
The first study discussed by Dr. Apolo was by Dr. Brian Rini “Pembrolizumab plus axitinib versus sunitinib as first-line therapy for advanced clear cell renal cell carcinoma: Results from 42-month follow-up of KEYNOTE-426.” With a 42.8-month median follow-up and a 35.6 month minimum follow-up, the final analysis of the KEYNOTE-426 trial reported that compared with sunitinib, pembrolizumab + axitinib improved OS (median: 45.7 vs 40.1 months; HR 0.73, 95% CI 0.60-0.88) and PFS (median 15.7 vs 11.1 months; HR 0.68, 95% CI 0.58-0.80). Furthermore, the 42-month OS rate was 57.5% with pembrolizumab + axitinib versus 48.5% with sunitinib, and the 42-month PFS rate was 25.1% with pembrolizumab + axitinib versus 10.6% with sunitinib. For pembrolizumab + axitinib versus sunitinib, the objective response was 60.4% versus 39.6% (p < 0.0001), complete response rate was 10.0% versus 3.5%, and median duration of response was 23.6 months (range: 1.4+ to 43.4+) versus 15.3 months (range: 2.3-42.8+). Dr. Apolo notes that KEYNOTE-426 and CheckMate-214 both have the longest follow-up (~42 months). As follows is a table summarizing the latest data comparison of CheckMate-214, KEYNOTE-426, CheckMate-9ER, and CLEAR:

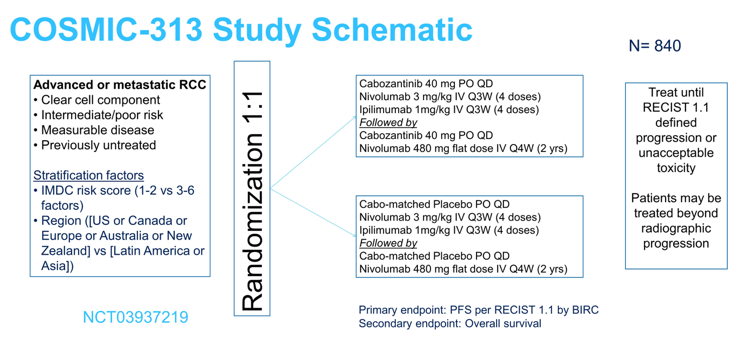
Dr. Apolo highlighted that over the next several years we will see the next generation of first-line mRCC trials, notably triplet trials such as the COSMIC-313 trial:
Given the wealth of combination therapy options in the first-line setting, Dr. Apolo highlighted several important considerations moving forward:
- The importance of treatment free-survival, incorporating therapy stopping options within clinical trials
- Assessing subsequent therapies, noting how many patients are moving on to second-and third-line therapies
- Assessing treatment tolerance, grade 3/4 events, and dose holdings/reductions/discontinuation
- Analyzing the health-related quality of life outcomes
The second study discussed by Dr. Apolo was by Dr. Robert Motzer “Health-related quality-of-life analysis from the phase 3 CLEAR trial of lenvatinib + pembrolizumab or everolimus versus sunitinib for patients with advanced RCC.” For comparisons of lenvatinib + pembrolizumab versus sunitinib, overall changes from baseline at mean follow-up (week 46) favored lenvatinib + pembrolizumab with significant differences between treatments for physical functioning (least-squares mean difference: 3.0, 95% CI 0.5, 5.5) and fatigue (least-squares mean difference: −2.8, 95% CI −5.5, −0.1), dyspnea (least-squares mean difference: −2.8, 95% CI −5.3, −0.3), and constipation (least-squares mean difference: −2.2, 95% CI −4.2, −0.2). Least squares mean difference of the FKSI-DRS total score was 0.2 (95% CI −0.4, 0.7). A summary of these findings is as follows:
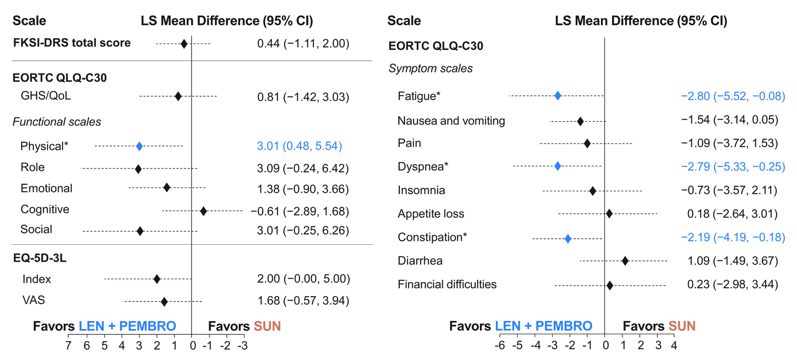
For comparisons of lenvatinib + everolimus versus sunitinib, overall changes from baseline at week 46 favored sunitinib with significant differences in overall HRQoL (least-squares mean difference: −2.8, 95% CI −5.1, −0.5 assessed by the EORTC QLQ-C30 GHS/QoL scale) and pain (least-squares mean difference: 2.8, 95% CI 0.1, 5.5), appetite loss (least-squares mean difference: 4.2, 95% CI 1.3, 7.1), and diarrhea (least-squares mean difference: 5.3, 95% CI 2.6, 7.9). Least squares mean difference of the FKSI-DRS total score was −0.4 (95% CI −1.0, 0.2). A summary of these findings is as follows:

When looking at CheckMate-214, KEYNOTE-426, CheckMate-9ER, and CLEAR, HRQoL is quite comparable between intervention and control arms for CLEAR and KEYNOTE-426, however, both CheckMate-214 and CheckMate-9ER show improved HRQoL for nivolumab + ipilimumab and nivolumab + cabozantinib, respectively:

The third study discussed by Dr. Apolo was by Dr. Nazir Tannir “CANTATA: Primary analysis of a global, randomized, placebo-controlled, double-blind trial of telaglenastat + cabozantinib versus placebo + cabozantinib in mRCC patients who progressed on immune checkpoint inhibitor or anti-angiogenic therapies.” Dr. Apolo notes that pre-clinically, telaglenastat synergized with cabozantinib, a VEGFR2/MET/AXL inhibitor, against RCC tumors utilizing dual inhibition of glucose and glutamine metabolic pathways. In a phase 1 study, telaglenastat + cabozantinib showed encouraging safety/efficacy as >=second-line therapy for mRCC with an objective response rate of 50% and 100% disease control rate. For CANTATA, patients were randomized 1:1 to receive cabozantinib (60 mg PO QD) with either telaglenastat (800 mg PO BID) or placebo.
There were 444 patients randomized (221 to telaglenastat + cabozantinib; 223 to placebo + cabozantinib). Baseline characteristics were balanced between arms, including 67% with IMDC intermediate-risk disease in the telaglenastat + cabozantinib arm and 68% in the placebo + cabozantinib arm. Prior nivolumab + ipilimumab was received by 29% of patients in each arm. The median follow-up for the trial was 11.7 months, and 276 patients received prior immune checkpoint inhibitors. Median PFS was 9.2 months for telaglenastat + cabozantinib versus 9.3 months for placebo + cabozantinib (HR 0.94, 95% CI 0.74-1.21). The overall response rate (confirmed) was 31.2% with telaglenastat + cabozantinib versus 27.8% placebo + cabozantinib. In a prespecified subgroup analysis in patients with prior immune checkpoint inhibitor, median PFS was numerically longer with telaglenastat + cabozantinib than placebo + cabozantinib (11.1 versus 9.2 months, respectively; unstratified HR 0.77, 95% CI 0.56, 1.06). Dr. Apolo notes that the previously published phase 3 METEOR trial1 also looked at outcomes based on prior therapy.2 For patients treated with prior checkpoint inhibitor therapy, median PFS was not reached for cabozantinib compared with 4.1 months for everolimus (HR 0.22; 95% CI 0.07–0.65):

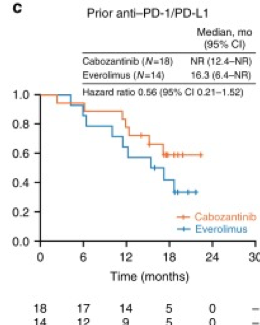
Similarly, median OS was not reached for cabozantinib versus 16.3 months for everolimus (HR 0.56; 95% CI 0.21–1.52), albeit this was not statistically significant:
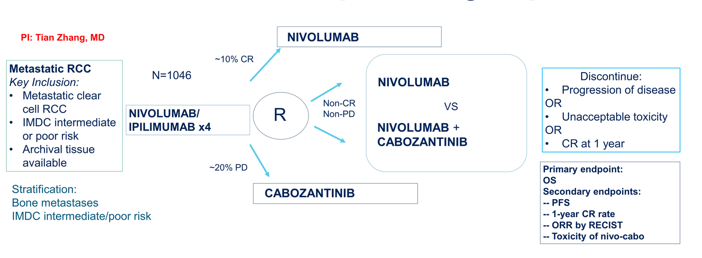
We await the results of the PDIGREE NCI cooperative group trial and its adaptive design based on response to initial therapy:
Dr. Apolo concluded her discussant presentation of these three clinical trials with the following take-home messages:
- With a median follow-up of 42.8 months in the KEYNOTE-426 study of axitinib + pembrolizumab, combination therapy maintained a survival benefit (HR 0.73), PFS benefit (HR 0.68) and objective response rate benefit (60% vs 40%) over sunitinib
- HRQoL analyses in the CLEAR study were exploratory and showed that the combination of lenvatinib + pembrolizumab has superior efficacy compared to sunitinib but there were no differences in HRQoL outcomes between the arms
- Telaglenastat added to cabozantinib did not improve PFS/OS/ORR efficacy of second/third-line cabozantinib in patients with mRCC in CANTATA, and this study provides a valuable dataset of cabozantinib therapy in the second/third-line setting after checkpoint inhibitor therapy
Presented by: Andrea B. Apolo, MD, Genitourinary Malignancies Branch, NCI, NIH, Bethesda, MD
References:
- Choueiri TK, Escudier B, Powles T, et al. Cabozantinib versus Everolimus in Advanced Renal-Cell Carcinoma. N Engl J Med2015;373(19):1814-1823.
- Powles T, Motzer RJ, Escudier B, et al. Outcomes based on prior therapy in the phase 3 METEOR trial of cabozantinib versus everolimus in advanced renal cell carcinoma. Br J Cancer.2018 Sep;119(6):663-669.

