From the 1990s the death rate from prostate cancer has been gradually declining. Death from prostate cancer usually occurs when it is in the stage of metastatic hormone-sensitive (mHSPC) or metastatic castrate-resistant prostate cancer (mCRPC), which usually occurs approximately 10-15 years from the initial diagnosis of localized disease.
Next, Dr. Oh raised the issue of patient monitoring while on systemic therapy. If we monitor these patients closely there is a potential to improve disease control by switching early to more effective therapy. It also reduces the risks of treatment-related adverse effects of ineffective therapy. Lastly, it reduces the costs of ineffective therapy. In contrast, if we decide to monitor the patient not so closely, we risk premature abandonment of effective therapy, using false-positive imaging, misinterpreting disease flare and insignificant progression. There is however an advantage of reducing the costs of imaging. Importantly, there is still no strong evidence that detecting treatment failure early is associated with improved outcomes.
Recently, the RADAR III paper was published, which is a clinician’s guide to next-generation imaging in patients with advanced prostate cancer.1 According to the RADAR III group, repeat imaging using traditional scans (bone scan and CT scan) is indicated when at least one of the following occurs:
- Every doubling of the PSA since the last imaging was performed.
- Change in symptomatology.
- Change in performance status.
- Every 6-9 months in the absence of a PSA rise.
In summary, no consensus regarding the frequency and type of imaging currently exists in mHSPC. Data from early mCRPC suggests we may be over-reliant on PSA to determine progression, as 25% of patients may progress on scans only with no PSA change. It is also not clear if the biology of recurrence is currently being altered by the earlier use of novel androgen receptor-targeted therapies.
Next, Dr. Oh discussed the side effects of ADT which are quite significant and include fatigue, hot flashes, sexual dysfunction, weight gain, decreased muscle mass, falls, increased glucose intolerance, anemia, and altered lipids. There is also an increased cardiovascular risk, cognitive changes including dementia, and osteoporotic fractures. It is currently unclear how the novel androgen receptor inhibiting therapies affect the long-term complication rate of ADT alone. These novel therapies have increased the duration of survival, and therefore increasing the duration of being treated with ADT with no clear knowledge on the anticipated effect on the ADT-complication rate.
A good example is the osteoporotic fracture rate, which has been shown to be more common in mCRPC patients taking ADT + a novel androgen receptor inhibiting treatments (Figure 1).
Figure 1 – The rate of osteoporotic fractures among mCRPC patients on ADT + additional various treatments:
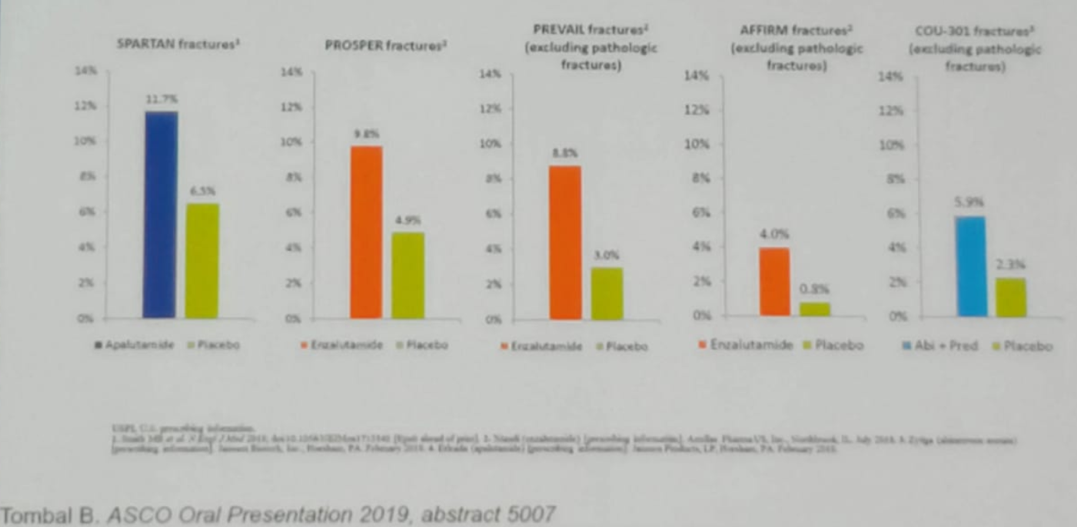
Concluding his talk, Dr. Oh sated that androgen receptor-targeted therapy has moved earlier in the disease trajectory, resulting in improved survival. This results in patients being exposed to longer durations of ADT + androgen receptor inhibitor treatments, with the ensuing increased toxicity and cost. Our goal should be to figure out how these increased complication rate may be managed while mandating the benefits of therapy on outcomes.
Dr. Oh ended his talk with a slide showing his point of view on which treatment to use at different patient scenarios (Figure 2).
Figure 2 – Which treatment first? Various possible options:

Presented by: Willian K. Oh, MD, Medicine, Hematology and Medical Oncology, Ichan School of Medicine at Mount Sinai, USA
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New-York, USA @GoldbergHanan at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 - 31, 2019 in Basel, Switzerland
References:
- Crawford ED, et al. A Clinician's Guide to Next Generation Imaging in Patients With Advanced Prostate Cancer (RADAR III). Journal of Urology. 2019 Apr;201(4):682-692. doi: 10.1016/j.juro.2018.05.164.

