Men with progressive mCRPC following at least 1 potent AR-pathway inhibitor (ie. abiraterone or enzalutamide) and chemotherapy (or unfit/refuse chemotherapy) without limit of number of prior therapies (including Radium-223 or prior 177Lu-PSMA) provided adequate organ function were eligible. Baseline 68Ga-PSMA11 PET was performed, but not used for eligibility. Dose-escalation was in single-subjects x4 followed by 3+3 with a single infusion of 225Ac-J591 (13.3 KBq/kg with planned escalation up to 93.3 KBq/kg). Dose-limiting toxicity was defined as attributable grade 4 hematological toxicity or grade 3/4 non-hematological toxicity. Imaging, genomic, patient-reported outcomes (PRO), and immune correlates embedded.
There were 32 men treated with a single dose of 225Ac-J591 on 7 dose levels with expansion at the highest dose level (n = 16). The median age was 69.5 (range 52-89) years and median PSA was 149.1 ng/mL (range 4.8-7168.4). There were 75% of patients with >2 prior AR-pathway inhibitors, 62.5% had previous chemotherapy, 28% previous Radium-223, and 43.7% previous 177Lu-PSMA. While PSMA uptake was not a prerequisite for treatment, among 28 patients with pre-treatment PSMA PET:
- None had a tumor SUVmax < liver
- 5 (17.8%) with tumor SUVmax 1-2.5x liver
- 2 (7.2%) with tumor SUVmax5-5x liver
- 21 (75%) with tumor SUVmax > 5x liver SUVmean
One of six patients in cohort 6 (80 KBq/kg) had dose-limiting toxicity (grade 4 anemia and platelets) with 0 of 6 patients at the highest dose level (93.3 KBq/Kg) and thus this dose was expanded. High grade adverse events were restricted to hematologic: in addition to dose-limiting toxicity, 4 (12.5%) patients with grade 3 platelets and 2 (6.2%) patients with grade 3 neutropenia. Non-hematological adverse events were restricted to grade 1/2 and included: 10 patients (31.2%) with fatigue, 5 patients (15.6%) with pain flare, 14 patients (43.7%) with nausea, 8 patients (25%) with grade 1 xerostomia (of which 5 received prior 177Lu-PSMA), and 12 patients (37.5%) with AST elevation. Despite prior treatment including 177Lu-PSMA and no selection for PSMA expression, 22 (68.8%) patients had any PSA decline, and 14 (43.8%) had > 50% PSA decline: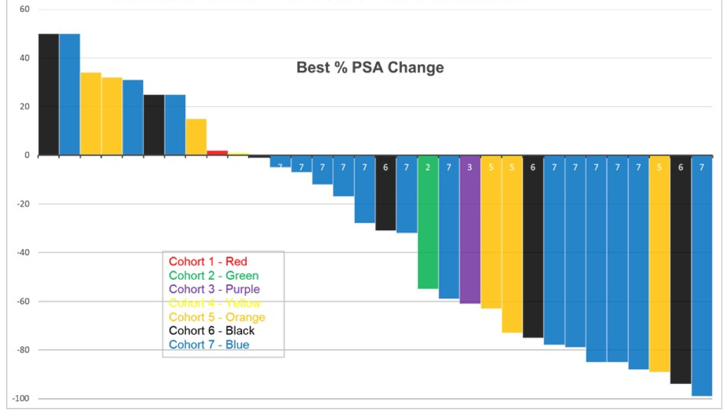
Among 21 patients with paired baseline and 12-week circulating tumor cell counts, 12 patients declined (5 converting from unfavorable to favorable and 5 converting detectable to 0), 5 patients remained 0, and 4 patients increased their circulating tumor cell count. In the subset with PRO data, pain scores by BPI-SF tended to improve by week 12. Following a single dose of 225Ac-J591, median PFS was 7.2 months (95% CI 4.6-not reached), and median OS was 10.9 months (95% CI 7.6-21.1).
Dr. Tagawa concluded his presentation of this phase 1 trial with the following summary statements:
- PSMA-targeted alpha-emitter 225Ac utilizing intact antibody J591 is tolerable with early evidence of clinical activity
- Early evidence of clinical activity including PSA and circulating tumor cell count decline in heavily pre-treated population, without selection by PSMA imaging and 44% with prior PSMA radioligand therapy
- Analysis of imaging, genomic, and immune correlates, and patient reported outcomes is ongoing
- Based upon these results, a follow up study [NCT04506567] testing multiple and fractionated dosing of 225Ac-J591 is underway
Clinical trial information: NCT03276572
Presented by: Scott T. Tagawa, MD, Associate Professor of Clinical Medicine, Clinical Urology, Medical Director, Genitourinary Oncology Research Program, Weill Cornell Medicine, New York, NY
Co-Authors: Michael Sun, A. Oliver Sartor, Charlene Thomas, Sharon Singh, Mahelia Bissassar, Escarleth Fernandez, Muhammad Junaid Niaz, Benedict Ho, Shankar Vallabhajosula, John Babich, Ana M. Molina, Cora N. Sternberg, David M. Nanus, Joseph Osborne, Neil Harrison Bander; Weill Cornell Medicine, New York, NY; Tulane Cancer Center, New Orleans, LA; Weill Cornell Medical Center, New York, NY; Cornell University, Ithaca, NY; Sandra and Edward Meyer Cancer Center, New York, NY
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021
Related Content:
Determining Dose-Limiting Toxicity and Maximum Tolerated Dose of 225Ac-J591 in Metastatic Castration-Resistant Prostate Cancer - Scott Tagawa

