be surprised if patients have personal reasons to not test. You must recognize and accept that patients may NOT want genetic testing (insurance premiums, cost, etc).
There is also a spectrum of testing –either single gene for suspected monogenic syndrome vs. expanded panels (up to 80 genes!). However, as you broader your search, you may end up with results of unknown significance –and you must have the expertise to be able to explain these findings (variants of unknown significance, etc):

After providing a few examples, his concluding statements were:
- Patients with RCC genetic conditions will be seen in your clinic... but if you don’t look for it, you won’t find it!
- Early onset, multifocality, family history, and personal history should guide referral for counseling.
- Establish a workflow for referral to a cancer genetics provider
- Not all individuals who are counseled should get tested!
- Not everyone who should get tested will agree to testing.
- There are many options for testing –be smart about which test to select!
Written by: Thenappan Chandrasekar, MD, Clinical Instructor, Thomas Jefferson University, Philadelphia, Pennsylvania, twitter: @tchandra_uromd, @JEFFUrology at the 2019 American Society of Clinical Oncology Genitourinary Cancers Symposium, (ASCO GU) #GU19, February 14-16, 2019 - San Francisco, CA


