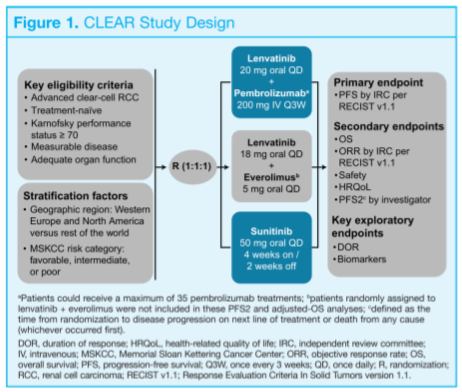
Three patient questionnaires were used to assess HRQoL as a planned secondary endpoint of the study. There were the FKSI-DRS (functional assessment of cancer therapy kidney symptom index – disease related symptoms), the EORTC QLQ-C30 (European organization for research and treatment of cancer quality of life questionnaire for patients with cancer – core 30), and the EQ-5D-3L (European quality of life 5-dimension 3-level system). These questionnaires were administered at baseline prior to randomization, then on day one of each cycle starting with cycle 2, and finally off treatment. The details of each questionnaire are summarized in the table below.

Two different analyses were performed: (1) longitudinal change from baseline and (2) time to deterioration. The longitudinal change from baseline was assessed using a mixed model analysis. The time to deterioration was measured both as time to first deterioration, which is the number of weeks between randomization and first deterioration event, and time until definitive deterioration, measured as the number of weeks between randomization and the earliest deterioration event with no subsequent recovery. Each scale had a different threshold for defining deterioration. Deterioration for the FKSI-DRS was a decrease of more than 3 points, more than 10 points for the EORTC QLQ-C30, and 0.08 points or more for the DQ-5D-3L scale. No adjustments for multiple testing were performed, and all p-values are nominal and descriptive.
With regards to the len/eve treatment arm, quality of life across different metrics was either similar or favored sunitinib such as with regards to pain, appetite loss, and diarrhea. The least squares mean difference between len/eve and sunitinib of greater than 5 is likely to be clinically very significant.
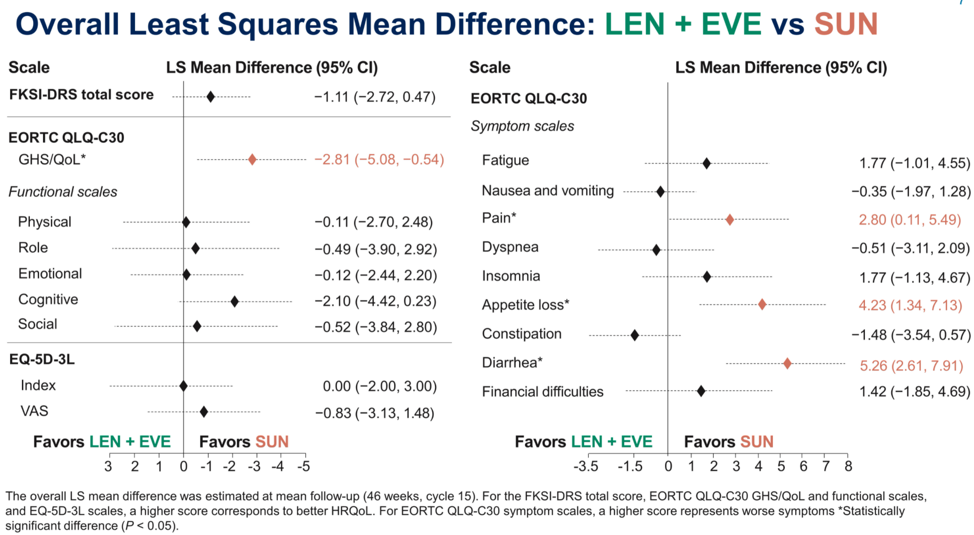
For len/pembro, HRQoL metrics were either similar or favored len/pem, such as with physical function, fatigue, dyspnea and constipation.
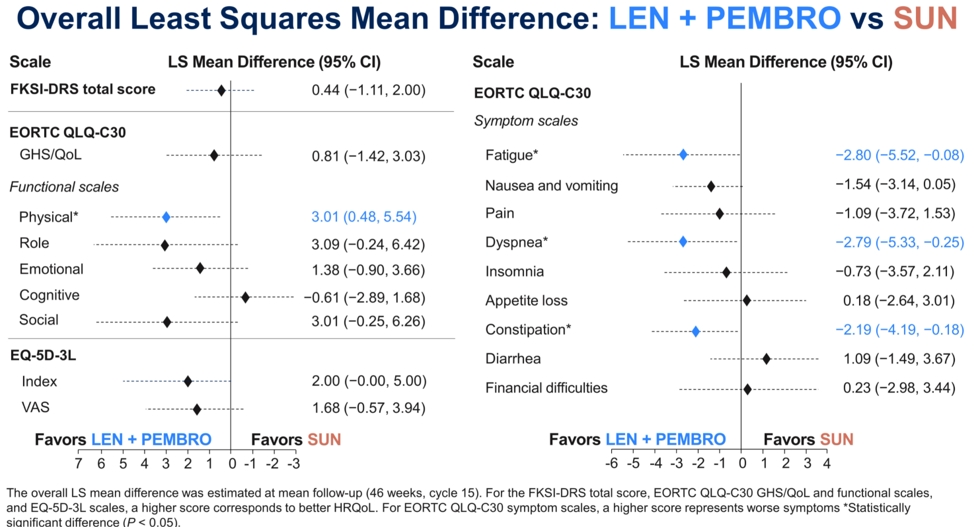
Given both the superior clinical efficacy as well as preliminary HRQoL data for len/pembro, subsequent time to deterioration analyses focused only on len/pem. Combination len/pem had superior HRQoL relative to sunitinib with regards to physical functioning, dyspnea, appetite loss, and the EQ visual analog scale (VAS).

With regards to time until definitive deterioration, len/pem was superior to sunitinib across almost all domains tested.

Dr. Motzer concluded that combination lenvatinib and pembrolizumab demonstrated similar or improved HRQoL and disease-related symptoms scores compared with sunitinib. In contrast, the combination of lenvatinib and everolimus resulted in similar or worse HRQoL scores. Combined with the efficacy and safety data that have previously been presented from the CLEAR trial, these data support lenvatinib/pembrolizumab as a first-line treatment option for advanced RCC.
Presented by: Robert J. Motzer, MD, Kidney Cancer Section head and Jack and Dorothy Byrne Chair in Clinical Oncology, Memorial Sloan Kettering Cancer Center, New York, New York.
Written by: Alok Tewari, MD, PhD, Medical Oncologist at the Dana-Farber Cancer Institute, at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021

