(UroToday.com) In a podium presentation at the Friday morning plenary of the 2022 American Urologic Association Annual Meeting held in New Orleans and virtually, Dr. William Aronson presented a State-of-the-Art Lecture on prostate specific membrane antigen (PSMA) Diagnostics and Theranostics.
Dr. Aronson began by highlighting the history of PSMA investigations that have led forward to the recent approvals of PSMA imaging tracers and theranostics. In the 1990s, pre-clinical studies in PSMA biology were performed at Cornell University and Johns Hopkins. However, it wasn’t until 2012 that researchers in Heidelburg Germany reported the first patient imaging using 68Ga-labelled PSMA-11. Subsequently, the FDA approved the 68Ga radiotracer in 2020 based on pivotal data from trials at UCLA and UCSF and the F-18 radiotracer in 2021 based on data from the CONDOR and OSPREY trials.
He then discussed the underlying biology of PSMA. While the function of PSMA is still unknown, PSMA is heavily overexpressed in prostate cancer cells. As PSMA is primarily extracellular, it is readily accessible for binding; it is to this extracellular component that 68Ga and F-18 radiotracers bind.
In interpreting PSMA-PET imaging, he emphasized that both 68Ga and F-18 radiotracers have similar performance characteristics. While there are number of grading systems for imaging, ultimately the performance of these tests relies upon the interpreting radiologist. He emphasized that the standardized uptake value (SUV) is a measure of uptake intensity. Both bone and lymph node tissue have low SUV levels based on low PSMA expression. This provides a favourable single to background noise ration allowing for good visualization of disease in these locations. Further, benign prostate tissue has low PSMA expression relative to prostate cancer allowing for visualization of local disease.
Dr. Aronson then highlighted clinical scenarios for the use of PSMA imaging, beginning with biochemical recurrence. Here, data from the CONDOR and UCLA/UCSF trials are relevant for F-18 and DCFPyL radiotracers, respectively. In this setting, PSMA-PET/CT has been shown to superior to conventional imaging including better sensitivity and specificity. In the CONDOR trial, the correct localization rate was 86% and nearly two-thirds of patients (64%) had a change in management. Both of these imaging approaches out-perform fluciclovine in this setting. Perhaps not surprisingly, the likelihood of radiographically detecting disease increased with increasing PSA.
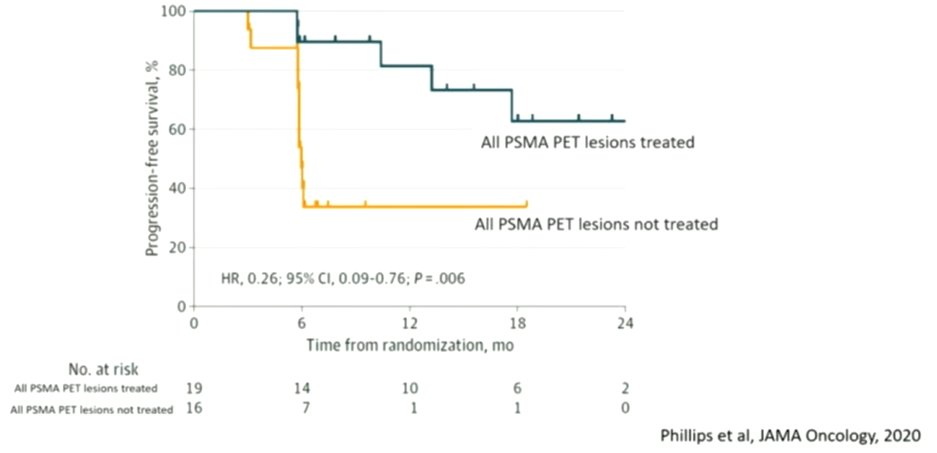
The real question though is whether use of these approaches can improve patient outcomes. To this end, ORIOLE is a phase 2 trial which randomized patients with oligometastatic disease following primary therapy to SBRT or no SBRT to the metastases. While all patients underwent PSMA imaging, radiation oncologists were blinded to these results for treatment planning. Among those patients who had all PSMA PET sites of disease treated had substantially improved progression free survival.
Next, he considered the utility of PSMA-PET in the context of pre-treatment staging, considering data from the OSPREY trial and the UCLA/UCSF trials of DCFPyL and 68Ga-PSMA PET, respectively. He highlighted that PSMA-PET has been shown to be superior to conventional imaging. While specificity is 98% with a positive predictive value of 87%, sensitivity is only 40% and negative predictive value is 83%. This low sensitivity reflects an inability of PSMA-PET/CT to identify small pelvic lymph node metastases. As result, he emphasized that we should not exclude pelvic lymph node dissection on the basis of a negative PSMA. He highlighted the role of pre-operative PSMA-PET based staging with a case of a patient with high-grade disease ho had evidence not just of nodal disease but bony metastasis in the sacrum despite negative conventional imaging.
Dr Aronson then moved to discussing PSMA-based theranostics. The principle of theranostics is based on the combination of diagnostics and therapy. As of March 2022, the FDA has approved Lu-177-PSMA-617 (lutetium) as treatment for metastatic castration resistant prostate cancer (mCRPC) who had progressed on prior therapy with novel hormonal therapies and docetaxel. Lutetium is a beta emitter – these particles travel approximately 2mm and cause primarily single-strand DNA breaks. Lutetium is linked to PSMA-617 which binds to PSMA and localizes this radiation. In the VISION trial, treatment with Lu-177-PSMA-617 was associated with improved overall survival, progression-free survival, quality of life, and pain scores.
Again, Dr. Aronson highlighted the role of PSMA-based theranostics with a case example. Unfortunately, he highlighted a recent press release from Novartis as of May 5, 2022 stating that Lu-177-PSMA-617 has temporarily been suspended due to quality control issues. However, this is expected to be resolved in the coming 6 weeks.
Dr. Aronson then discussed future directions for PSMA-PET imaging, including a potential role for utilization in patients prior to prostate cancer diagnosis. PSMA-PET imaging may be used, for example, to guide prostate biopsy for the initial diagnosis of prostate cancer. Additionally, there is an unmet need and a clinical opportunity for PSMA-PET imaging in the monitoring of treatment response and disease progression. Further, artificial imaging analysis may allow a more objective assessment of this imaging. Further, PSMA targeting may be used for intraoperative detection of lymph node metastasis during lymphadenectomy. Further, novel radiotracers and theranostic payloads (including alpha emitters) are currently being developed and studies.
In conclusion, Dr. Aronson highlighted that PSMA-PET has redefined patterns of prostate cancer spread resulting in profound transformation of prostate cancer management. To this end, the NCCN guidelines have recently been updated as of May 2022 to emphasized that PSMA-PET is equally if not more effective than conventional imaging in the front-line staging setting and detection of biochemical recurrence. As a result, conventional imaging is not required before PSMA-PET. Additionally, PSMA-PET may be considered for patients with PSA progression while on androgen deprivation therapy. Further lutetium is recognized in the NCCN guidelines as an effective treatment for mCRPC in patients who have progression following novel hormonal therapies and docetaxel.
Presented by: William J. Aronson, MD, University of California Los Angeles, Los Angeles, CA

