A retrospective analysis was conducted to evaluate the response to the platinum rechallenge after ICIs in patients with progressive disease (PD) less than 1 year after the first-line platinum-based chemotherapy.
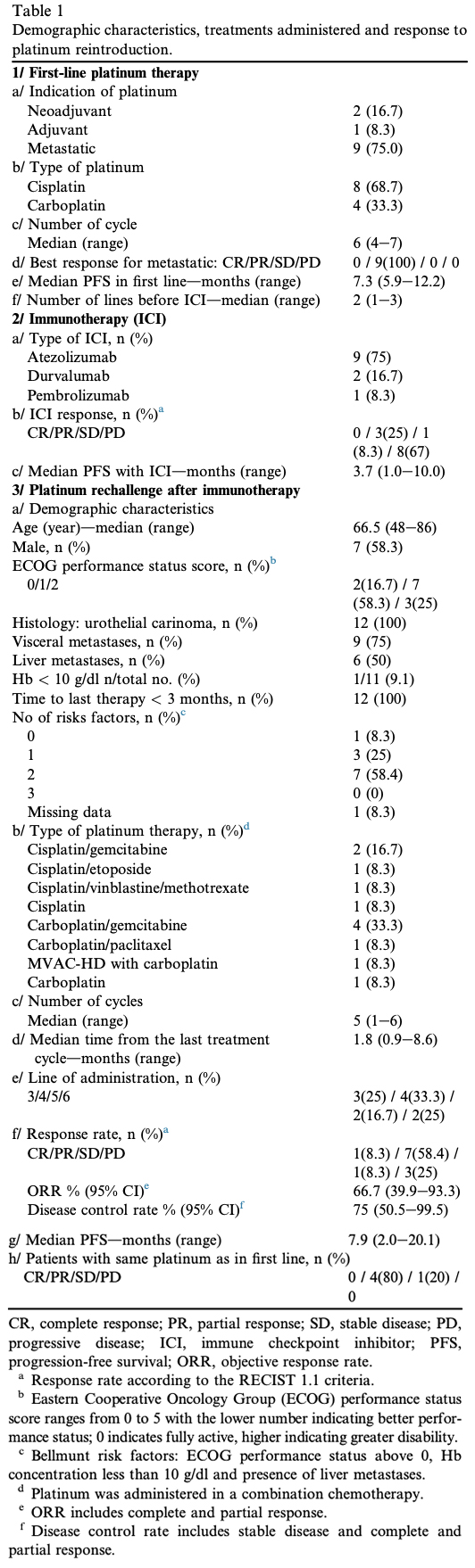
Data from 12 patients from four centres were collected. Patients’ characteristics and prior therapies are summarised in Table 1. Median progression-free survival (PFS) after first-line platinum-based treatment initiation was 7.3 months (5.9e12.2 months); all metastatic patients (75%) showed partial response to chemotherapy. Patients received ICIs in second-line treatment (41.7%). Platinum reintroduction was mainly performed as fourth-line treatment (33.3%). According to Bellmunt classification, 83.4% of patients had poor prognosis. Median follow-up was 6.03 months, the objective response rate (ORR) was 66.7% (95% confidence interval 39.9e93.3%), the median PFS was 7.9 months (2.0e20.1 months) and median survival was 11.2 months (2.2e20.1 months).
The ORR for patients with liver metastases was 83.3% (5/6). Five patients had platinum reintroduction in second line before ICIs; four of them had PD but responded to platinum administered after ICIs. Four patients in progression after ICIs received non-platinum chemotherapy; all had PD and were responders to subsequent platinum. Five subjects received the same platinum molecule at first line and at rechallenge (three received cisplatin and two carboplatin), four of them were in partial response and one in stable disease. Five patients received cisplatin at first line and carboplatin at rechallenge and two received carboplatin followed by cisplatin. In our cohort, three of the five patients responding to ICIs were responders to platinum rechallenge, and five of the seven patients who were non-responders to ICIs became responders at platinum-based rechallenge.
Recent retrospective analyses in solid tumours suggest an unexpected response to chemotherapy when administered after ICIs. In urothelial carcinoma, data suggest that chemotherapy conserves its activity when administered after ICIs, without cross-resistance between chemotherapy and ICIs 3. In this retrospective study, the authors describe an interesting response rate to chemotherapy rechallenge after ICIs with 2/14 patients in partial response and 10/14 with stable disease. It is of note that patients were not specifically refractory to platinum-based chemotherapy at first line, and patients received platinum or noneplatinum-based chemotherapy after ICIs. Patients with mUC eligible to receive chemotherapy at third line are rare, particularly those eligible for platinum-based regimens. Patients have poor performance status, and most of the patients die already after second-line treatment. Furthermore, oncologists often are reluctant to reintroduce chemotherapy regimens to patients who have been progressing less than 1 year after administration.
