(UroToday.com) Intermittent supraphysiologic testosterone administration in metastatic castration-resistant prostate cancer (mCRPC) is an intriguing modulatory strategy being explored to augment the efficacy of anti-androgen therapies. This strategy has its roots in case reports sixty years ago, where rare patients with metastatic prostatic cancer experienced symptomatic and lab improvements after treatment with testosterone. Subsequent small clinical studies and preclinical work suggest that supraphysiologic testosterone may uniquely target mCRPC cells that have adapted to androgen deprivation therapy by upregulating androgen receptor axis signaling. Bipolar androgen therapy (BAT) specifically refers to patients on androgen-deprivation therapy (ADT) receiving monthly injections of 400 mg of testosterone cypionate, which cause transient supraphysiologic levels of serum testosterone in excess of 1500 mg/dL, then a subsequent reduction in serum testosterone levels to castrate levels (< 50 ng/dL).
In this study, mCRPC patients on ADT that progressed on abiraterone were randomized to BAT or enzalutamide, with the option to crossover to the alternative therapy once clinical or radiographic progression was noted. The primary endpoint was to detect a difference in progression-free survival between BAT and enzalutamide therapy. Secondary endpoints included safety, radiographic response, PSA response within each arm and at crossover, PFS2 (time from initiation of therapy to progression on crossover), and overall survival.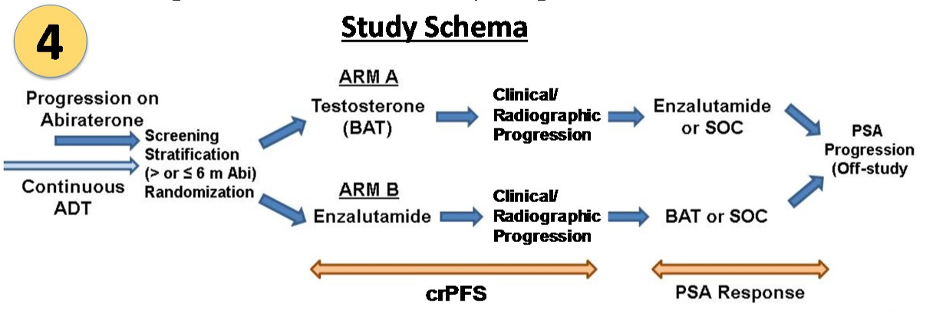
The overall response characteristics are shown below. There was a numerically higher number of objective responses in the BAT arm compared to enzalutamide, but no differences were statistically significant. In the analysis of patients that crossed over to the alternative therapy, patients who underwent BAT then received enzalutamide had improved time to PSA progression, higher rates of 50% decline in PSA, higher objective response rates, and longer PFS2.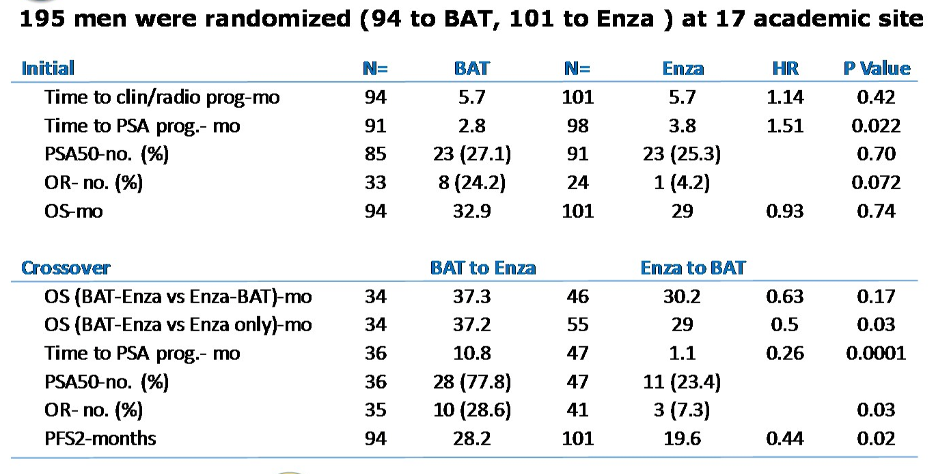
Patients in the BAT arm had favorable safety profiles, with less fatigue, insomnia, nausea, diarrhea, and hot flashes relative to patients on enzalutamide.
Overall, these data are very interesting and suggest that bipolar androgen therapy may sensitize patients who have progressed on abiraterone to derive greater benefit from enzalutamide therapy. Further investigation with larger cohorts and randomized study design as well as molecular analysis will help further delineate the efficacy of BAT in patients with mCRPC.
Presented by: Samuel R. Denmeade, MD, Professor of Oncology and Urology, Sidney Kimmel Cancer Center, Johns Hopkins University, Baltimore, Maryland
Written by: Alok Tewari, MD, Ph.D., Medical Oncology Fellow at the Dana-Farber Cancer Institute, at the 2020 American Society of Clinical Oncology Virtual Annual Meeting (#ASCO20), May 29th-May 31st, 2020
Related Content:
Watch: Testosterone Replacement in the Treatment of Advanced Prostate Cancer - Sam Denmeade and Michael Schweizer


