The authors performed an open-label, randomized trial among patients with previously untreated, confirmed clear cell or sarcomatoid mRCC who had intermediate or poor-risk disease by IMDC criteria (NCT03829111). Patients were randomized in a 2:1 fashion to receive either nivolumab and ipilimumab with CBM-588 or nivolumab and ipilimumab alone. Nivolumab and ipilimumab were dosed at 3 mg/kg and 1 mg/kg IV, respectively, every 3 weeks for 12 weeks, followed by nivolumab at 480 mg IV every 4 weeks. CBM-588 was dosed orally at 80 mg bid. Bacteriomic profiling was performed on the basis of stool samples collected at baseline and 12 weeks using metagenomic sequencing. The primary endpoint was a change in Bifidobacterium spp. from baseline to week 12 with secondary endpoints including change in microbial diversity and clinical outcomes including response rate (RR) and progression-free survival (PFS).
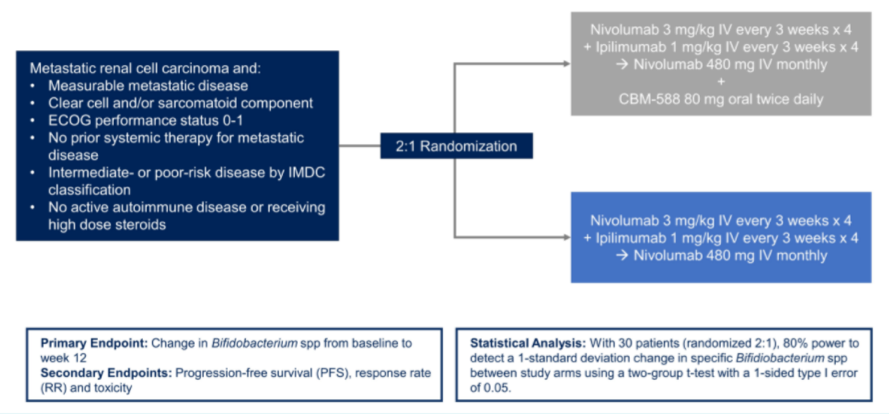
A total of 30 patients were randomized between April 2019 and Nov 2020 of whom one was subsequently excluded after genomic sequencing confirmed that they had sarcoma, rather than RCC. Among 29 evaluable patients, 21 were male, 8 were female and the median age was 66 years. In terms of disease characteristics, 10 patients (34%) had sarcomatoid features and 24 (83%) had intermediate-risk disease. 11 patients had undergone prior nephrectomy and all had at least two or more metastatic sites with lung and lymph node the most common sites.
Metagenomic sequencing showed an 8-fold increase from baseline to week 12 in Bifidobacterium bifidum and a 6-fold increase in Bifidobacterium adolescentis among those patients receiving nivolumab and ipilimumab and CBM-588.

C. butyricum was detected only among those receiving CBM-588. Further, pathogenic species including Escherichia. coli and Klebsiella spp. were more prevalent in patients who did not receive CBM-588.
In terms of clinical outcomes, RR was significantly higher among patients receiving nivolumab and ipilimumab and CBM-588 vs nivolumab and ipilimumab alone (58% vs 20%; P = 0.024).
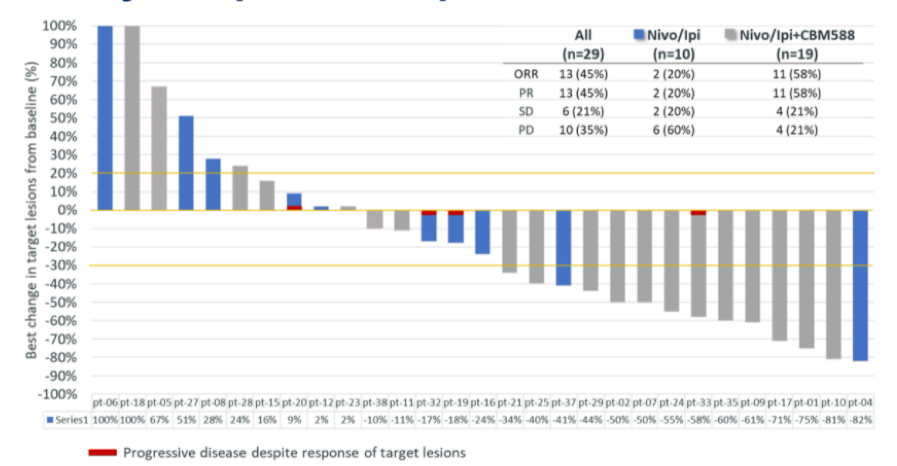
Further, median PFS was also prolonged with the addition of CBM-588 to nivolumab and ipilimumab (NR vs 11 weeks; P < 0.001). No significant difference in grade 3/4 toxicities were observed between study arms.
The authors concluded that this first randomized, prospective study of a live bacterial product in combination with immunotherapy enhances the response. However, the study is limited by the small sample size and that it was performed at a single site. Thus, further exploration is warranted.
Presented by: Luis A. Meza, MD, Postdoctoral Fellow, City of Hope
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021

