For PROfound, patients were randomized to olaparib tablets (300 mg bid; n=256) or physician’s choice of new hormonal agent (n=131). Patients completed the Brief Pain Inventory-Short Form (BPI–SF) questionnaire every four weeks up to six months after progression or treatment crossover. Responses were analyzed to determine time to progression to worst pain, pain severity and first opiate use for cancer-related pain, and also pain interference in daily activity.
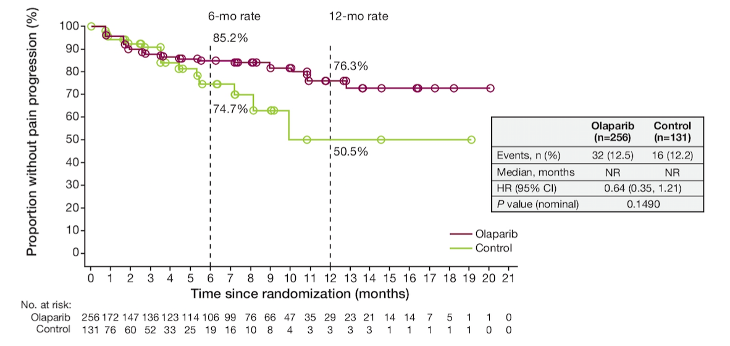
The overall BPI-SF compliance rate was high at 92.6% in the olaparib arm and 93.1% in the control arm for the overall population and 90.1% and 92.8%, respectively, for Cohort A. There were 85% of patients at 6 months and 76% of patients at 12 months taking olaparib that were free of pain progression (worst pain item) compared with 75% of patients at 6 months and 51% of patients at 12 months in the physician’s choice of new hormonal agent arm:
The proportion of patients without pain progression (overall pain severity) also favored olaparib: 12-month rate of 81.0% for olaparib vs 65.2% for physician’s choice of new hormonal agent arm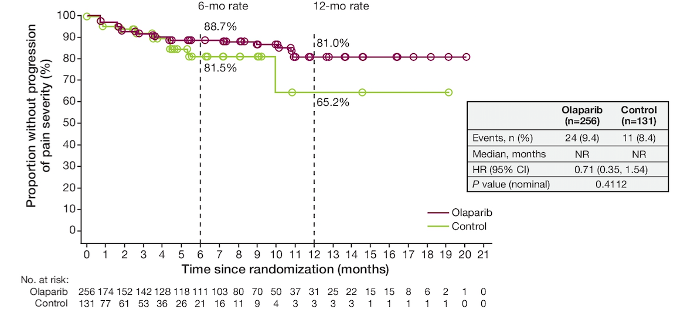
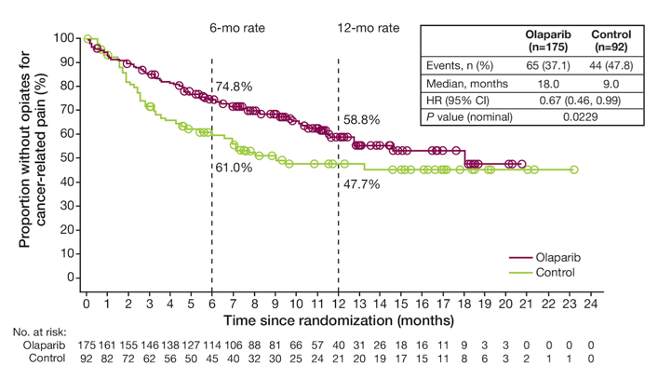
The median time to first opiate use was significantly prolonged in olaparib arm compared with physician’s choice of new hormonal agent arm: 18 months for olaparib versus 9 months for physician’s choice of new hormonal agent:
BPI-SF pain interference scores were also more favorable for olaparib than physician’s choice of new hormonal agent, with a difference in overall adjusted mean change from baseline score of −0.75 (95% CI −1.14 to −0.36) p = 0.0002:

Concluding statements for this assessment of pain burden among patients in PROfound are as follows:
- The reduced burden of pain observed with olaparib over the control treatment was manifested by a statistically significant delay in time to pain progression in Cohort A and a trend toward a delay in the overall population
- Furthermore, olaparib treatment lead to a trend toward delayed time to progression of pain severity in both the overall population and Cohort A
- A greater proportion of patients on olaparib were free of pain progression or worsening of pain severity at 6 and 12 months compared with their control-treated counterparts
- The symptomatic benefit observed with olaparib over physician’s choice of enzalutamide or abiraterone in men with mCRPC and HRR gene alterations, evidence by a reduced burden of pain, further supports the clinical benefit of improved radiographic progression-free survival
Clinical Trial Information: NCT02987543.
Presented by: Fred Saad, MD, FRCS, Professor and Chief of Urology, Director of GU Oncology, Raymond Garneau Chair in Prostate Cancer, University of Montreal Hospital Centre (CHUM), Director, Prostate Cancer Research, Institut du cancer de Montréal/CRCHUM
Co-Authors: Guilhem Roubaud, Giuseppe Procopio, Neal D. Shore, Karim Fizazi, Antoine Thiery-Vuillemin, Gabriel dos Anjos, Gwenaelle Gravis, Jae Young Joung, Nobuaki Matsubara, Daniel Castellano, Maha H. A. Hussain, Arnold Degboe, Christopher Gresty, Jinyu Kang, Joseph E Burgents, Johann S. De Bono; Dept of Medical Oncology, Institute Bergonié, Bordeaux, France; Medical Oncology Dept, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, Italy; Carolina Urologic Research Center, Myrtle Beach, SC; Institut Gustave Roussy, University of Paris Sud, Villejuif, France; Centre Hospitalier de Besancon, Besancon, France; Hospital Ernesto Dornelles, Porto Alegre, Brazil; Centre de Recherche en Cancerologie de Marseille (CRCM), Institut Paoli-Calmettes, Aix-Marseille University, Marseille, France; Center for Prostate Cancer, National Cancer Center, Goyang, South Korea; Dept of Breast and Medical Oncology, National Cancer Center Hospital East, Chiba, Japan; Hospital Universitario, Madrid, Spain; Robert H. Lurie Comprehensive Cancer Center, Northwestern University Feinberg School of Medicine, Chicago, IL; AstraZeneca, Gaithersburg, MD; AstraZeneca, Cambridge, United Kingdom; Merck & Co., Inc., Kenilworth, NJ; The Institute of Cancer Research and Royal Marsden Hospital, London, United Kingdom
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md at the 2020 American Society of Clinical Oncology virtual annual meeting (#ASCO20), May 29th-May 31st, 2020
References:
Related Content:View: PROfound Study - PARP-inhibitor Olaparib in Advanced Prostate Cancer Patients with Specific Tumor Mutations - Maha Hussain
Read: ESMO 2019: PROfound: Phase 3 Study of Olaparib vs. Enzalutamide or Abiraterone for Metastatic Castration-Resistant Prostate Cancer with Homologous Recombination Repair Gene Alterations
Read: Survival with Olaparib in Metastatic Castration-Resistant Prostate Cancer


