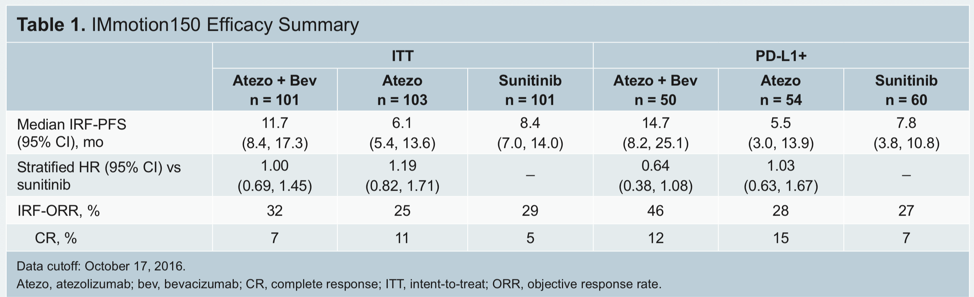
The Phase 3 IMmotion151 trial demonstrated that the combination of atezolizumab (atezo) and bevacizumab (bev) demonstrated an improved progression-free survival (PFS) compared to sunitinib (sun) alone in untreated mRCC pts, 362 (40%) of 915 patients had PD-L1 positive disease. In that study, they found that in the PD-L1 positive population, the median progression-free survival was 11.2 months in the atezolizumab plus bevacizumab group versus 7.7 months in the sunitinib group (hazard ratio [HR] 0·74 [95% CI 0·57-0·96]; p=0·0217). In the ITT population, the median overall survival had an HR of 0.93 (0·76-1·14) and the results did not cross the significance boundary at the interim analysis. The results are summarized below:
The full study protocol is seen below:


Delayed TTD of symptom severity and interference with daily life was seen with atezo vs sunitinib and atezo + bev vs sunitinib and was longest with atezo alone; the full results are seen below:

Patients reported milder symptoms and less interference during the first 6 cycles with atezo vs sunitinib: ES mean (range) was 0.36 (0.07-0.68) for core symptom severity and 0.36 (0.04-0.83) for symptom interference. The five worst patient-reported symptoms during treatment were dry mouth, fatigue, rash, drowsiness, lack of appetite – and all were in the sunitinib arm. All 16 symptoms measured were milder with atezolizumab than with sunitinib – seen below:

Clinical trial information: NCT01984242
Presented by: Sumanta K. Pal, MD, Co-director, Kidney Cancer Program, Medical Oncologist, City of Hope
Co-authors: David F. McDermott, Michael B. Atkins, Bernard Escudier, Brian I. Rini, Robert J. Motzer, Lawrence Fong, Richard Wayne Joseph, Stephane Oudard, Alain Ravaud, Sergio Bracarda, Cristina Suarez Rodriguez, Elaine Tat Lam, Toni K. Choueiri, Beiying Ding, Caroleen Quach, Kenji Hashimoto, Christina Schiff, Elisabeth Piault, Thomas Powles
Written by: Thenappan Chandrasekar, MD, Clinical Instructor, Thomas Jefferson University, @tchandra_uromd, @JEFFUrology, at the 2019 ASCO Annual Meeting #ASCO19, May 31- June 4, 2019, Chicago, IL USA
References:


