The authors enrolled Chinese patients with cytologically or histologically confirmed RCC who had disease progression after one prior VEGFR-TKI (NCT03095040). Patients were randomized in a 1:1:1 ratio to receive matching placebo plus vorolanib or everolimus, or the combination of the two active agents. The primary endpoint was progression-free survival (PFS) with secondary endpoints including overall survival (OS), objective response rate (ORR), and safety.
The authors enrolled 399 patients between November 2016 and June 2019, with 133 patients allocated to each treatment group. As would be expected based on the randomized design, the three arms were well balanced in terms of baseline characteristics. The majority of included patients had intermediate-risk disease and the majority of included patients had visceral metastatic disease.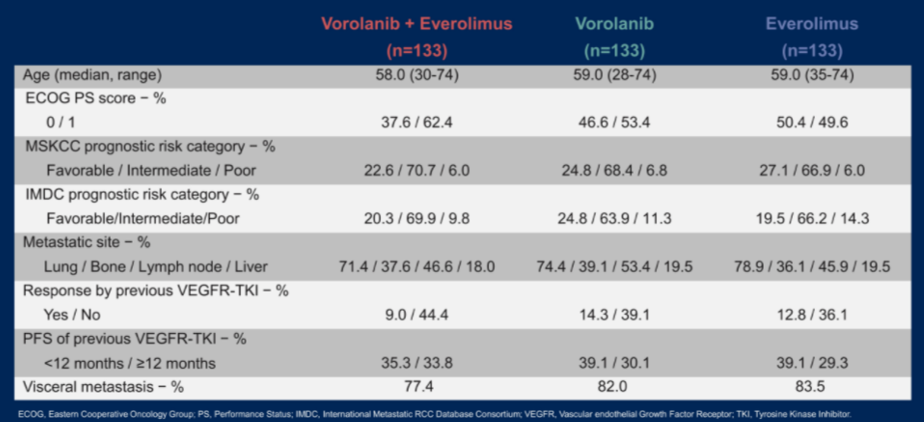
As of the data cut-off of October 23, 2020, the combination of vorolanib and everolimus had a significantly longer median PFS (10.0 months [95% CI, 8.2-10.4]) than that in the single-agent everolimus group (6.4 months [95% CI, 4.7-8.3]; HR = 0.70 [95% CI, 0.52-0.94]; p=0.017). In contrast, the median PFS was similar between single-agent vorolanib (6.4 months [95% CI, 4.6-8.3]) and single-agent everolimus (6.4 months [95% CI, 4.7-8.3]; HR = 0.94 [95% CI, 0.69-1.24]; P = 0.69).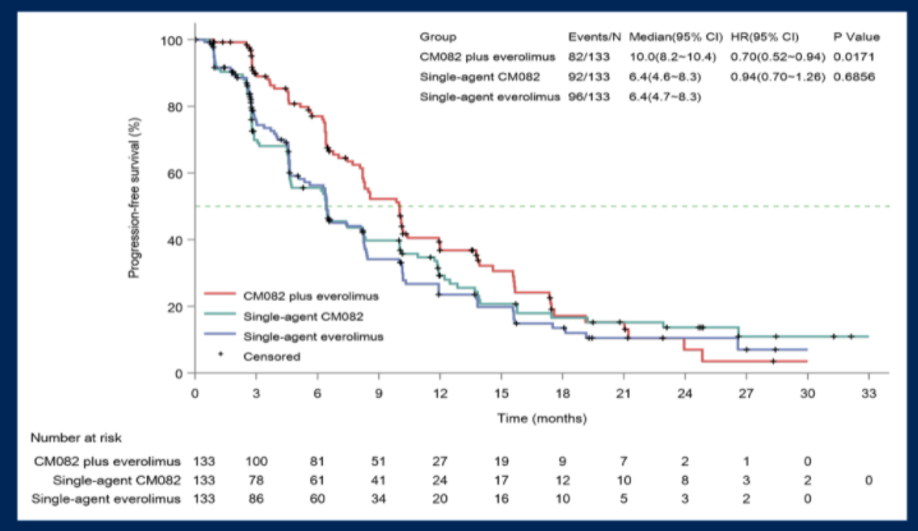
Additionally, the authors examined this effect across numerous subgroups defined on the basis of patient demographic and clinical characteristics, finding consistent results.
Assessing secondary outcomes, objective response was observed in 33 (24.8%) of 133 pts allocated vorolanib plus everolimus compared with 11 (8.3%) who received everolimus and 14 (10.5%) who received vorolanib. Duration of complete response was longer among those who received vorolanib plus everolimus (84.2 months, 95% CI 76.9-90.0) than vorolanib (66.9 months, 95% CI 58.2-74.8) or everolimus (74.4 months, 95% CI 66.2-81.6).
