While transurethral resection of the bladder tumor (TURBT) remains the gold standard ‘first step’ in bladder cancer management (both diagnosis and tumor removal), there can be a high rate of residual tumor left behind after TURBT. With some series showing as many as 34-76% of patients with a tumor on repeat TURBT at 2-6 weeks.2 Incomplete initial resection has been associated with a higher risk of tumor recurrence, despite the administration of adjuvant intravesical therapies, emphasizing the importance of the completeness of TURBT. Importantly, new technology has helped surgeons to continue to improve their TURBT techniques to achieve a more complete TURBT, which is something we need to do as a urology community to better manage the disease and improve patient care.
Updated Guidelines
Recently updated guidelines that included a joint effort between the American Urological Association (AUA) and the Society of Urologic Oncology (SUO) emphasize that a precise and thorough removal of all tumor before initiating any adjuvant therapies allows for not only early and accurate diagnosis, but proper risk stratification, and this is paramount to ensuring the optimal care of patients with bladder cancer. The importance of the completeness of TURBT is further underscored in recommendations presented at the European Association of Urology (EAU) guidelines panel for NMIBC, where Dr. Babjuk emphasized that “the most important modality in the treatment of NMIBC remains a complete and precisely performed transurethral resection of the bladder, independent of the availability of BCG on the market.”
In addition, because the clinical course of NMIBC is dynamic and variable, and often characterized by multiple recurrences and changing therapeutic interventions, the AUA/SUO guidelines recommend clinicians should classify patients as low, intermediate, or high risk of developing another tumor at each tumor recurrence. The updated guidelines are particularly notable for providing a risk-stratified clinical framework for post-surgical management and emphasizing that individualizing the care of patients prior to each treatment decision is critical.4
Furthermore, before risk stratification, a repeat TURBT, also known as a re-staging TURBT, should be considered for intermediate and high-grade tumors according to the AUA/SUO NMIBC guidelines. This allows the surgeon to ensure thorough removal of all visible tumors, and allow adjuvant therapy (e.g. BCG), the optimal milieu for activity. In addition, the risk of under staging NMIBC is significant, with up to 30% of cases being up staged to T2 at the repeat TURBT.6
Enhanced Cystoscopy
To help accomplish improved detection and resection, the new 2016 AUA/SUO guidelines recommend the use of enhanced cystoscopic techniques at the time of TURBT. Specifically, the guidelines state that clinicians should offer blue light cystoscopy (BLC), if available, as an adjunct to traditional white light cystoscopy (WLC) to increase tumor detection, decrease recurrence and improve staging (and risk stratification (Moderate Recommendation; Evidence Strength: Grade B)).4 Similar recommendations have been added to other major guidelines, including those by the National Comprehensive Cancer Care Network (NCCN), the EAU4 and the National Institute for Health and Care Excellence (NICE) in the UK. The AUA/SUO guidelines also state that clinicians may consider use of narrow band imaging (NBI) at the time of TURBT (Conditional Recommendation; Evidence Grade: C).
Hexaminolevulinic acid (HAL) the agent used with BLC is selectively sequestered in proliferating cells, leading the tumor and tumor margins to be directly identified by red fluorescence when exposed to blue light, whereas NBI identifies blood vessels, which are likely to proliferate around tumors. Hexaminolevulinate (HAL, Cysview®) is currently the only optical imaging agent approved in the US and Europe for use with BLC. A meta-analysis using raw pooled data from nine prospective trials demonstrated that BLC with HAL significantly improved the detection of bladder cancers.3
