Prior studies have established that AR activation promotes tumor growth through a feedback loop involving DNA repair factor DNA-dependent protein kinase catalytic subunit (DNA-PKcs)5,6. The activated AR promotes resolution of double strand breaks and promotes resistance to DNA damage. AR may bind to PRKCDC, the gene encoding DNA-PKcs, which induces PRKDC and further DNA-PKcs activity6. Additionally, Goodwin et al found that androgen receptor (AR) activity may be induced by DNA damage and further promotes the activation of the gene expression program governing DNA repair6. DNA-PK catalytic subunit was identified as a key player in controlling AR-mediated DNA repair and cell survival after DNA double strand damage. With regards to the mTOR pathway, pre-clinical studies have shown that prostate cancer is dependent on PI3K signaling in addition to AR signaling, and in PTEN deficient mice, combined inhibition of PI3K and AR signaling could result in near complete prostate cancer regression7.
This phase I study by Dr. Rathkopf and colleagues sought to evaluate the combination of mTOR/DNA-PK inhibition with CC-115 for patients with metastatic castration resistant prostate cancer. Finasteride, bicalutamide, and nilutamide were discontinued at least 4 weeks prior to the study. Exclusion criteria include prior exposure to enzalutamide, abiraterone, any agents targeting the PI3K/AKT pathways, and chemotherapy for CRPC. The study schema is below.

In brief, a total of 16 patients were given CC-115 plus enzalutamide. The median age was 68 in all cohorts. 63% of patients were white. All patients had an ECOG of 0. Median PSA was 9.5 ng/mL and baseline hemoglobin was 13.2 g/dL.
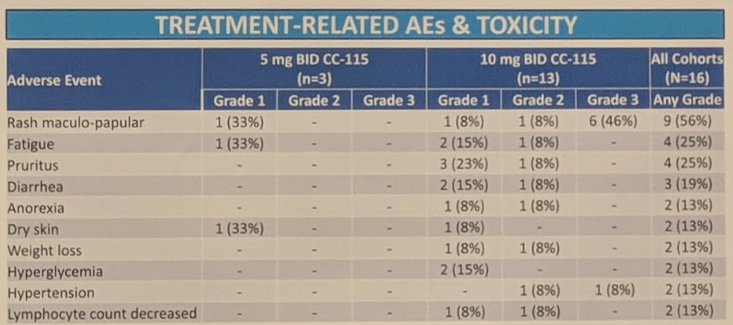
In terms of treatment-related adverse events (AE) and toxicities, rash was the most common grade 3 AE, with 46% of patients developing grade 3 rash. Due to the high observed rate of grade 3 rash, the protocol has been amended to accrue additional patients at a 7.5 mg BID dose for the dose expansion. One patient had grade 3 hypertension. There were no other grade 3 AEs observed.

In conclusion, the combination of enzalutamide and abiraterone was safe and active in men with mCRPC. There was a higher than expected incidence of rash with this combination therapy and the dose has been adjusted to mitigate this AE for the phase II expansion. It will be exciting to see what the phase II data shows!
References:
1. Scher HI, Fizazi K, Saad F, et al. Increased Survival with Enzalutamide in Prostate Cancer after Chemotherapy. New England Journal of Medicine 2012;367:1187-97.
2. Beer TM, Armstrong AJ, Rathkopf DE, et al. Enzalutamide in Metastatic Prostate Cancer before Chemotherapy. The New England journal of medicine 2014;371:424-33.
3. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration-resistant prostate cancer: the STRIVE trial. Journal of Clinical Oncology 2016;34:2098-106.
4. Li Y, Chan SC, Brand LJ, Hwang TH, Silverstein KAT, Dehm SM. Androgen Receptor Splice Variants Mediate Enzalutamide Resistance in Castration-Resistant Prostate Cancer Cell Lines. Cancer Research 2013;73:483-9.
5. Goodwin Jonathan F, Kothari V, Drake Justin M, et al. DNA-PKcs-Mediated Transcriptional Regulation Drives Prostate Cancer Progression and Metastasis. Cancer Cell 2015;28:97-113.
6. Goodwin JF, Schiewer MJ, Dean JL, et al. A Hormone–DNA Repair Circuit Governs the Response to Genotoxic Insult. Cancer Discovery 2013;3:1254-71.
7. Carver Brett S, Chapinski C, Wongvipat J, et al. Reciprocal Feedback Regulation of PI3K and Androgen Receptor Signaling in PTEN-Deficient Prostate Cancer. Cancer Cell 2011;19:575-86.
Presented By: Dana E. Rathkopf, MD, Memorial Sloan Kettering Cancer Center
Written By: Jason Zhu, MD. Fellow, Division of Hematology and Oncology, Duke University, Twitter: @TheRealJasonZhu at the 2018 ASCO Annual Meeting - June 1-5, 2018 – Chicago, IL USA


