Physicians are often hesitant to replete vitamin D levels in this population for fear of driving new stone formation secondary to increased intestinal absorption of calcium from vitamin D supplementation and the potential for subsequent hypercalciuria .
While data remains sparse to draw definitive conclusions, a few studies have examined the relationship between various vitamin D repletion protocols and urinary calcium excretion and found no convincing direct association.8-10 These studies, however, analyzed a variety of repletion doses and durations in varying patient populations. For these reasons, we sought to directly compare two of the most common repletion protocols of vitamin D supplementation in known calcium kidney stone formers and assess each protocol’s effect on urinary calcium excretion as well as other lithogenic risk factors such as calcium salt supersaturations.
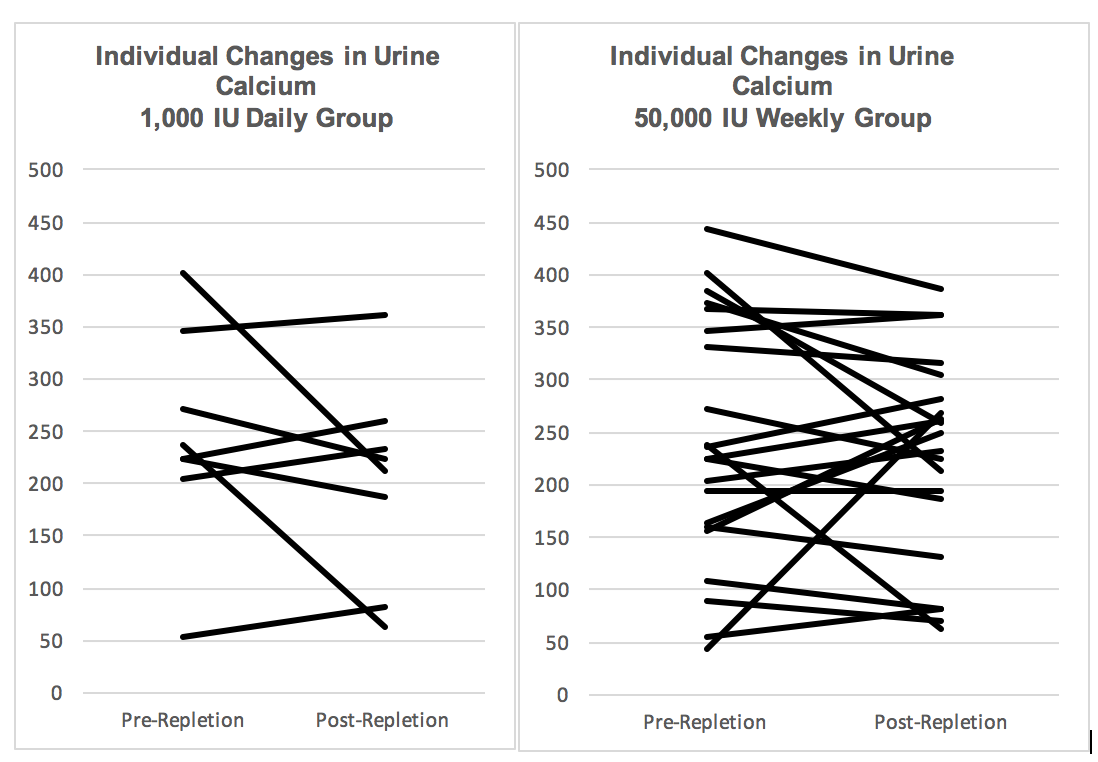
Upon diagnosis of vitamin D deficiency in the urology clinic, kidney stone patients were enrolled in the study and randomized to receive a 6-week course of vitamin D3 oral supplementation with either 1,000 IU daily or 50,000 IU weekly. Patients completed a 24-hour urine collection before and immediately after treatment to analyze changes in urinary excretion of calcium and other 24 hour urine parameters. Twenty-one patients completed the study, 8 in the low dose daily group and 13 in the high dose weekly group. Both groups showed improvement in serum vitamin D levels after repletion, from a median of 19.5 to 27.5ng/ml in the 1,000 IU daily group and 17 to 37ng/ml in the 50,000 IU weekly group, however, only in the latter group was this change statistically significant (p<0.01) and resulted in a median post-repletion level above the threshold for deficiency (>30ng/ml). There was no significant change in urine calcium excretion in either group after repletion; from 231 to 219mg in the 1,000 IU group with a median change of -11mg (p=0.26) and from 195 to 263mg in the 50,000 IU group with a median change of -16mg (p=0.75). The change in urine calcium for individual patients, separated by group, is depicted in the figure below to illustrate the variation among the patients within each group. There was also no significant change in the supersaturation of calcium salts in each group, with median delta of supersaturation of calcium oxalate and calcium phosphate of -0.8 and -0.5 respectively in the 1,000 IU daily group and 0.7 and 0.4 in the 50,000 IU weekly group.
