(UroToday.com) The ANZUP 2021 virtual annual scientific meeting included a presentation by Dr. Sima Porten discussing the use of molecular subtypes in muscle invasive bladder cancer (MIBC) to improve patient selection for treatment. Today’s personalized medicine can be looked at through a variety of lenses:
- Disease presentation: age, gender, tumor characteristics (stage, grade, histology, hydronephrosis), social determinants of health, frailty, comorbidities, mental health, symptom burden, and risk stratification
- Treatment: prehabilitation, enhanced recovery after surgery (ERAS) programs, chemotherapy, surgery, radiation, symptom management, and coping
- Outcomes: cure (pT0/1, disease-specific survival, overall survival), crystal ball (“what can I expect?”), quality of life (ADLs, avoiding financial toxicity, “return to joy”)
Dr. Porten notes that the AUA guidelines suggest treatment options for patients that we should use in the context of comorbidities, tumor characteristics, and quality of life implications. These include: radical cystectomy + pelvic lymphadenectomy, neoadjuvant chemotherapy (cisplatin based), adjuvant chemotherapy and immunotherapy, and bladder preservation with trimodal therapy (maximal TURBT, chemotherapy plus radiation). To date, the depth and extent of tumor invasion determines the stage and treatment, irrespective of the molecular alterations or accuracy of overall clinical stage.
Dr. Porten then continued the discussion by outlining three different patients in her practice:

Patient #1 unfortunately had a pure urothelial pT3aN2(38 nodes)M0 tumor after cystectomy and had bone metastasis within six months. Patient #2 had an exceptional response to chemotherapy, pT0N0(25 nodes)M0 and is alive and well at most recent follow-up. Patient #3 declined surgery after significant side-effects from neoadjuvant chemotherapy and despite no radical cystectomy after neoadjuvant chemotherapy is alive and well with no evidence of metastatic disease. Despite similar tumors at presentation, these patients all had different courses and outcomes. To try and better understand why this may be the case, Dr. Porten notes that perhaps this has something to do with the underlying tumor biology, which may be assessable with molecular subtype signatures. Early work from the MD Anderson Cancer Center, of which Dr. Porten contributed to during her fellowship, suggested that patterns of RNA expression defined basal and luminal marker subtypes. The basal subtype tumors have an overexpression of stromal keratin and cell adhesion markers. The luminal subtype, both the p53-like and the pure luminal group are characterized by an overexpression of surface keratins. Together, the pathophysiology of luminal and basal MIBC is summarized in the following figure:
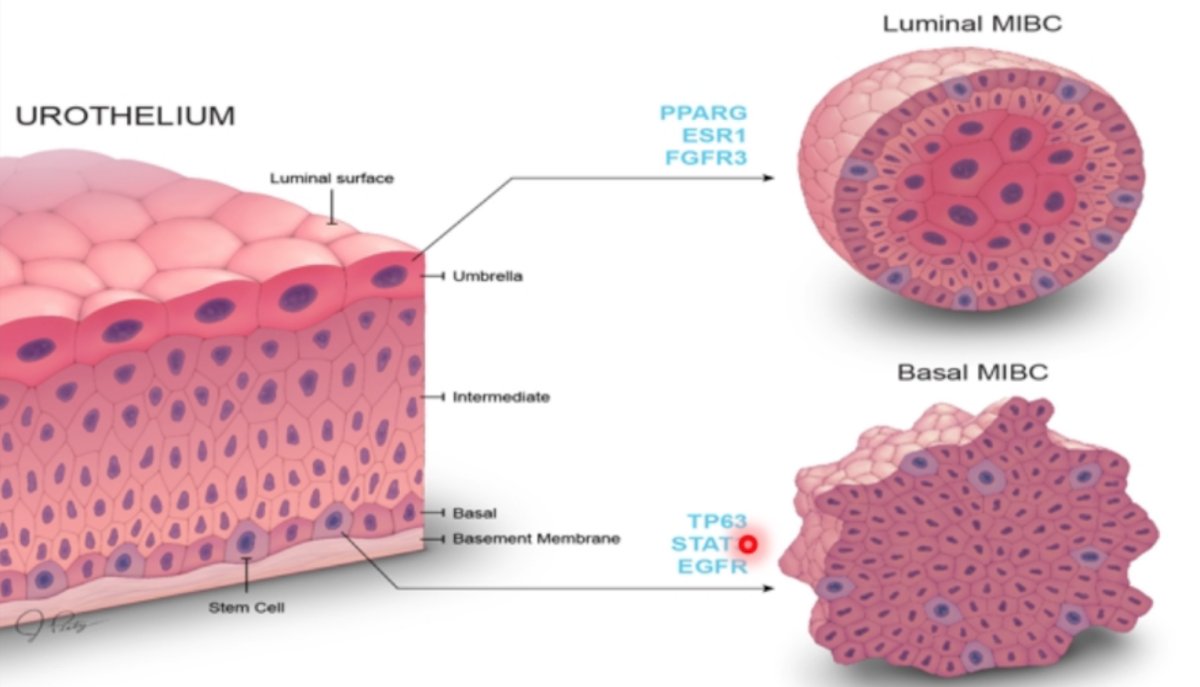
It is also possible to look at these subtypes as subtype-associated gene expression signatures, or pathway specific alterations. For example, the basal tumors have overexpression of p63 pathway genes (Stat-3, NFkB, Hif-1, p63, MYC, and EGFR), the p53-like tumors have overexpression of p53 pathway genes (p16, CDKN2a, RB1, SMARCB1, and SMAD7), and the luminal-like tumors have overexpression of PPARy pathway genes (ER/TRI, M24, FOXA1, and GATA3).
