(UroToday.com) The 2023 ASCO annual meeting included a testicular cancer session, featuring a presentation by Dr. Torgrim Tandstad discussing combined results of the SWENOTECA and Cologne experience of primary retroperitoneal lymph node dissection (RPLND) in seminoma stage IIA-IIC ≤3 cm tumors. Chemotherapy or radiotherapy has been standard treatment for stage II seminomas with excellent survival but is associated with significant long-term treatment-related toxicities. This is important given the very high cure rates for CSI tumors (5-year overall survival >98%), and also for metastatic tumors (nonseminoma – 5-year overall survival > 90%; seminoma – 5-year overall survival > 96%). Additionally, there is the long life-expectancy following treatment (40-70 years):

However, and importantly, late effects affecting mortality do not show up until 20+ years after diagnosis, specifically related to long-term toxicity. In a population-based study from Norway [1], over a median follow-up of 18.7 years, the non-testicular cancer death was 12%. Overall excess non-testicular cancer mortality was 23% compared to the general population, with increased risks after platinum-based chemotherapy and radiotherapy:
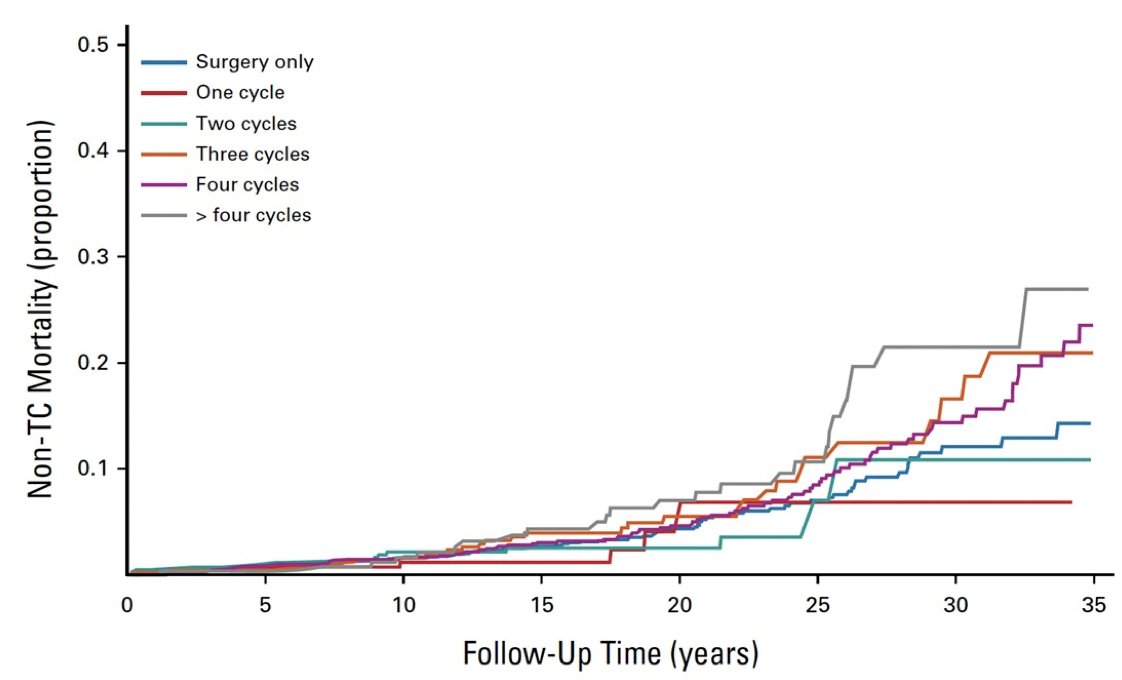
There is interest in primary RPLND, as there is no increased risk of late toxicity affecting mortality, there are many small series and recent newer trials (SEMS and PRIMETEST) suggesting feasibility and safety, with COTRIMS starting in 2018.

The current SWENOTECA treatment program recommends primary RPLND in seminoma stage IIA to IIB ≤ 3 cm with 1-2 metastatic nodes as the standard treatment modality. At the 2023 ASCO annual meeting, Dr. Tandstad and colleagues presented preliminary results combined with data from Cologne.
Within SWENOTECA, RPLND is centralized into five hospitals and Cologne is a tertiary center for patients recruited from different parts of Germany. Both patients with recurrence after initial stage I disease (n = 58) and patients with primary stage IIA and IIB at diagnosis (n = 36) were included. Dr. Tandstad and his group collected information on peri- and postoperative complications, and also analyzed histologic outcome and whether adjuvant chemotherapy was given.
This study included 94 patients operated from May 2018 to November 2022, and the number of operations per site varied from 4 to 28. The median age of patients at the time of RPLND was 41.8 years (range 21-79) and median follow-up since RPLND was 21 months (range 4-61). Twenty-six patients (28%) were operated with robotic laparoscopic technique. 10 patients (10.6%) had a Clavien-Dindo postoperative complication > 2, and 23 of the patients were given adjuvant oncological treatment after RPLND, most of them one course of BEP chemotherapy. The histology from RPLND showed seminoma in 83 patients, benign/necrosis in eight patients, vital non-seminomatous germ cell tumor in one patient, teratoma only in one patient, and lymphoma in one patient. Mean number of resected nodes was 19, and mean number of positive nodes was 1.5. The patients have been followed for a median 18 months (range 2-59), of which nine patients (9.6 %) have had recurrences, all but one in the first year after RPLND.
Dr. Tandstad concluded this presentation by discussing combined results of the SWENOTECA and Cologne experience of primary RPLND in seminoma stage IIA-IIC ≤ 3cm tumors with the following take-home points:
- These early results of primary RPLND of seminomas IIA-IIB ≤ 3 cm are promising and can be performed at experienced centers
- Longer follow-up is required to ensure this as a safe treatment option, but currently risk of late morbidity and mortality is minimal
Presented by: Torgrim Tandstad, MD, PhD, St. Olav's University Hospital, Trondheim, Norway
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Associate Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, Fri, June 2 – Tues, June 6, 2023.
References:
- Hellesnes R, Myklebust TA, Fossa SD, et al. Testicular cancer in the cisplatin era: Causes of death and mortality rates in a population-based cohort. J Clin Oncol. 2021 Nov 10;39(32):3561-3573.


