Cabozantinib is a small TKI of VEGFR2, MET and AXL, demonstrating a statistically significant improvement for mRCC patients in all three endpoints of clinical efficacy (objective response rate [ORR], progression free survival [PFS], and overall survival [OS]) in a phase III randomized trial compared to everolimus. Furthermore, this drug has shown significant clinical benefit in PFS and ORR when compared to sunitinib as first-line therapy in patients with IMDC intermediate- or poor-risk disease. Cabozantinib offers better ORR than sunitinib in 1st line (20-33%) which forecasts a higher response prior nephrectomy.
The authors present CABOPRE, which is a multicenter, non-randomized, uncontrolled phase II trial, assessing the efficacy and safety of cabozantinib as perioperative therapy in patients with advanced metastatic RCC who are appropriate candidates for cytoreductive nephrectomy.
This was a national multicenter, prospective phase 2 proof of concept study. Eligibility criteria included, age>18 years, metastatic RCC in patients suitable for cytoreductive nephrectomy. Patients must have measurable disease, with a performance status of 0,1, and have had no previous therapy for MRCC.
The study endpoints include primary ORR prior the cytoreductive nephrectomy, freedom from progression at 12 months, safety and tolerability. The trial schema is depicted in figure 1.
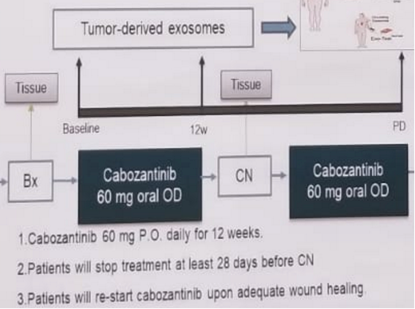
The authors plan to accrue 50 patients. ORR is planned to be assessed, using the RECIST criteria 1.1
Patients will be followed using imaging starting 12 weeks after initiation of Cabozantinib.
Tumor biopsies and peripheral blood will be collected to assess changes in tumor microenvironment at several different time points, including baseline, before cytoreductive nephrectomy, and at disease progression.
Presented by: Guillermo de Velasco, Madrid, Spain
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, Twitter:@GoldbergHanan at the 2018 ASCO Annual Meeting - June 1-5, 2018 – Chicago, IL USA


