However, it is important to remember that platinum-based chemotherapy is the backbone of first-line therapy in metastatic urothelial carcinoma:
• Gemcitabine-cisplatin: has a median overall survival of 14 months and an objective response rate of 49%
• Dose-dense MVAC: has a median overall survival of 15 months and objective response rate of 70%
• Gemcitabine-carboplatin: has a median overall survival of ~13 months and an objective response rate of 43%
• Only a minority of patients receive second-line therapy in metastatic urothelial carcinoma
• There is an unmet need to improve survival with first-line treatment
Previous studies have suggested that gemcitabine-cisplatin + drug X is not better than gemcitabine-cisplatin alone in phase 3 trials to date. This includes the EORTC 30987 trial assessing gemcitabine-cisplatin + paclitaxel versus gemcitabine-cisplatin1 and the recently published CALGB 90601 trial assessing gemcitabine-cisplatin + bevacizumab versus gemcitabine-cisplatin + placebo2:

Furthermore, first-line chemotherapy + immunotherapy did not improve overall survival (OS) compared to chemotherapy alone, including data from the IMvigor 130 trial3, which showed a progression-free survival (PFS) improvement that wasn’t clinically meaningful (and no OS benefit so far), and KEYNOTE-361 trial, with no PFS or OS improvement4.
The first trial discussed by Dr. Gupta was by Dr. Monty Pal “A randomized phase II study comparing cisplatin and gemcitabine with or without berzosertib in patients with advanced urothelial carcinoma.” Dr. Gupta notes that ATR kinase is a major regulator of DDR and is a potential therapeutic target in cancer and has shown promise in ovarian cancer and small cell lung cancer (SCLC). There is the rationale for combination therapy as (i) cisplatin and gemcitabine induce DNA damage, (ii) berzosertib can regulate DNA repair, and (iii) preclinical synergy of berzosertib with gemcitabine and cisplatin. Unfortunately, this was a completely negative study. Median PFS was 8.0 months for both arms (Bajorin risk-adjusted HR 1.17, 95% CI 0.69-1.98):
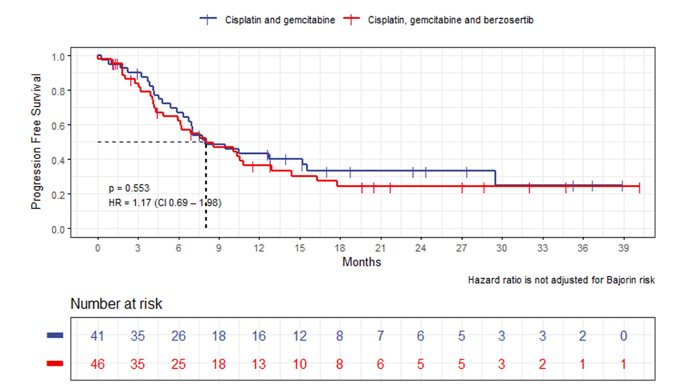
RR was 54% (4 complete responses, 21 partial responses) in the cisplatin + gemcitabine with berzosertib arm and 63% (4 complete responses, 22 partial responses) in cisplatin + gemcitabine alone arm (p = 0.66). Additionally, median OS was shorter with cisplatin + gemcitabine with berzosertib as compared to cisplatin + gemcitabine alone (14.4 versus 19.8 months; Bajorin risk-adjusted HR 1.33, 95% CI 0.71-2.48):

Additionally, the median cumulative cisplatin dose in the experimental arm was 250 mg/m2, as compared to 370 mg/m2 in the control arm (P < 0.001). The authors concluded that:
• The addition of berzosertib to cisplatin + gemcitabine did not prolong PFS in patients with metastatic urothelial carcinoma
• The addition of berzosertib appeared to augment hematologic toxicities, most notably causing higher rates of thrombocytopenia
• More frequent dose reductions were required on the berzosertib arm, which used attenuated doses of both cisplatin and gemcitabine at baseline
• Cumulative doses of cisplatin were significantly lower on the berzosertib arm
• Compromised delivery of cytotoxic therapy may account for the concerning trends in OS observed in this study
Dr. Gupta notes that these findings are in line with single-agent PARP inhibitor therapy not showing an efficacy benefit in metastatic urothelial carcinoma based on the ATLAS trial of rucaparib in unselected metastatic urothelial carcinoma patients.
Dr. Gupta’s take-away points from this trial are as follows:
• Cisplatin doses should not be compromised in metastatic urothelial carcinoma
• PARP inhibitors and ATR inhibitor have not shown promise in metastatic urothelial carcinoma
• DDR pathway appears to be prognostic and predictive in metastatic urothelial carcinoma, but not promising as a therapeutic target in metastatic urothelial carcinoma so far
The second trial discussed by Dr. Gupta was “First-line pembrolizumab in cisplatin-ineligible patients with advanced urothelial cancer: Response and survival results up to five years from the KEYNOTE-052 phase 2 study.” Indeed, the evolution of first-line therapy in cisplatin-ineligible metastatic urothelial carcinoma has been busy over the last few years:

Dr. Gupta notes that when KEYNOTE-052 is compared to KEYNOTE-361, the response rate is lower with pembrolizumab compared to gemcitabine + carboplatin:

Additionally, Dr. Gupta notes that overall survival for pembrolizumab catches up but does not cross significantly enough for a positive trial:

Putting things into perspective, Dr. Gupta highlights that data from the IMvigor 130 [3] and JAVELIN Bladder 1005 trials shows that PD-L1 seems to predict atezolizumab responses in exploratory analyses. Dr. Gupta’s key takeaway messages from this trial are (i) that there is a lower response rate and more early deaths with pembrolizumab compared to gemcitabine-carboplatin in KEYNOTE-361, (ii) PD-L1 was not predictive of benefit to pembrolizumab in KEYNOTE-361, and (iii) the field has moved forward with avelumab approval after first-line platinum-chemotherapy in non-PD patients, (iv) pembrolizumab use in cisplatin-ineligible patients with locally advanced/metastatic urothelial carcinoma should be limited to a select population not eligible to receive carboplatin.
The third trial discussed by Dr. Gupta was by Dr. Arjun Balar “Inducible T-cell co-stimulatory receptor agonist, feladilimab, alone and in combination with pembrolizumab: Results from INDUCE-1 urothelial carcinoma expansion cohorts.” Feladilimab is a first-in-class humanized IgG4 mAb selected for its agonist activity through the inducible T-cell co-stimulatory receptor, with low/no T-cell depleting effects. In the monotherapy expansion cohort arm of this trial, the median duration of follow-up was 10.6 months (range: 1.1–22.8); the overall response rate was 7% (1 partial response; 95% CI 0.2, 36.0) with a duration of response of 6.1 months. Disease control rate (response or stable disease for ≥9 weeks) was 21% (95% CI 4.7, 50.8), and median overall survival (OS) was 14.5 months (95% CI 3.0, NR), with 77% of patients alive at 6 mo. In the combination expansion cohort, the median duration of follow-up was 9.6 months (range: 0.9–28.3); the objective response rate was 22% (7 partial responses; 95% CI 9.3, 40.0) with a median duration of response of 8.3 months (range: 3.5–23.3+). The disease control rate was 63% (95% CI 43.7, 78.9), and median OS was 10.7 months (95% CI 5.2, 18.4), with 64% of patients alive at 6 months.

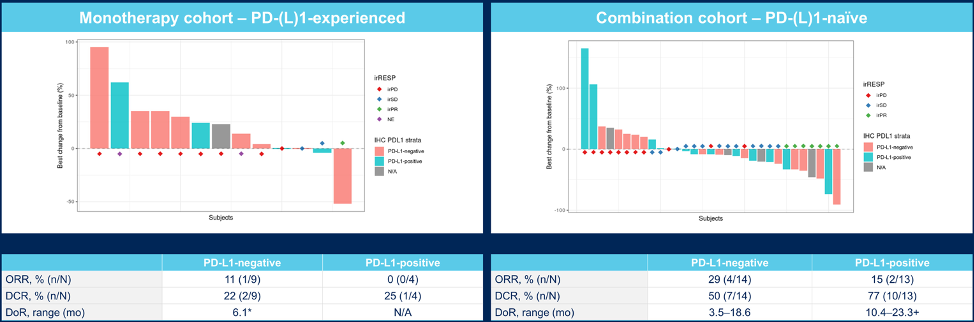
Best tumor response in PD-L1 positive subcohorts is as follows for the monotherapy and combination cohorts:
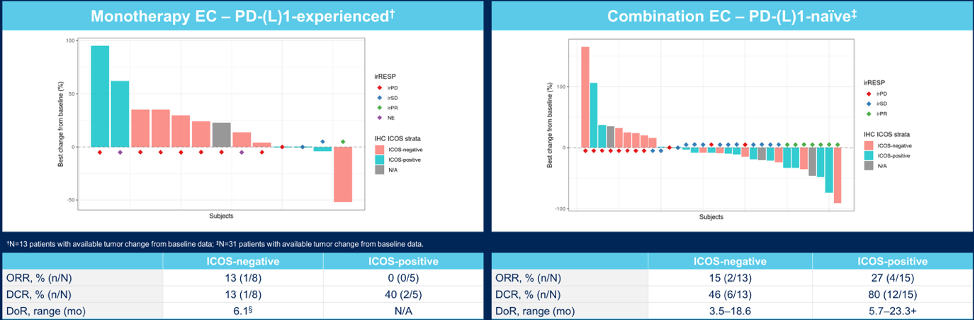
Best tumor response in inducible T-cell co-stimulatory-enriched subcohorts is as follows for the monotherapy and combination cohorts:
Grade ≥3 treatment-related adverse events were reported for 0% and 9% of patients in the monotherapy (n = 16) and combination (n = 44) safety populations, respectively. PD-L1 expression and inducible T-cell co-stimulatory-specific biomarkers are being evaluated, with promising trends observed in the enrichment of clinical activity in preliminary analyses.
Dr. Gupta’s key takeaway messages from this trial are as follows:
• Inducible T-cell co-stimulation represents a novel immunotherapy target in urothelial carcinoma
• Feladilimab monotherapy and in combination had a manageable safety profile in metastatic urothelial carcinoma in this first in human phase 1 trial with early efficacy signal
• Definitive conclusions cannot be drawn from biomarkers
• Future phase 2 trials are needed to further explore this agent in metastatic urothelial carcinoma and understand the significance of inducible T-cell co-stimulation in metastatic urothelial carcinoma
Dr. Gupta notes that the current treatment paradigm in metastatic urothelial carcinoma in June 2021 looks as follows:

However, first-line therapy in metastatic urothelial carcinoma continues to evolve, with several ongoing phase 3 clinical trials:

Dr. Gupta concluded her presentation with the following take-home messages and future directions:
• Platinum-based chemotherapy is the kingpin in first-line metastatic urothelial carcinoma and the addition of more agents is NOT better
• PD-L1 does not appear to be predictive for immunotherapy benefit
• The metastatic urothelial carcinoma landscape is evolving rapidly and traditional control arms become quickly outdated
• There is a need to design pragmatic, biomarker-based trials instead of large “me too” trials
• There is a need for harmonization of biomarkers and incorporation of artificial intelligence for predicting biomarkers of response and resistance
Presented by: Shilpa Gupta, MD, Staff Member, Department of Solid Tumor Oncology, Cleveland Clinic Taussig Cancer Institute, Cleveland, Ohio
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021
References:
1. Bellmunt J, von der Maase H, Mead GM, et al. Randomized phase III study comparing paclitaxel/cisplatin/gemcitabine and gemcitabine/cisplatin in patients with locally advanced or metastatic urothelial cancer without prior systemic therapy: EORTC Intergroup Study 30987. J Clin Oncol. 2012 Apr 1;30(10):1107-1113.
2. Rosenberg JE, Ballman KA, Halabi S, et al. Randomized phase III trial of gemcitabine and cisplatin with bevacizumab or placebo in patients with advanced urothelial carcinoma: Results of CALGB 90601 (Alliance). J Clin Oncol. 2021 May 14 [Epub ahead of print].
3. Galsky MD, Arranz Arija JA, Bamias A, et al. Atezolizumab with or without chemotherapy in metastatic urothelial cancer (IMvigor130): A multicentre, randomized, placebo-controlled phase 3 trial. Lancet. 2020 May 16;395(10236):1547-1557.
4. Powles T, Csoszi T, Ozguroglu M, et al. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): A randomized, open-label, phase 3 trial. Lancet Oncol. 2021 May 26;S1470-2045(21)00152-2.
5. Powles T, Park SH, Voog E, et al. Avelumab Maintenance Therapy for Advanced or Metastatic Urothelial Carcinoma. N Engl J Med 2020 Sept 24;383(13):1218-1230.
Related Content:
ASCO GU 2020: Rucaparib for Recurrent, Locally Advanced, or Metastatic Urothelial Carcinoma (mUC): Results from ATLAS, A Phase II Open-Label Trial