To date, overall survival data are immature with no significant difference between pts assigned vorolanib plus everolimus (30.4 months [95% CI, 16.5-NE]) and that allocated everolimus (25.4 months [95% CI 19.4-NE]) or vorolanib (30.5 months [95% CI, 22.8-NE]).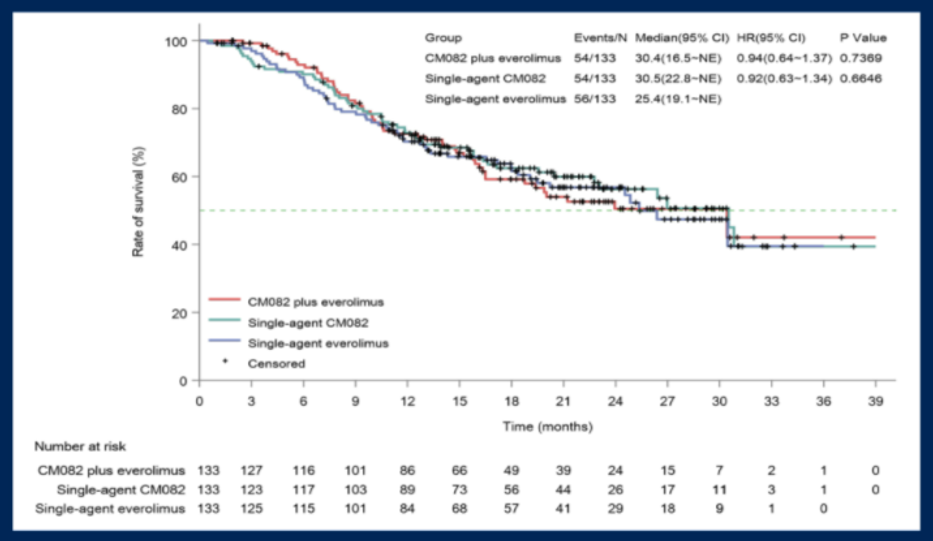
Treatment-related adverse events (TRAE) were common regardless of treatment approach, occurring in 132 (99%) patients receiving vorolanib plus everolimus, 127 (96%) receiving vorolanib, and 131 (99%) receiving everolimus. Grade 3 or higher TRAEs occurred in fewer patients who received single-agent vorolanib (52 [39%]) than in those who received single-agent everolimus (71 [53%]) or the combination of vorolanib and everolimus (96 [72%]). Treatment-related deaths occurred in 5 patients in the vorolanib plus everolimus combination arm, 4 in the vorolanib monotherapy, and 3 in the everolimus monotherapy arms.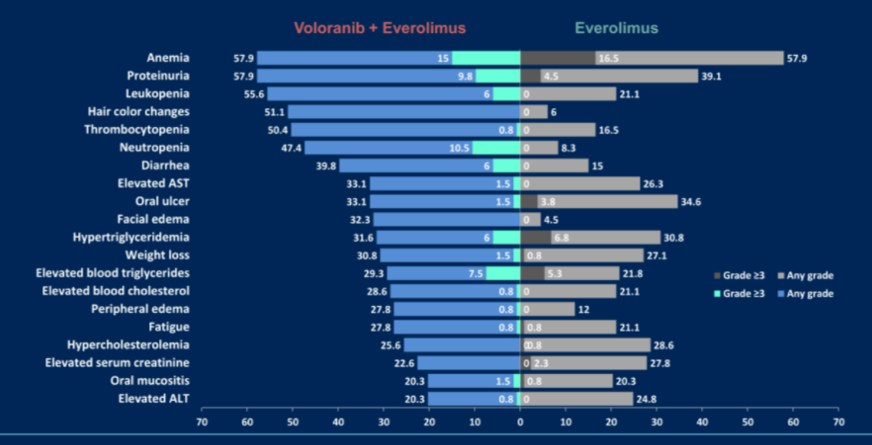
The authors conclude that this first phase 3 study of the combination of mTOR- and VEGF-targeted agents in second-line treatment of RCC demonstrates a progression-free survival benefit to the combination of vorolanib plus everolimus in patients who previously received VEGF-targeted therapy. Further, the combination of vorolanib plus everolimus was well-tolerated.
Presented by: Xinan Sheng, MD, of Peking University Cancer Hospital & Institute, Beijing, China
Written by: Christopher J.D. Wallis, MD, Ph.D., Urologic Oncology Fellow, Vanderbilt University Medical Center, Twitter: @WallisCJD on Twitter at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, #ASCO21, June, 4-8, 2021