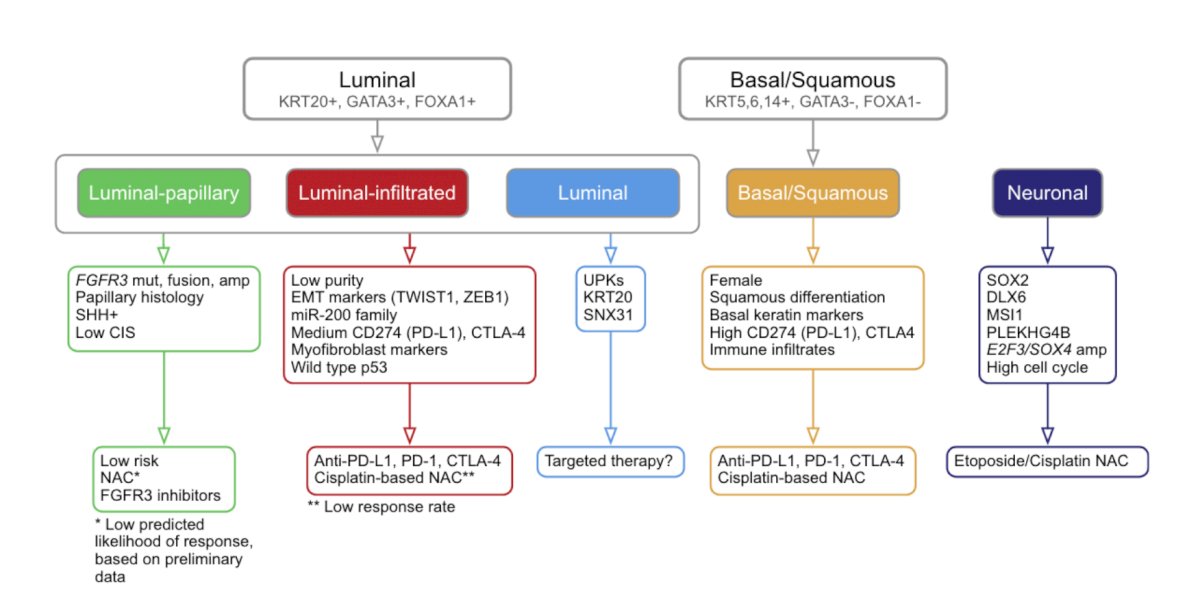
Following the work from MD Anderson was the seminal publication in Cell that transformed biomarkers in the TCGA dataset [1]. Among 412 MIBC patients, the luminal and basal subtypes were further substratified to luminal-papillary, luminal-infiltrated, luminal, basal-squamous, and neuronal:
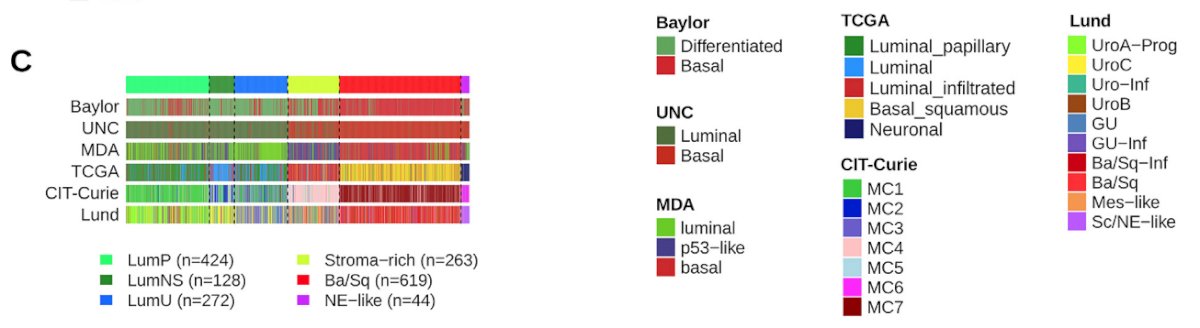
Because of the variability of the classification systems identifying different types of patients, the push was for a consensus molecular classification of MIBC, which was subsequently published in European Urology in 2020 [2]. The following figure demonstrates the relationship between subtyping results from six input classification schemes summarized in the consensus classification:
With regards to prognostication, the early work from the MD Anderson group showed that p-53-like patients had the best survival followed by luminal and the worst survival among basal-like tumors. These survival patterns were also recapitulated in the TCGA dataset with the more stratified subtyping:

Additionally, early work from MD Anderson also showed that effective neoadjuvant chemotherapy may also be able to reverse subtype related outcomes, specifically demonstrating that basal-like tumors had improved overall survival after neoadjuvant chemotherapy. Based on all of the above information, the next step was commercialization into a usable tool, which is now known as the Decipher Bladder Genomic Classifier. This classifier provides results of luminal versus basal based on TURBT tissue from patients with MIBC.
