To do so, the authors utilized the nationwide Flatiron Health electronic health records-derived database to identify patients diagnosed with mRCC and treated with front-line axitinib and pembrolizumab or ipilimumab and nivolumab between 2018 and 2020. They examined primary endpoints of overall survival (OS) and real-world progression-free survival (rwPFS). To account for baseline differences, the authors used propensity score-based Inverse Probability of Treatment Weighting (IPTW), accounting for age, gender, insurance, race, IMDC risk group, practice type, and nephrectomy. The authors examined survival from the initiation of systemic therapy and compared survival by treatment groups using weighted and unweighted Kaplan-Meier curves with log-rank tests and weighted Cox proportional hazards regression models.
In the Flatiron dataset, the authors identified 821 patients who received first-line axitinib and pembrolizumab (n=259) or ipilimumab and nivolumab (n= 562). The median age of the cohort was 66 years, 73% were male, and 54.9% had undergone prior nephrectomy.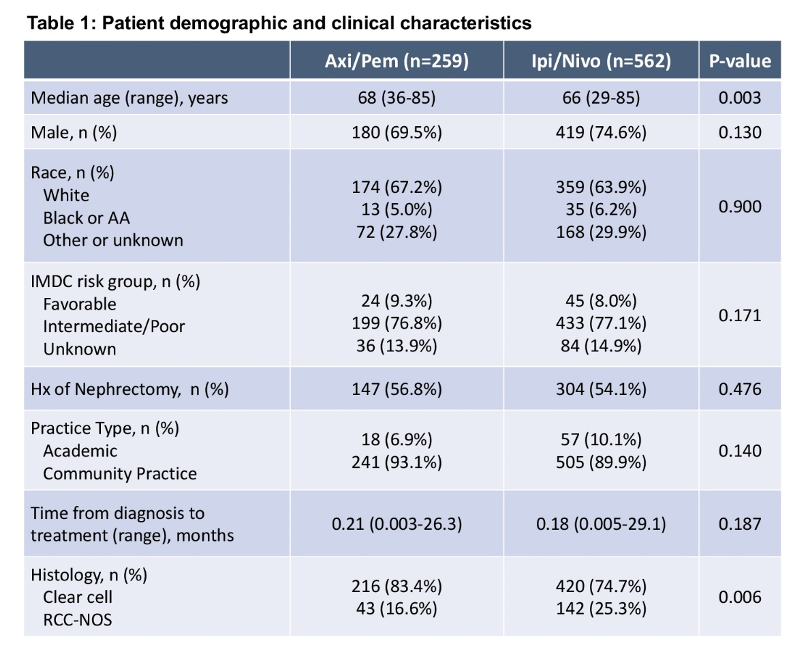
They found that the likelihood of receiving nivolumab and ipilimumab was inversely associated with increasing age at diagnosis (p=0.030) and with care in community settings (p=0.044). Gender, history of nephrectomy, insurance status IMDC risk group, and BMI were not significantly associated with treatment choice.
The authors found no significant difference in adjusted median OS between treatment groups (p=0.40): not reached for patients who received axitinib and pembrolizumab and 22 months (95% CI 19.8-NR) for those who received ipilimumab and nivolumab.
Similarly, 12-month overall survival did not significantly differ between groups (p=0.41): 68.5% for those who were treated with axitinib and pembrolizumab and 65.8% of those who were treated with ipilimumab and nivolumab. There was no difference seen across IMDC risk group, either.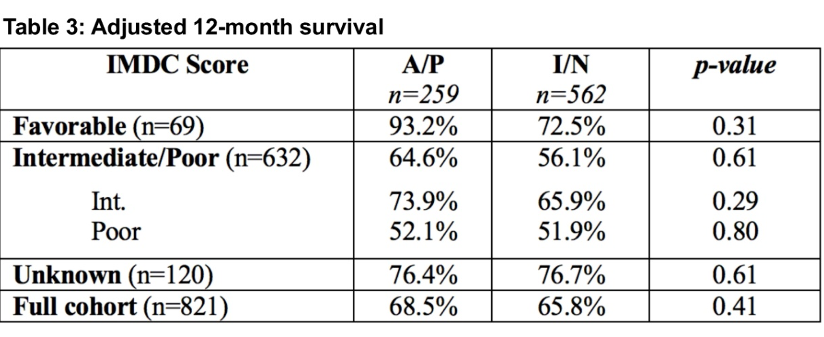
Not surprisingly, mPFS was also comparable (p=0.14): 8.4 months among those receiving axitinib and pembrolizumab and 6.8 months among those receiving ipilimumab and nivolumab.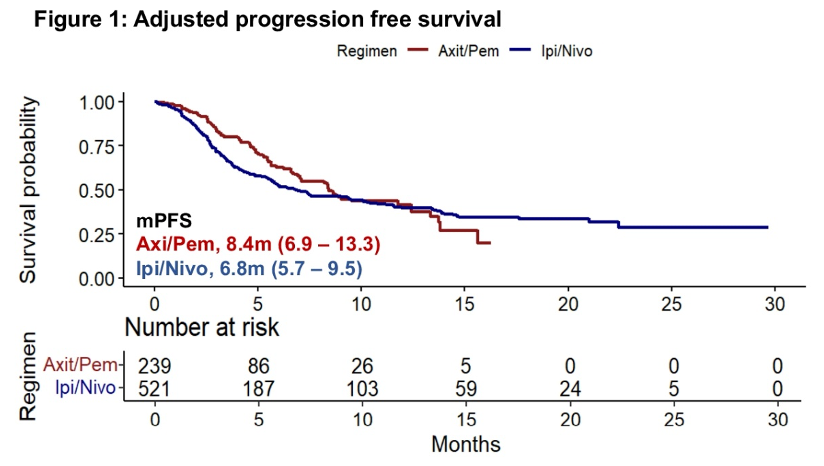
The authors conclude that, based on retrospective, real-world study, short-term survival outcomes are similar between patients treated with axitinib and pembrolizumab or ipilimumab and nivolumab. Future studies may assess more recently approved treatment approaches as well as provide longer follow-up.
Presented by: Kevin Kayvan Zarrabi, MD, Fox Chase Cancer Center, Philadelphia, PA
Written by: Christopher J.D. Wallis, MD, Ph.D., Urologic Oncology Fellow, Vanderbilt University Medical Center, Twitter: @WallisCJD at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, #ASCO21, June, 4-8, 2021