Based on our findings, it seems that ICIs could restore platinum sensitivity in mUC being refractory to platinum-based chemotherapy at first line. Data in lung cancer suggest that in selected patients, the sequence of immunotherapy followed by chemotherapy may be better than chemotherapy followed by immunotherapy in PFS 4. Our data suggest a similar observation.
The biological mechanisms involved in platinum resensitisation are still unclear. There are arguments in favour of the sequential association of ICIs and chemotherapy. The DNA damage induced by platinum based agent leads to an interferon-g production and results in an immune reactivation after ICI cessation, explaining the observed response 5. Our hypothesis is that an ICI induced modification in the tumour homoeostasis, resulting in a response improvement to platinum-based rechallenge chemotherapy. Recent data suggest the existence of a reciprocal regulation between immunity and angiogenesis 6. CD4þ T-cell activation induced by ICIs increases vessel normalisation with a positive feedback loop between Th1 enhancement and vessel normalisation. These vessel architecture modifications could enhance the platinum diffusion in the tumour, increasing the intratumoural platinum concentration and explain the observed improvement of the response.
Our study shows a promising response rate to platinum rechallenge after ICIs for platinum refractory mUC, and it could suggest that ICIs can resensitise mUC to platinum chemotherapy. The results of this small retrospective cohort need to be validated prospectively and possibly in other tumour types. The biological mechanisms involved in platinum resensitisation remain unclear and need to be investigated.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Authors: Gwenaelle Gravis1,2, Emilien Billon2, Capucine Baldini3, Christophe Massard3, Werner Hilgers4, Remy Delva5, Jochen Walz6, Geraldine Pignot6, Stanislas Rybikowski6, Slimane Dermeche2, Jeanne Thomassin7, Serge Brunelle8, Pernelle Lavaud9, Yohann Loriot9 On behalf of the French Genito-Urinary Tumor Group (GETUG)
Author Affiliation:
1. From the Centre de Recherche en Cance´rologie de Marseille, INSERM UMR1068, CNRS UMR7258, Institut PaoliCalmettes, Aix Marseille Universite´, Marseille, France
2. Medical Oncology, Institut Paoli-Calmettes, Marseille, France
3. Drug Development Department (DITEP), Gustave Roussy, University of Paris Sud, Villejuif, France
4. Medical Oncology, Institut Sainte Catherine, Avignon, France
5. Medical Oncology, Centre Paul Papin, Angers, France
6. Surgical Urology, Institut Paoli-Calmettes, Marseille, France
7. Biopathology Department Institut Paoli-Calmettes, Marseille, France
8. Radiology Department Institut Paoli-Calmettes, Marseille, France
9. De´partement de Me´decine Oncologique, Gustave Roussy, University of Paris Sud, Villejuif, France Received 15 May 2018; received in revised form 5 September 2018; accepted 6 September 2018 Available online ---
Gravis G, et al., Unexpected response to cisplatin rechallenge after immune checkpoint inhibitors in patients with metastatic urothelial carcinoma refractory to platinum regimen, European Journal of Cancer (2018), https://doi.org/10.1016/ j.ejca.2018.09.002
References
1. von der Maase H, Sengelov L, Roberts JT, Ricci S, Dogliotti L, Oliver T, et al. Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Urol 2006;175:482. https://doi.org/10.1016/S0022- 5347(05)00370-8.
2. Bellmunt J, de Wit R, Vaughn DJ, Fradet Y, Lee J-L, Fong L, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med 2017;376:1015e26. https: //doi.org/10.1056/NEJMoa1613683.
3. Szabados B, van Dijk N, Tang YZ, van der Heijden MS, Wimalasingham A, de Liano AG, et al. Response rate to chemotherapy after immune checkpoint inhibition in metastatic urothelial cancer. Eur Urol 2017. https://doi.org/10.1016/j.eururo.2017.08.022.
4. Park SE, Lee SH, Ahn JS, Ahn M-J, Park K, Sun J-M. Increased response rates to salvage chemotherapy administered after PD-1/PD-L1 inhibitors in patients with non-small cell lung cancer. J Thorac Oncol 2018;13:106e11. https: //doi.org/10.1016/j.jtho.2017.10.011.
5. Brzostek-Racine S, Gordon C, Van Scoy S, Reich NC. The DNA damage response induces IFN. J Immunol 2011;187:5336e45.
6.Mortara L, Benest AV, Bates DO, Noonan DM. Can the codependence of the immune system and angiogenesis facilitate pharmacological targeting of tumours? Curr Opin Pharmacol 2017; 35:66e74.
Read an Expert Commentary by Bishoy Faltas, MD

