(UroToday.com) In a podium presentation in the Late-Breaking Abstracts session at the 2022 American Urologic Association Annual Meeting held in New Orleans and virtually, Dr. David Crawford presented a post hoc analysis of safety and adverse events among men with metastatic hormone-sensitive prostate cancer (mHSPC) treated in the context of the ARASENS trial (NCT02799602).
ARASENS demonstrated that the triplet combination of darolutamide+androgen-deprivation therapy (ADT)+docetaxel significantly reduced risk of death by 32.5% (hazard ratio [HR] 0.675; 95% confidence interval [CI] 0.568–0.801; P <0.0001) compared to the doublet of ADT+docetaxel.

While previously reported and published, the ARASENS trial enrolled patients with mHSPC and randomized them in a 1:1 fashion to receive darolutamide 600 mg twice daily or matching placebo in addition to ADT+docetaxel. The authors examined adverse event (AE) incidences by time interval, defined as the number patient pts with new-onset AE or worsening AE in an interval (every 3 months for year 1; 6 months for year 2) divided by number of patients in that interval. For AEs specifically associated with AR pathway inhibitors, exposure-adjusted incidence rates (EAIR) are presented per 100 patients years to adjust for differences in treatment duration between groups.
The ARASENS trial randomized 1306 patients of whom 1302 patients could be evaluated for safety including 652 who received darolutamide and 650 who received placebo.
The incidence of any-grade AEs (99.5%; 98.9%), grade 3–5 AEs (70.2%; 67.5%), serious AEs (44.8%; 42.3%), and discontinuations of study treatment (darolutamide or placebo) due to AEs (13.5%; 10.6%) were comparable for the two study arms. The most frequently reported AEs were alopecia (40.5%; 40.6%), neutropenia (39.3%; 38.8%), and fatigue (33.1%; 32.9%). Perhaps not surprisingly, the incidence of AEs were highest in the first 6 months of treatment during the docetaxel treatment period and progressively decreased thereafter, in both study arms.
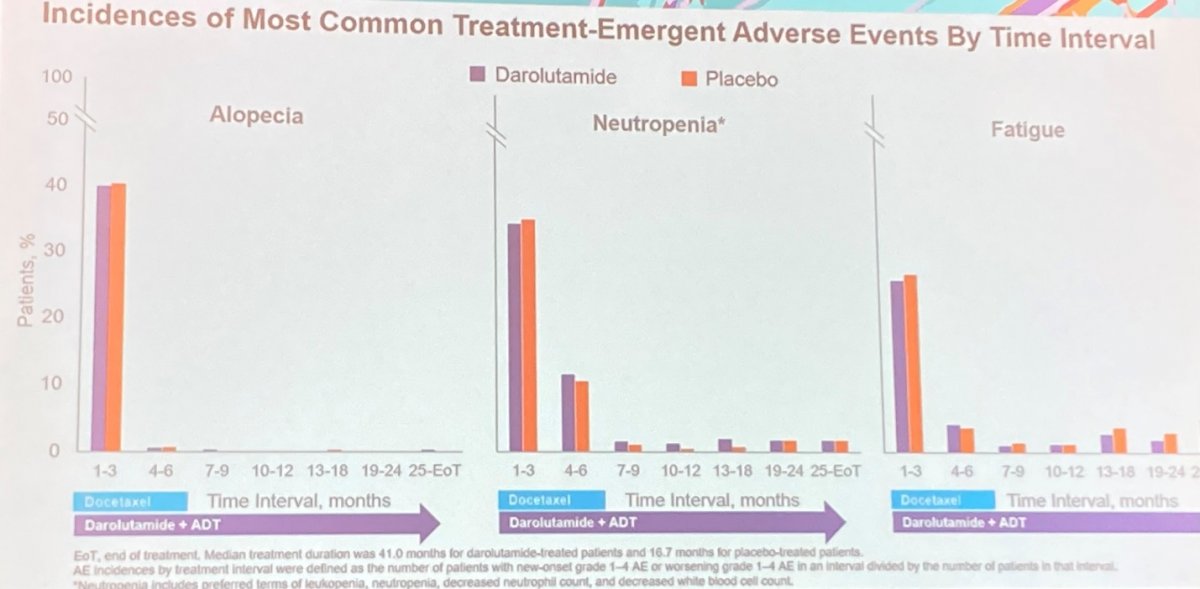
A similar pattern was observed for other common AEs (defined as those reported in >10% of patients in either treatment group), including peripheral edema, diarrhea, nausea, increased serum liver enzyme concentrations, and peripheral neuropathy.

Among AEs specifically associated with AR pathway inhibitors, most showed minimal (≤2%) difference between those patients treated with darolutamide or placebo. Both rash (16.6%; 13.5%) and hypertension (13.7%; 9.2%) occurred in more patients receiving darolutamide, though when exposure duration was considered, EAIRs were similar between groups (rash: 6.2; 7.3; hypertension: 5.1; 5.0).
Thus, Dr. Crawford concluded that these data from the ARASENS trial demonstrate that the triplet approach of darolutamide+ADT+docetaxel significantly increased overall survival without added toxicity. The highest incidence of toxicity, regardless of treatment arm, was highest in the first 6 months of overlapping docetaxel treatment, and declined thereafter.
Presented by: E. David Crawford, MD, Urologist, Professor of Urology, UC San Diego Health
Written by: Christopher J.D. Wallis, University of Toronto Twitter: @WallisCJD during the 2022 American Urological Association (AUA) Annual Meeting, New Orleans, LA, Fri, May 13 – Mon, May 16, 2022.

