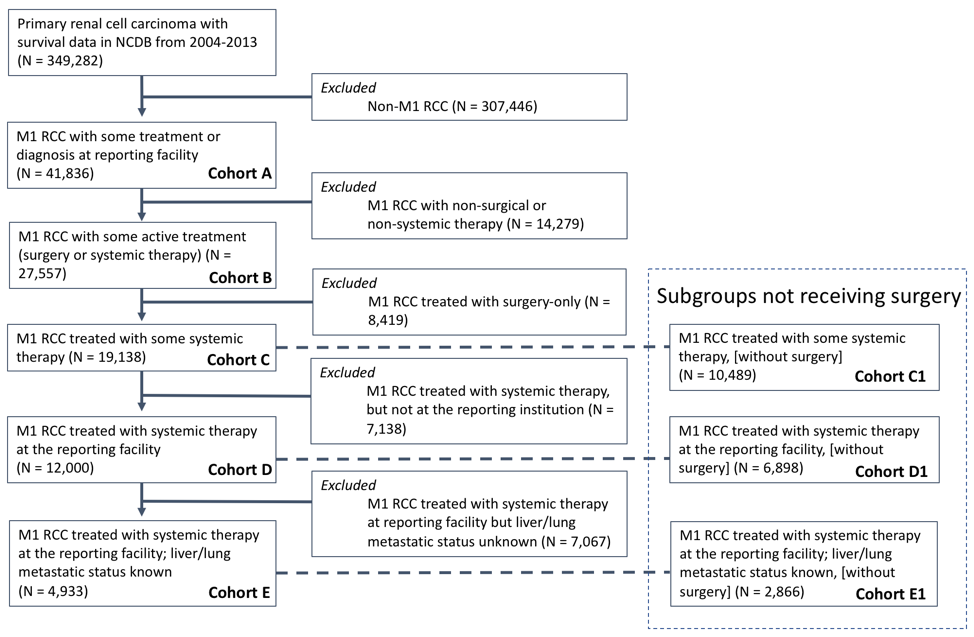
Another way of looking at the data, however, is to split TFs into high- and low-volume facilities, which we did in the supplementary materials. This may be more intuitive for clinicians as they determine the best treatment environment for their patients.
In our separate analysis, high volume TFs were defined a priori as those in the top 20th percentile of mean number of mRCC patients treated per year, which was determined to be ≥4.8 patients per year. The unadjusted median OS of all mRCC patients (cohort A) treated at high vs. low volume TFs was 9.5 vs. 6.5 months (p < 0.0001). The figure below demonstrates the Kaplan-Meier survival estimates for high and low volume TFs (cohort A and cohort E) [see cohort Figure below].

Either way you look at it, our data suggest that there is a survival benefit to being treated at higher volume facilities. Efforts to improve outcomes at lower volume facilities, especially those that treat less than 5 patients/year, are crucial to narrow this survival gap.
Read the Abstract
Written by: Shreyas S. Joshi, MD, & Daniel M. Geynisman, MD, Fox Chase Cancer Center, Temple Health, Philadelphia, PA

