(UroToday.com) The Society of Urologic Oncology (SUO) 2021 annual meeting in Orlando, FL was host to an excellent overview by Dr. Maria Carlo, MD of the future of genomic evaluation to direct therapy in renal cell carcinoma (RCC). Dr. Carlo began her presentation by defining genomic biomarkers in cancer as: “A measurable DNA or RNA characteristic that is an indicator of normal biologic processes, pathogenic processes, or response to therapeutic or other interventions”. The role of targetable biomarkers has been best defined in the non-small cell lung cancer disease space with multiple actionable targets currently present. In bladder cancer, Erdafitinib is being used to selectively target FGFR3 or FGFR2 altered metastatic clones and poly (ADP-ribose) polymerase (PARP) inhibitors have become featured in the management of select prostate cancer patients with DNA damage repair gene alterations. There is a paucity of genomic biomarkers in RCC due to the presence of few actionable somatic mutations with very rare kinase fusions.
Currently, the International Metastatic RCC Database Consortium (IMDC) risk group stratification criteria (anemia, high platelets, high neutrophils, high calcium, low Karnofsky Performance Score, <1 year from diagnosis) is used to guide the treatment of metastatic RCC.
- Favorable risk (0 risk factors):
- Anti-VEGF + PD-1 inhibitor
- Cabozantinib + nivolumab
- Axitinib + pembrolizumab
- Lenvatinib + pembrolizumab
- Anti-VEGF + PD-1 inhibitor
- Intermediate/poor risk (1+ risk factors):
- Anti-VEGF + PD-1 inhibitor
- Cabozantinib + nivolumab
- Axitinib + pembrolizumab
- Lenvatinib + pembrolizumab
- CTLA-4 + PD-1 inhibitor
- Anti-VEGF + PD-1 inhibitor
How does one choose between a tyrosine kinase inhibitor (TKI) + immunotherapy (IO) combination versus IO + IO regimen? One such potential biomarker is Polybromo 1 (PBRM1). In an ad hoc analysis of CheckMate-025 (Nivolumab versus Everolimus in advanced RCC patients previously receiving anti-angiogenic agents), patients with PBRM1mutations had superior overall survival (HR 0.65, p=0.03). Further analysis from COMPARZ demonstrated similar results (HR 0.63, p=0.0019).1,2 It also appears that increased CD8+ T-cell infiltration is associated with superior response to PD-1 blockade.3
An ad hoc analysis of IMmotion 151 (atezolizumab/bevacizumab versus sunitinib) identified molecular subsets associated with differential clinical outcomes to angiogenesis blockade alone or combined with a checkpoint inhibitor. Combination therapy with atezolizumab/bevacizumab was noted to be most effective in patients with high T-effector and/or cell-cycle transcription with both treatment arms benefiting subsets with high angiogenesis.4
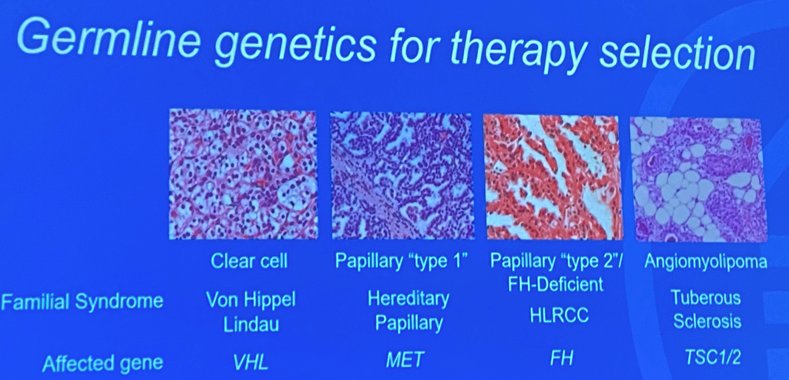
Dr. Carlo argued that we do currently have germline genetics for therapy selection:
- Clear cell (VHL)
- Papillary type 1 (MET)
- Papillary type 2 (FH)
- Angiomyolipoma (TSC1/2)
Food and Drug Association- (FDA) approved genomically targeted therapies are currently available:
- HIF-2 alpha inhibitors for VHL germline mutations
- mTOR inhibitors for TSC1 or TSC2 germline mutations
