(UroToday.com) The 2022 Annual Meeting of the American Urological Association was host to an exhibition hosted by UroGen® and presented by Dr. Jennifer Linehan. Dr. Linehan began her presentation by highlighting that the long-term standard of care has delivered suboptimal outcomes for low-grade upper tract urothelial cancer (LG-UTUC) patients.
The gold standard treatment approach with radical nephroureterectomy (RNU) may have critical consequences. There is a significant rate of progression to renal insufficiency post-RNU, with nearly 80% of patients with UTUC having stage 3, 4, or 5 CKD post-RNU. This may limit adjuvant/salvage therapeutic options due to dose-limiting, post-operative estimated glomerular filtration rates (eGFR).

Current guidelines expand the use of kidney-sparing procedures to both imperative cases and low-risk UTUC.
- Imperative:
- Bilateral UTUC
- Solitary kidney
- Renal insufficiency
- Hereditary diseases such as hereditary non-polyposis colorectal cancer
- Low risk:
- Low-grade cytology
- Low-grade biopsy on ureteroscopy
- Unifocal, size <1.5 cm
- Non-invasive on contrast-enhanced, cross-sectional imaging
Despite kidney-sparing benefits, current endoscopic management paradigms are associated with a high rate of recurrence. Petros et al. have demonstrated overall recurrence rates of 65% of 597 low/intermediate-grade UTUC patients after a median follow up of 43 months.1
Complexity of the renal anatomy can also make endoscopic procedures more challenging, with respect to:
- Limitations for unresectable patients
- Up to 45% of patients are unresectable at presentation
- Poor visualization of tumors
- Some endoscopic techniques may cause a significant decrease in irrigation flow and thus lead to poor visualization, especially when hematuria is present
- Continued risk of progression to RNU
- Up to 33% of patients progress to RNU or ureterectomy after repeated endoscopic procedures
Mitomycin has shown promise but has been limited by upper tract physiology. Urine flow constantly dilutes and flushes out aqueous chemotherapy, which decreases dwell time. In a pharmacokinetic study of mitomycin patients, a minimum concentration of 200 ug/ml was required to penetrate the entire urothelium, and the duration of topical mitomycin exposure >200 ug/ml was approximately five minutes only.2 Sustained exposure to mitomycin makes effective chemoablation of LG UTUC tumors possible. With RTGelTM reverse-thermal hydrogel technology, JELMYTO® (mitomycin) allows for a dwell time of 4-6 hours:
- Instilled as a chilled liquid via a ureteral catheter or nephrostomy tube
- Fills and conforms to the renal pelvis, even in hard to reach areas
- Changes into a semi-solid gel at body temperature for 4-6 hours of sustained exposure to tumor-ablative chemotherapy
- Excreted via normal urinary flow
Next, Dr. Linehan went on to present the study design of the pivotal OLYMPUS trial, which was a phase 3, open-label, single arm, multicenter prospective study that evaluated six weekly induction course of mitomycin gel in patients with new or recurrent non-invasive LG UTUC. The primary endpoint was complete response (CR) and patients who achieved this outcome went on to a once monthly maintenance dose of JELMYTO for up to 12 months. 12-month durability of CR was a significant secondary endpoint.
- All patients had biopsy-confirmed LG UTUC with cytology negative for high grade disease.
- At least one papillary low-grade tumor located superior to the ureteropelvic junction had to be present
- Size: 5-15 mm
- Patients who had larger tumors could have had tumor debulking prior to treatment in order to meet the criteria
- The primary endpoint of CR was defined as:
- Complete absence of tumor lesions in the ipsilateral pyelocaliceal system at 3 months after initiation of JELMYTO
- Evaluated via urine cytology, ureteroscopy and biopsy where warranted
- Safety objective was to evaluate the safety and tolerability of JELMYTO in patients with low-grade UTUC
- Trial allowed for the instillation of JELMYTO into the pyelocaliceal system via ureteral catheters or nephrostomy tubes

Seventy-one patients were included in the intention-to-treat analysis. The study patient characteristics are displayed in the image below.
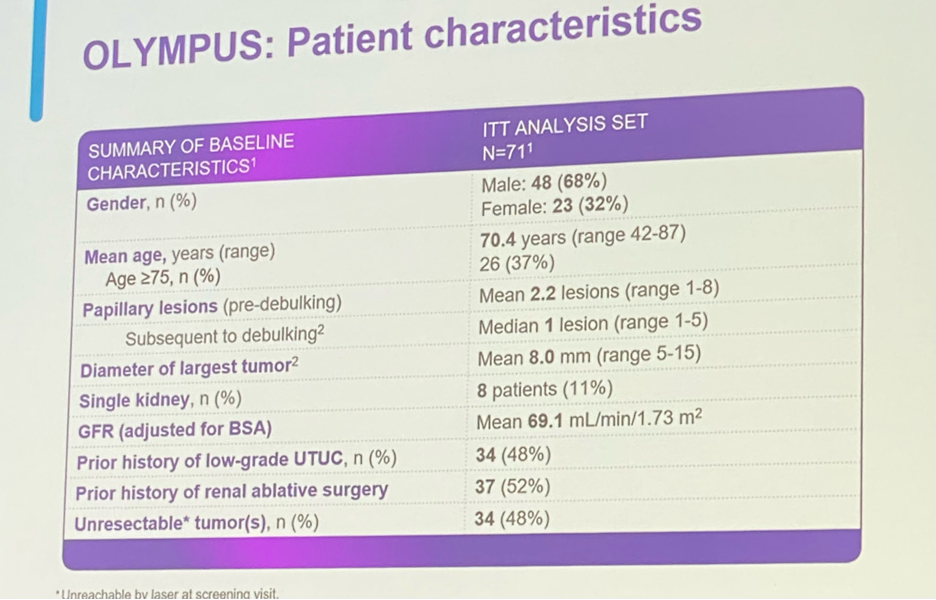
The primary endpoint of CR was achieved in 58% of patients with LG-UTUC. With regards to the remaining 42% of patients, 11% of the total patients had a partial response and 17% had no response (remaining 13% indeterminate or other).