In 2017, Seiler et al. [3] investigated the ability of molecular subtypes to predict pathological downstaging and survival after neoadjuvant chemotherapy on a larger scale. Whole transcriptome profiling was performed on pre-neoadjuvant chemotherapy transurethral resection specimens from 343 patients with MIBC. Samples were classified according to four published molecular subtyping methods. A single-sample genomic subtyping classifier was used to predict consensus subtypes (claudin-low, basal, luminal-infiltrated and luminal) with highest clinical impact in the context of neoadjuvant chemotherapy. The genomic subtyping classifier was able to predict four consensus molecular subtypes with high accuracy (73%), and clinical significance of the predicted consensus subtypes could be validated in independent neoadjuvant chemotherapy and non-neoadjuvant chemotherapy datasets. Luminal tumors had the best OS with and without neoadjuvant chemotherapy, and claudin-low tumors were associated with poor OS irrespective of treatment regimen. Basal tumors showed the most improvement in OS with neoadjuvant chemotherapy compared with surgery alone.
In the neoadjuvant disease space, the PURE-01 trial tested pembrolizumab prior to cystectomy, demonstrating that 42% of patients had a pathological complete response, which was enriched in patients that were CPS positive versus those that were not CPS positive [4]. When these patients were assessed on the Decipher-based platform, the basal claudin low patients happen to have similar outcomes with or without neoadjuvant chemotherapy and excellent results with the neoadjuvant immune checkpoint inhibitor therapy.
Going back to the three patients that Dr. Porten discussed at the beginning of the presentation, the first patient’s tumor (no response to neoadjuvant chemotherapy) was Claudin low, and as such perhaps he should have received neoadjuvant immunotherapy. Patient #2 had a basal subtype tumor, which reflects his exceptional response to chemotherapy, and patient #3 (declined cystectomy after neoadjuvant chemotherapy) was luminal, so perhaps he would have done well regardless of the treatment received.
Dr. Porten then transitioned to discussing DNA alterations and RNA subtypes. In early studies lead by Dr. Plimack [5], alterations in any DNA repair genes (ATM, RB1 and FANCC) predict pathologic response and better overall survival. Furthermore, the ATM/RB1/FANCC signature did not correlate with subset assignment, but were predictive of neoadjuvant chemotherapy response and were prognostic for survival.