Improved identification using HAL-BLC, particularly in high grade tumors, may lead to modifications in the treatment strategy and, potentially, have an impact on management decisions.,
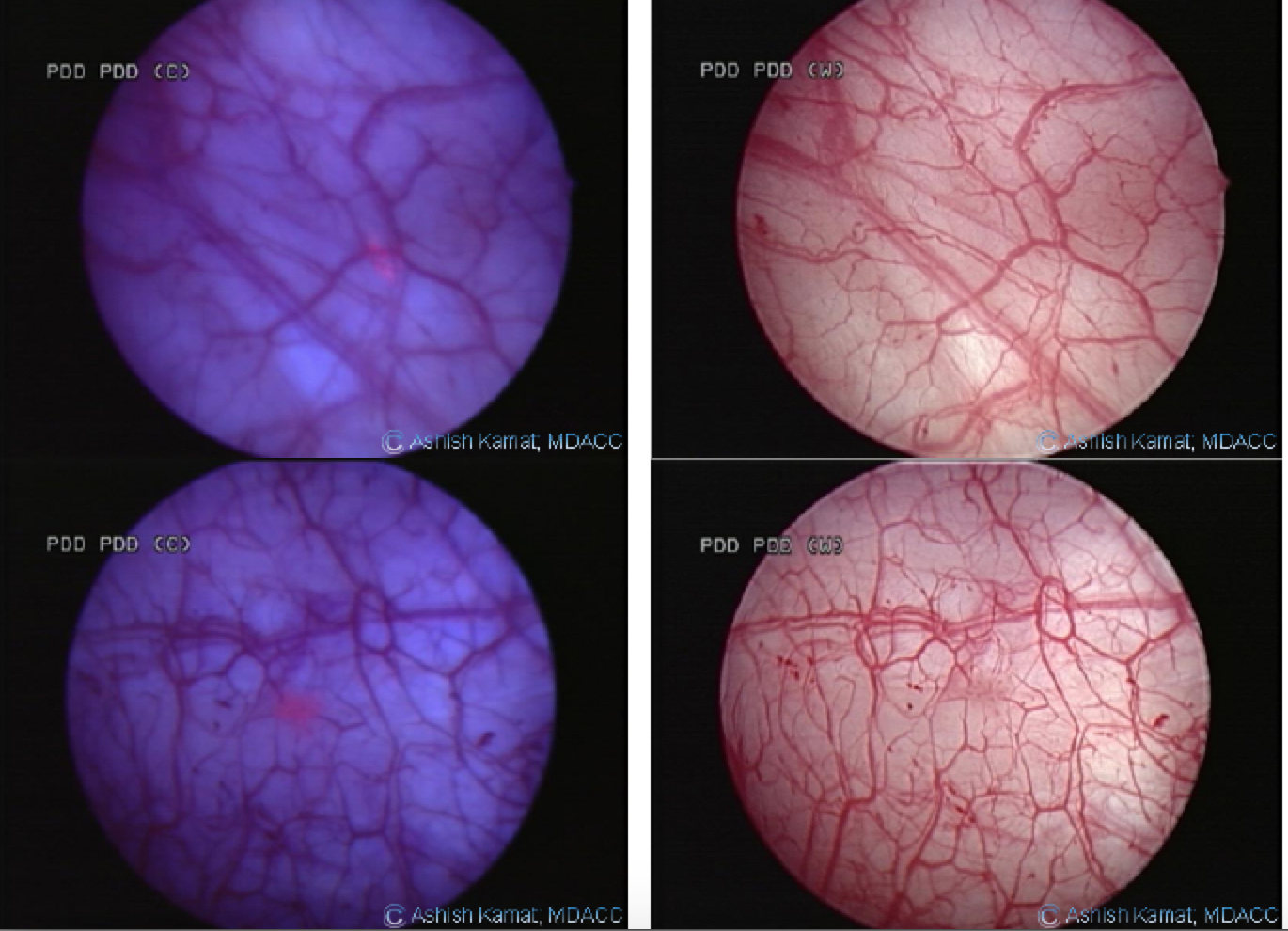
Pictured here are multiple lesions in the same patient. Shown on the right is a bladder image using white light cystoscopy alone. The image on the left shows the use of blue light cystoscopy with Cysview®
as an adjunct to white light.
Summary
A precise and thorough resection of all existing tumors remains a critical factor influencing patient outcomes, not only by reducing the incidence of under-staging, but also for providing the optimal milieu for adjuvant therapy. Enhanced cystoscopy has a critical role to play in helping urologists in this area, as recommended by the updated AUA/SUO guidelines on NMIBC.
Written by: Ashish Kamat, MD and Kamal Pohar, MD
Ashish M. Kamat, MD is a Professor of Urology and Director of Urologic Oncology Fellowship at M.D. Anderson Cancer Center. Dr. Kamat serves as a consultant and clinical trial investigator for Photocure.
Kamal S. Pohar, MD is Associate Professor of Urology at Ohio State University Wexler Medical Center. Dr. Pohar serves as a consultant and clinical trial investigator for Photocure.
References:
1. Cancer Statistics Center: Urinary bladder. American Cancer Society. https://cancerstatisticscenter.cancer.org/#/cancer-site/Urinary%20bladder. Accessed February 2017.
2. Babjuk M, Burger M, Zigeuner R, Shariat SF, van Rhijn BW, Compérat E, et al. EAU Guidelines on non-muscle-invasive bladder cancer (Ta, T1 and CIS). Eur Urol. 2016 Guidelines Edition:1-40.
3. Burger M, Grossman HB, Droller M, et al. Photodynamic diagnosis of non-muscle-invasive bladder cancer with hexaminolevulinate cystoscopy: a meta-analysis of detection and recurrence based on raw data. Eur Urol. 2013 Nov;64(5):846-54.
4. Chang SS, Boojian B, Chou R, et al. Diagnosis and treatment of non-muscle invasive bladder cancer: AUA/SUO Guideline. J Urol. 2016;196(4):1021-1029.
5. Babjuk M. How can the urologist comply with the EAU Guidelines in times of BCG shortage? In: Managing Patients with NMIBC: Therapeutic Strategies in Times of BCG Shortage Satellite Symposium. Madrid, Spain. European Association of Urology; 2015. http://medacse.ezyserver.se/halsosjukvard/wp-content/uploads/2015/06/Medac-symposierapport-EAU-2015.pdf. Accessed January 18, 2017.
6. Herr HW: Role of re-resection in non-muscle-invasive bladder cancer. ScientificWorldJournal 2011; 11: 283.
7. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) – Bladder Cancer. National Comprehensive Cancer Network. http://www.nccn.org/professionals/physician_gls/pdf/bladder.pdf. Accessed April 2016
8. Bladder Cancer: Diagnosis and Management – 1 Recommendations. National Institute for Health and Care Excellence (NICE). https://www.nice.org.uk/guidance/ng2/chapter/1-Recommendations#diagnosing-andstaging-bladder-cancer-2. Accessed April 2016.
9. Frampton JE, Plosker GL. Drugs 2006; 66: 571–578
10. Bryan RT et al. BJU Int 2007; 101: 702–706.
11. Kamat AM, Cookson M, Witjes A, Stenzl A, Grossman HB. The impact of blue light cystoscopy with hexaminolevulinate (HAL) on progression of bladder cancer: a new analysis. Bladder Cancer. 2016;2:273-278.
12. Danitchenko D, Riedl CR, Sacha MD, et al. Long-term benefit of 5-aminolevulinic acid fluorescence assisted transurethral resection of superficial bladder cancer: 5-year results of a prospective randomized study. J Urol. 2005;175:2129-2133.