Furthermore, germline variants are frequent in advanced non-clear cell RCC:
- A series of 254 patients with advanced RCC found that 9% had FH germline mutations (diagnostic of Hereditary Leiomyomatosis and RCC [HLRCC])
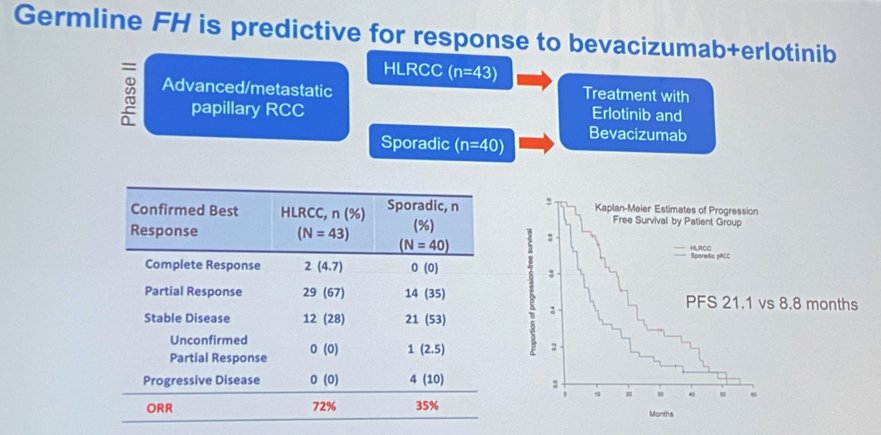
Approximately one in 1,00 individuals have a germline FH mutation diagnostic of HLRCC. To put things in perspective, Von Hippel Lindau (VHL) is present in only one in 36,000 individuals. A phase II trial of 83 patients with advanced/metasatatic papillary RCC treated with Erlotinib and Bevacizumab demonstrated superior progression-free survival (PFS) in patients with HLRCC compared to sporadic papillary RCC (PFS 21.1 versus 8.8 months, ASCO 2020).
A further analysis of 32 patients with metastatic FH-deficient RCC seen at MSKCC demonstrated that anti-VEGF therapies have efficacy in FH-RCC A phase II trial of cabozantinib and nivolumab presented at ASCO 2021 demonstrated that 5/6 (83.3%) of patients with FH-deficient RCC had an overall response rate.

Furthermore, MET activating mutations predict response to MET inhibitors such as Savolitinib in papillary RCC.
Currently, the National Comprehensive Cancer Network (NCCN) guidelines for hereditary RCC has the following therapy recommendations:
- HLRCC: Erlotinib + bevacizumab (useful in certain circumstances)
- TSC: Everolimus (useful in certain circumstances)
- VHL: Beltuzifan (preferred regimen) or pazopanib (useful in certain circumstances)
Dr. Carlo concluded her talk with the following summary points:
- Actionable genomic targets are (so far) very rare in RCC
- PRBM1 may be a prognostic and/or predictive biomarker
- Gene expression signatures are a promising biomarker to distinguish patients that benefit from immune therapy or anti-angiogenic therapy
- Tumor and germline sequencing should be considered for metastatic non-clear cell RCC, particularly papillary and unclassified
- Important for therapy selection, clinical trial eligibility, and cascade testing of family members
Presented by: Maria Isabel Carlo, MD, Assistant Attending Physician, Genitourinary Oncology Service, Clinical Genetics Service, Memorial Sloan Kettering Cancer Center, New York, NY
Written by: Rashid Sayyid, MD, MSc – Urology Chief Resident, Augusta University/Medical College of Georgia, @rksayyid on Twitter during the 2021 Society of Urologic Oncology (SUO) Winter Annual Meeting, Orlando, FL, Wed, Dec 1 – Fri, Dec 3, 2021.
References:
- Voss MH, Reising A, Cheng Y, et al. Genomically annotated risk model for advanced renal-cell carcinoma: a retrospective cohort study. Lancet Oncol. Dec 2018;19(12):1688-1698.
- Braun DA, Ishii Y, Walsh AM, et al. Clinical Validation of PBRM1 Alterations as a Marker of Immune Checkpoint Inhibitor Response in Renal Cell Carcinoma. JAMA Oncol. Sep 2019;5(11):1631-1633.
- Braun DA, Hou Y, Bakouny Z, et al. Interplay of somatic alterations and immune infiltration modulates response to PD-1 blockade in advanced clear cell renal cell carcinoma. Nat Med. 2020 Jun;26(6):909-918.
- Motzer RJ, Banchereau R, Hamidi H, et al. Molecular Subsets in Renal Cancer Determine Outcome to Checkpoint and Angiogenesis Blockade. Cancer Cell. 2020 Dec;38(6):803-817.

