(UroToday.com) The International Kidney Cancer Symposium 2021 annual hybrid meeting included a role of perioperative therapy in renal cell carcinoma (RCC) session and a presentation by Dr. Mohamad Allaf discussing surgical considerations with perioperative therapy. Dr. Allaf notes that TKIs have been used as monotherapy or in combination with immunotherapy in the neoadjuvant setting, adjuvant setting, or both. However, there is an element of overtreatment and toxicity associated with the treatment given that many patients are cured by surgery alone. As such, the role of perioperative TKIs in non-metastatic RCC is not well defined. Downstaging of tumor thrombus has been reported, which may shrink the tumor, but the level and approach are the same with the added toxicity of treatment. Additionally, TKIs have been used to facilitate resectability (may shrink some tumors +/- easier resection) and potentially enable nephron-sparing surgery (may shrink tumors, potentially enabling more partial nephrectomies). However, whether this therapy decreases recurrence or increases overall survival is unknown.
Several phase 2 trials have assessed the utility of neoadjuvant TKI therapy in patients with locally advanced RCC. Karam et al.1 assessed the utility of neoadjuvant axitinib for 12 weeks among 24 patients with locally advanced RCC, noting a 28% average reduction in tumor diameter, leading to 11 (46%) patients with partial responses, 13 (54%) with stable disease, and no patients that progressed. Notable complications included intraoperative assessment of the tumor being “stuck”, with grade 2 complications including chylous ascites, pulmonary embolism, and wound issues, as well as grade 3 complications including chylous ascites and bleeding. Rini and colleagues2 performed a phase 2 trial of pazopanib (8 weeks) in patients with localized RCC among 25 patients, noting that 92% of patients experienced a reduction in tumor volume. Additionally, six of 13 patients were able to undergo partial nephrectomy, with five patients having a urine leak and 8 patients having bleeding (7 requiring transfusion, and 1 requiring an embolization). Dr. Allaf notes that it is important to understand when to stop TKI therapy prior to surgery, as summarized in the following table:

With regards to perioperative systemic therapy, Dr. Allaf made the following observations:
- We have significant rationale to develop a novel perioperative systemic therapy
- There is a lack of widespread cure with the available agents in the metastatic setting
- There is no established perioperative therapy
- There is no benefit to adjuvant immunotherapy with interferon and IL-2 regimens based on data from >7 trials
- There is a controversial DFS benefit to 1 year of adjuvant VEGF blockade, with only the S-TRAC trial having positive results3
- The KEYNOTE-564 trial4 showed a DFS benefit for adjuvant pembrolizumab, however, we are still awaiting OS data
Dr. Allaf notes that there is a rationale for pre-nephrectomy anti-PD-1 priming, including (i) ongoing but unsuccessful anti-tumor T cell response in the primary tumor, tumor microenvironment, and draining lymph nodes, (ii) post-PD-1 blockade anti-tumor CD8 T cells may preferentially expand in these areas and then traffic to distant sites as memory cells where they can eradicate micrometastases, (iii) nephrectomy will remove the majority of these effector cells and cytokines potentially resulting in a less potent response, and (iv) there is a significant decrease in circulating PD-1+ cells after a nephrectomy. Mouse models in triple-negative breast cancer have shown an improved efficacy of neoadjuvant compared to adjuvant immunotherapy for eradication of metastatic disease, in addition to positive results for neoadjuvant immune checkpoint blockade in high-risk resectable melanoma and resectable lung cancer.
From a surgical perspective, there are several considerations:
- Will neoadjuvant therapy improve DFS and/or OS?
- How many will not make it to surgery?
- Do all patients need restaging and how do you assess these patients?
- Is surgery more complicated in these patients?
- How do you assess pathologic response?
