He began with an overview of the principle of precision medicine, defined as the tailoring of medical treatment to the individual characteristics of each patient. This may be informed by a variety of data sources including genomics, transcriptomics, epigenomics, proteomics, metabolomics, radiographic data, and information from the electronic medical record.
In prostate cancer, markers may be utilized across the spectrum of disease from screening to advanced treatment decisions.
Dr. Cucchiara described the role of prostate-specific antigen (PSA)-based prostate cancer screening as controversial on the basis of discrepant results from the European Randomized Study of Screening for Prostate Cancer (ERSPC) and Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial. However, the use of genomic markers may allow for a more tailored approach. For example, he highlighted STHLM3, a prospective population-based diagnostic study of prostate cancer screening in men aged 50-69 years. This integrated model comprising PSA data, other biomarkers, and clinical data significantly outperformed PSA alone while saving many biopsies.
Further, previous data have demonstrated an association between BRCA2 mutations and the development of prostate cancer. The IMPACT study highlighted the value of PSA-based prostate cancer screening in this population. Thus, current guidelines now recommend earlier PSA screening for those with BRCA2 mutation.
Moving onto the question of prostate cancer diagnosis, Dr. Cucchiara highlighted the revolutionary role of magnetic resonance imaging (MRI) in recent years. However, the negative predictive value of MRI remains its greatest limitation. It is in this space that biomarkers may have particular utility. These include the novel ExoDx Prostate IntelliScore, a urine-based assay, SelectMDx, also a urine-based assay, and ConfirmMDx, a tissue-based assay. In spite of a high negative predictive value for each of these biomarkers, the 2020 European Association of Urology (EAU) guidelines deem the use of these biomarkers a weak recommendation on the basis of the available data.
Dr. Cucchiara then addressed the role of biomarkers in prostate cancer treatment. For context, he highlighted that there remains a large proportion of men who begin active surveillance who convert to active therapy. Thus, there is an unmet need for tests to risk-stratify patients for suitability for surveillance. He highlighted data on three commercially available markers (Oncotype DX® GPS, Prolaris®, and Decipher®) which may be used for this goal.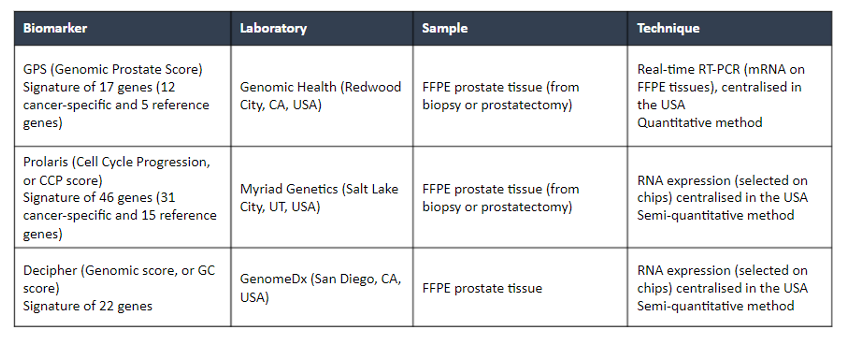
The use of these biomarkers, particularly Oncotype DX® GPS, has been shown to increase recommendations for active surveillance, as well as persistence on surveillance for those who initially opt for this approach. However, the use of biomarkers to enroll patients in active surveillance has not been prospectively validated.
Finally, in the context of adjuvant therapy, the ARTISTIC collaborative recently published data comparing adjuvant and early salvage radiotherapy following radical prostatectomy. In this pooled analysis, there was no difference in event-free survival between these approaches. In this context, it remains to be seen whether biomarkers can provide useful risk stratification in this setting. He highlighted data from Spratt et al. examining the ability of Decipher® assay results to predict metastasis following radical prostatectomy. However, there is a lack of prospective data to support this approach and thus it should not be routinely used.
In conclusion, he highlighted the importance of germline testing for patients with suspected germline mutations or familial syndromes as knowledge of BRCA status can influence screening and treatment decisions. Clinically available biomarkers may be considered in specific circumstances but, in Dr. Cucchiara’s perspective, should not be routinely recommended.
Presented by: Vito Cucchiara, MD, Department of Urology, Università Vita-Salute San Raffaele, Milan, Italy
Written by: Christopher J.D. Wallis, MD, PhD, Urologic Oncology Fellow, Vanderbilt University Medical Center, Nashville, Tennessee, Twitter: @WallisCJD at the 2020 Société Internationale d'Urologie Virtual Congress (#SIU2020), October 10th - October 11th, 2020