In a sub-analysis, we evaluated whether vitamin D supplementation has a more pronounced impact on patients who were already hypercalciuric (urinary calcium excretion of >200mg in females and >250mg in males) on pre-treatment 24 hour urine collection and found no significant risk of worsening hypercalciuria. In the low dose daily group, 5 patients had a median change of -47.8mg after treatment and 5 patients in the high dose weekly group had a median change of -56.6mg providing some evidence for the safety of vitamin D repletion in patients who have known hypercalciuria.
In conclusion, repletion with 50,000 IU weekly of vitamin D for 6 weeks provides a more robust increase in serum vitamin D levels than 1,000 IU daily, without significantly increasing urinary calcium excretion or calcium salt supersaturation in known calcium stone formers. Additionally, neither group was associated with increasing any lithogenic risk factors among kidney stone formers. We hope this analysis provides the basis for vitamin D repletion in this population and encourages further study, as larger sample sizes are needed.
Written by: Michelle J Semins and Matthew C Ferroni
References:
1. Forrest KY, Stuhldreher WL. Prevalence and correlates of vitamin D deficiency in US adults. Nutr Res. 2011 Jan;31(1):48-54.
2. Munger KL, Levin LI, Hollis BW, et al. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA. 2006 Dec 20;296(23):2832-8.
3. Hyppönen E, Läärä E, Reunanen A, et al. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. See comment in PubMed Commons below Lancet. 2001 Nov 3;358(9292):1500-3.
4. Garland CF, Gorham ED, Mohr SB,et al. Vitamin D and prevention of breast cancer: pooled analysis. J Steroid Biochem Mol Biol. 2007 Mar;103(3-5):708-11.
5. Garland CF, Gorham ED, Mohr SB, et al. Vitamin D for cancer prevention: global perspective. Ann Epidemiol. 2009 Jul;19(7):468-83.
6. Elkoushy MA, Sabbagh R, Unikowsky B, et al. Prevalence and metabolic abnormalities of vitamin D-inadequate patients presenting with urolithiasis to a tertiary stone clinic. Urology. 2012 Apr;79(4):781-5.
7. Ticinesi A, Nouvenne A, Ferraro PM, et al. Idiopathic calcium nephrolithiasis and hypovitaminosis D: a case-control study. Urology. 2016 Jan:87;40-5.
8. Leaf DE, Korets R, Taylor EN et al. Effect of vitamin D repletion on urinary calcium excretion among kidney stone formers. Clin J Am Soc Nephrol. 2012 May;7(5):829-34
9. Penniston KL, Jones AN, Nakada SY, et al. Vitamin D repletion does not alter urinary calcium excretion in healthy postmenopausal women. BJU Int. 2009 Nov;104(10):1512-6.
10. Ferraro PM, Taylor EN, Gambaro G. Vitamin D intake and the risk of incident kidney stones. J Urol. 2016 Aug 18. [Epub ahead of print].
Read the Abstract