Subset analysis evaluating JELMYTO response in patients with unresectable tumors was performed. These patients (48% of total study population) responded similarly to those in the overall population with a 59% CR rate. The secondary endpoint of 12-month durable response was achieved by 82% of all patients who achieved an initial CR.

With regards to the safety profile, any adverse reaction was encountered in >10% in patients who received JELMYTO in OLYMPUS. In addition to ureteric obstruction, urinary tract infections, vomiting, and hypertension were the most encountered grade 3 or worse adverse events.

One of the most common concerns raised by practicing urologists is the concern regarding the development of post-instillation ureteral strictures. Any grade ureteric obstruction was reported in 58% of patients receiving JELMYTO (n=41). When looking into this further, 24% of the total patients had Grade 1 or mild obstruction and the remaining 34% had Grade 2/moderate or Grade 3/severe obstruction. The definition of ureteric obstruction included: hydronephrosis, obstructive uropathy, pelvi-ureteric obstruction, ureteric obstruction, ureteric stenosis, and urinary tract obstruction. The median time to onset was 2.4 months post-completion of the six-week induction course. The important question that presents itself is: Are these ureteral strictures secondary to JELMYTO or the endoscopic instrumentation? Previous series have demonstrated stricture incidence rates ranging between 0% and 27% (median of ~10%).3,4 While less common, adjuvant administration of mitomycin and BCG was associated with stricture rates ranging from 4.4% to 15%. Thus, the higher stricture rates may be associated with repeated procedures/recurrent disease and just JELMYTO administration.

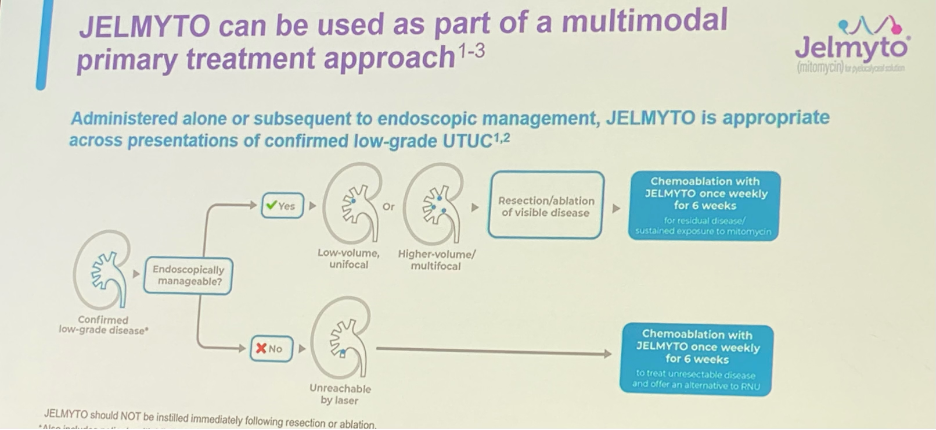
JELMYTO can be used as part of a multimodal primary treatment approach for both endoscopically manageable and non-manageable patients as detailed below:
There are certain dosing and administration considerations for JELMYTO:
- The dose of JELMYTO is 4 mg/mL, with the total instillation volume based on measurements using pyelography (not to exceed 15 mL)
- Can be administered via a Uroject12 Syringe Lever and a 5- or 7-Fr ureteral catheter or nephrostomy tube (available with the kit)
- General anesthesia is not a requirement
- Used in only 37% of patients for at least one instillation in the OLYMPUS study. Local anesthesia or sedation can then be used at discretion of the physician
- Can be administered in the clinic, ambulatory surgery center, or hospital depending on needs of the practice and patients
UroGen SupportTM provides unparalleled assistance with access, acquisition, and administrative support that are crucial to facilitating JELMYTO initiation in one’s practice, as Dr. Linehan reflected from her own real-world experience.

Dr. Linehan concluded her talk by providing two real-world experiences from her own practice, one a uninephric patient with multifocal UTUC non-resectable tumor and another with high volume, low-grade disease in the lower pole both of whom would have undergone RNU had JELMYTO not been available and both currently remain disease-free without a need for extirpative surgery.
Presented by: Jennifer A. Linehan, MD, Associate Professor, Urologic Oncology, John Wayne Cancer Institute, Santa Monica, CA
Written by: Rashid Sayyid, MD, MSc – Urology Chief Resident, Augusta University/Medical College of Georgia, @rksayyid on Twitter during the 2022 American Urological Association (AUA) Annual Meeting, New Orleans, LA, Fri, May 13 – Mon, May 16, 2022.
References:
- Petros FG, Li R, Matin SF. Urol Clin North Am. 2018;45(2):267-286.
- Au JL-S, Jang SH, Wientjes MG. J Control Release. 2002;78(1-3):81-95.
- Linehan J, Schoenberg M, Seltzer E, et al. Urology. 2021;147:87-95.
- Daneshmand S, Quek ML, Huffman. Cancer. 2003;98(1):55-60.