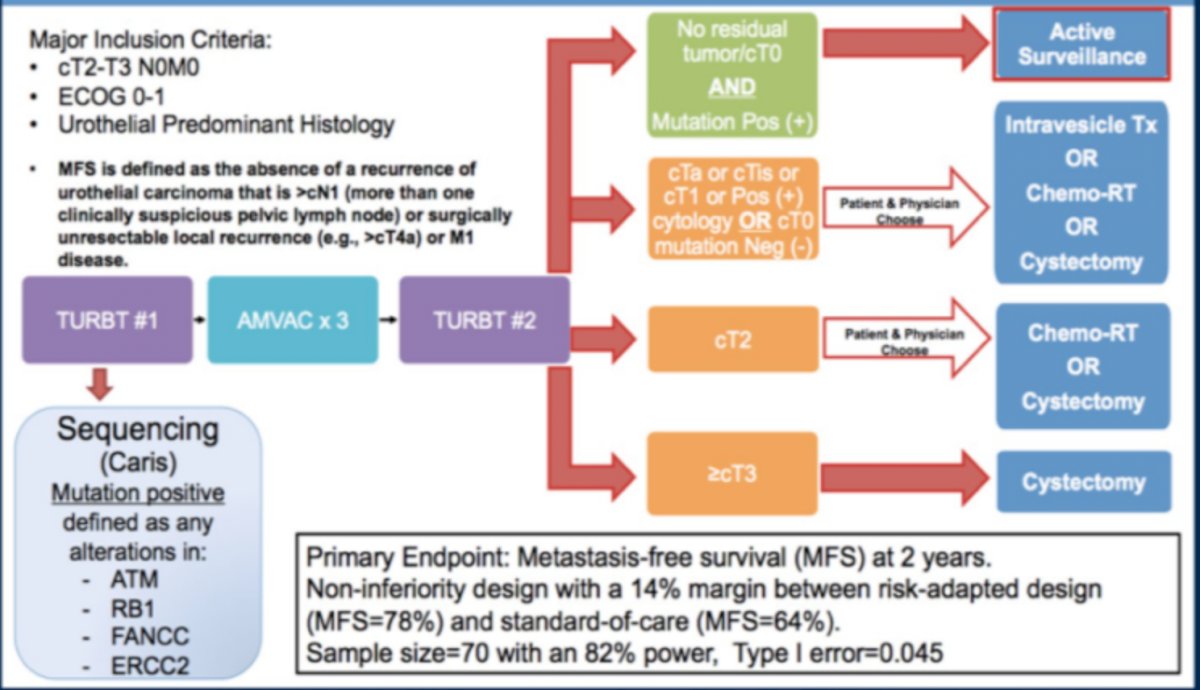
Dr. Porten notes that there is one prospective biomarker trial in MIBC, the S1314 COXEN trial. This study included patients randomized to either gemcitabine + cisplatin or dose-dense MVAC followed radical cystectomy + pelvic lymph node dissection. A COXEN score was assigned based on the regimen received, and the primary objective was to characterize the relationship of dose-dense MVAC and gemcitabine + cisplatin specific COXEN scores to pT0 rate. There were 167 patients enrolled with cT2-T4aN0M0 disease. Unfortunately, the trial did not meet its clinical endpoint, as the negative predictive value of COXEN was 50%, which was below imaging and an exam under anesthesia. However, the silver lining of this study was that the pathologic downstaging rate was 50-60%. Dr. Porten highlighted two trials currently accruing patients utilizing DNA alternations, including the A031701 trial using molecular profiling to identify MIBC patients who can be managed with definitive chemotherapy alone:

The second trial she highlighted is the RETAIN trial, with the schema noted in the following figure:
Dr. Porten concluded her presentation of moving toward better patient selection for treatment of patients with MIBC with the following concluding statements:
- The majority of “biomarkers” (both clinical and molecular) are prognostic
- Molecular subtype classification is promising and has predictive capability (with roots in biology) with a commercially available test
- Prospective biomarker trials are possible (ie. COXEN), however we need to continue cooperation/coordination and to learn from our past mistakes
- DNA damage repair alterations may have predictive capabilities and we are awaiting current clinical trial results
- We will need to be nimble and able to accommodate the changing landscape of the systemic treatment, akin to studies like I-SPY and STAMPEDE
Presented by: Sima Porten, MD, Department of Urology, University of California – San Francisco, San Francisco, CA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2021 Australian and New Zealand Urogenital and Prostate (ANZUP) Cancer Trials Group Annual Scientific Meeting (ASM), Sunday, Oct 17 – Monday, Oct 18, 2021.
References:
- Robertson AG, Kim J, Al-Ahmadie H, et al. Comprehensive molecular characterization of muscle-invasive bladder cancer. Cell 2017;171(3):540-556.
- Kamoun A, de Reynies A, Allory Y, et al. A Consensus Molecular Classification of Muscle-invasive bladder cancer. Eur Urol. 2020 Apr;77(4):420-433.
- Seiler R, Al Deen Ashab H, Erho N, et al. Impact of molecular subtypes in muscle-invasive bladder cancer on predicting response and survival after neoadjuvant chemotherapy. Eur Urol. 2017 Oct;72(4):544-554.
- Necchi A, Anichini A, Raggi D, et al. Pembrolizumab as Neoadjuvant Therapy Before Radical Cystectomy in Patients with Muscle-Invasive Urothelial Bladder Carcinoma (PURE-01): An Open-Label, Single-Arm, Phase II Study. J Clin Oncol 2018 Dec 1;36(34):3353-3360.
- Miron B, Hoffman-Censits JH, Anari F, O’Neill J, et al. Defects in DNA Repair Genes Confer Improved Survival after Cisplatin-based Neoadjuvant Chemotherapy for Muscle-invasive Bladder Cancer. Eur Urol Oncol. 2020;3(4):544-547.