At Johns Hopkins, Dr. Allaf and his colleagues recently published results of their prospective, open-label, single-arm trial evaluating neoadjuvant nivolumab in patients with nonmetastatic high-risk RCC.5 The trial schema is as follows:

The primary endpoint for this trial was the safety and tolerability of neoadjuvant nivolumab in non-metastatic RCC, with delay to surgery and surgical complications also recorded. Among 17 patients accrued to this trial, the median age was 65 years of age (IQR 62-69), 29.6% were female, and the most common clinical T-stage was cT3a (41.2%). All patients were cN0 and the median tumor diameter was 7.9 cm (IQR 6.7-8.5 cm). All patients underwent pre-enrollment biopsy safely with no complications, and all patients completed three doses of nivolumab with no delays to surgery. The median operative time was 120 minutes (IQR 100-147), estimated blood loss was 100 cc (IQR 50-240 cc), lymphadenectomy (performed at the surgeon’s discretion) was performed in 35% of patients, and median length of stay was 2 days (IQR 1-3). There were no grade 4-5 adverse events, and there were three grade 3 adverse events (kidney infection, lymphopenia), with the most common overall adverse events being fatigue (n=7), rash (n=5), pruritis (n=5), and hyperthyroidism (n=4); no patients received steroid treatment. Of note, one patient opted for continued off-label adjuvant nivolumab x 6 doses post-operatively and developed autoimmune diabetes and bullous pemphigous, requiring steroids >200 days post-trial. With regards to surgical complications, there were no wound complications, grade 1 complications included AKI (n = 1) and dyspnea (n = 1), grade 2 complications included pulmonary embolism/DVT (n = 1), UTI (n = 3), and urinary retention (n = 1); there were no Clavien 3 complications. Subjectively, there were no changes in tissue planes, and one patient underwent conversion to open surgery unrelated to tissue planes.
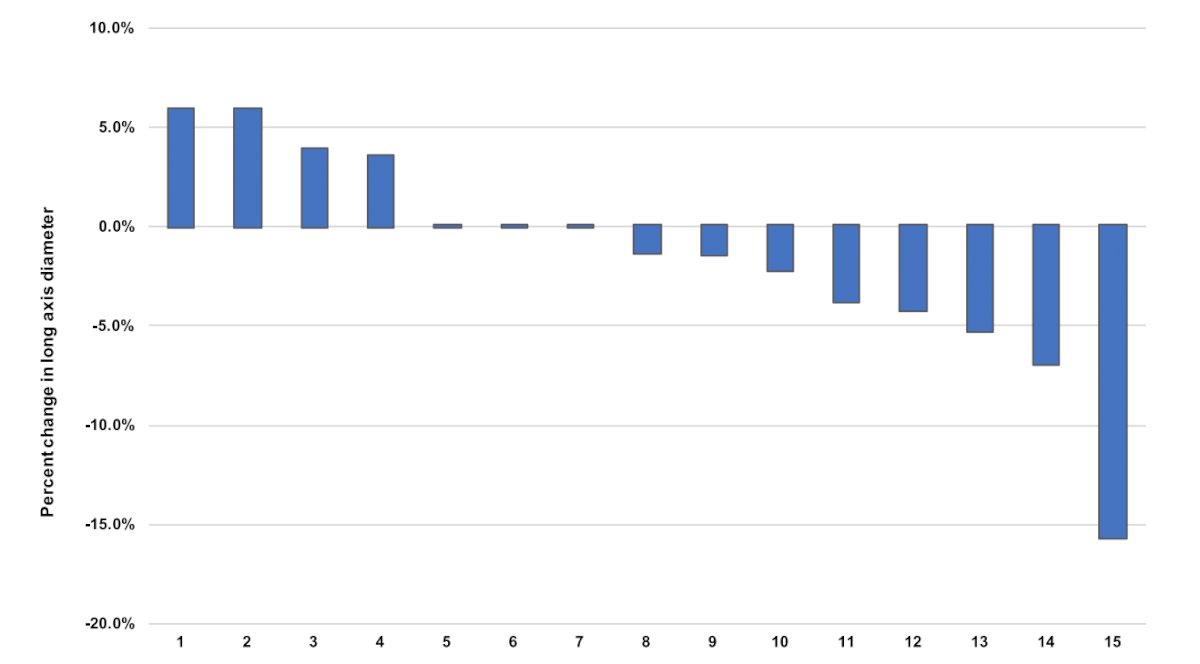
As follows is the percentage change in long axis tumor diameter after treatment with neoadjuvant nivolumab:
The following figure highlights features of immune-related pathologic response seen in the nephrectomy specimen from a responder to neoadjuvant nivolumab:
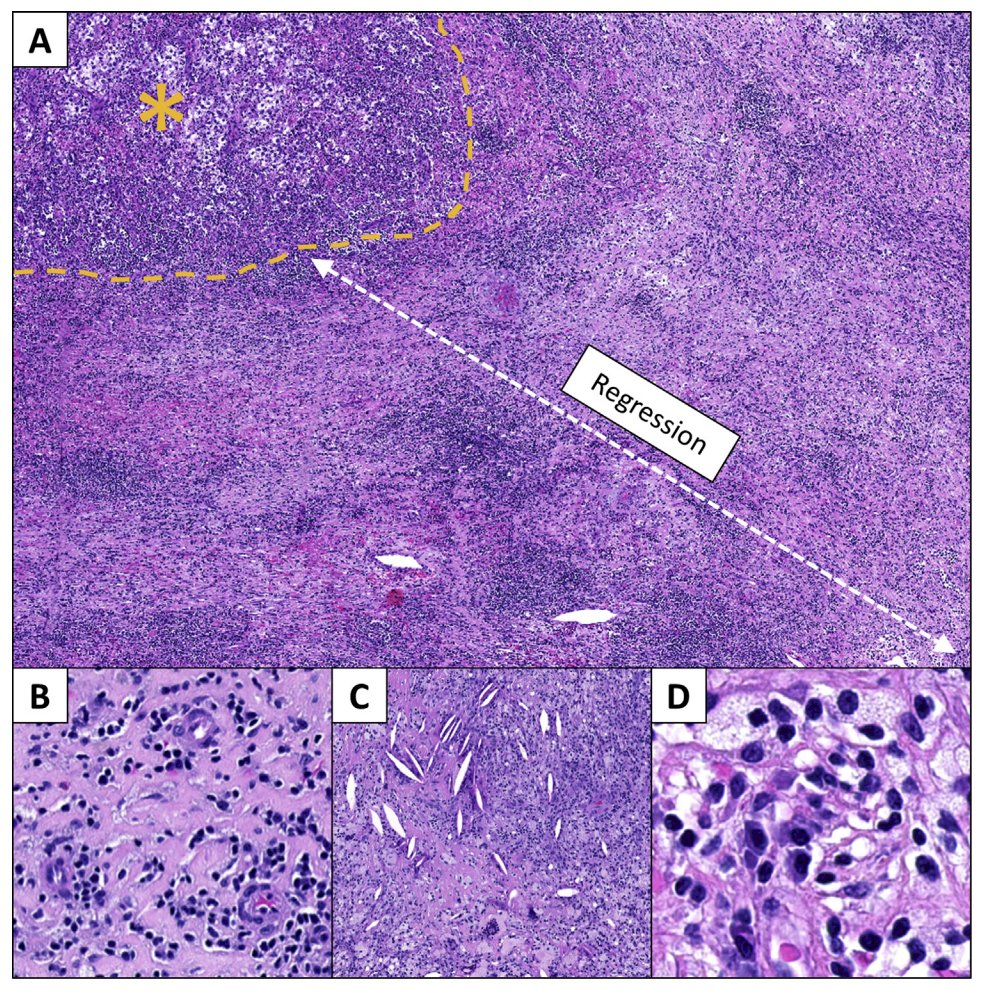
In panel A above, the immune-related pathologic response is characterized by a regression bed where the tumor used to be, with the remaining residual viable tumor marked by the yellow-asterisk.
Dr. Allaf then provided the following take-home messages from his presentation of surgical considerations with perioperative therapy:
- How many receiving neoadjuvant therapy will not make it to surgery? 0-10% of patients in existing reports (similar to chemotherapy), noting that it is very rare to delay surgery
- With regards to restaging and reassessment, all patients are restaged prior to surgery
- Surgical difficulty and postoperative complications are difficult to discern but were not a problem in the Johns Hopkins clinical trial
- How do we assess pathologic response? Immune activation, cell death, tissue repair, and regression bed can be seen in all tumor types, with pan-tumor pathologic scoring of response to PD-(L)1 blockade
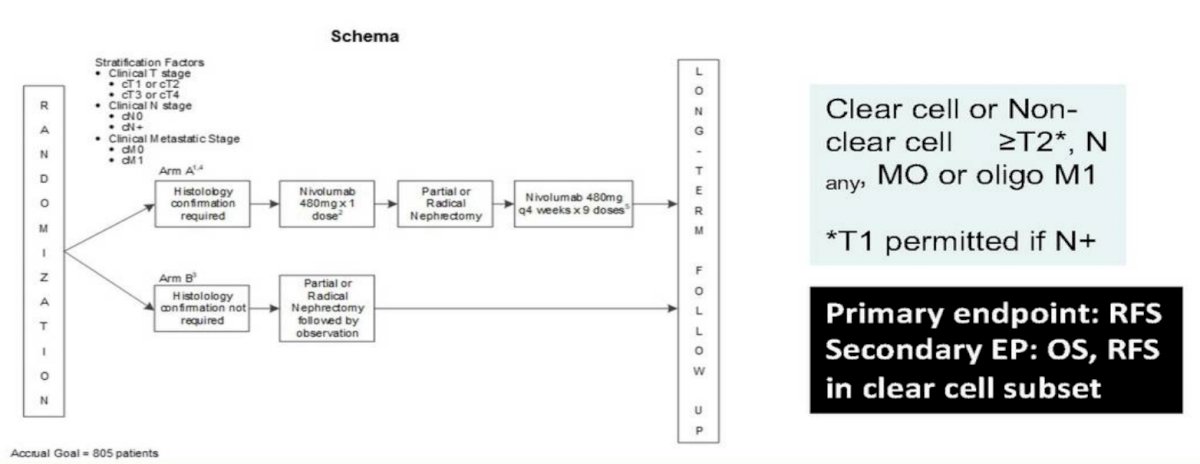
Dr. Allaf also noted that the EA8143 PROSPER RCC trial randomizing clear cell or non-clear cell RCC patients (>=T2, Nany, M0 or oligo M1 disease) to Arm A (neoadjuvant nivolumab x 1 dose followed by surgical resection followed by nivolumab x 9 doses) versus Arm B (surgical resection) has completed accrual. The trial schema for this trial is as follows:
Presented by: Mohamad Allaf, Professor of Urology and Oncology, Johns Hopkins Medicine, Baltimore, MD
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the International Kidney Cancer Symposium (IKCS) 2021 Annual Congress, November 5 and 6, 2021.
References:
- Karam JA, Devine CE, Urbauer DL, et al. Phase 2 trial of neoadjuvant axitinib in patients with locally advanced nonmetastatic clear cell renal cell carcinoma. Eur Urol. 2014;66:874-880.
- Rini BR, Plimack ER, Takagi T, et al. A phase II study of pazopanib in patients with localized renal cell carcinoma to optimize preservation of renal parenchyma. J Urol. 2015;194:297-303.
- Ravaud A, Motzer RJ, Pandha HS, et al. Adjuvant Sunitinib in High-Risk Renal-Cell Carcinoma after Nephrectomy. N Engl J Med 2016;375(23):2246-2254.
- Choueiri TK, Tomczak P, Park SH, et al. Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma. N Engl J Med. 2021 Aug 19;385(8):683-694.
- Gorin MA, Patel HD, Rowe SP, et al. Neoadjuvant nivolumab in patients with high-risk nonmetastatic renal cell carcinoma. Eur Urol Oncol. 2021 May [Epub ahead of print].

