In this study, presented by the lead author Dr. Duran, the authors evaluated the efficacy in the IMvigor210 cohort one patients by PD-L1 status over time. The authors evaluated the objective response rate, and overall survival in patients with the intention to treat analysis and in subgroups based on PD-L1 status across 4 data cuts, as shown in figure 1.
Figure 1 – Timing of evaluated analyses:
The objective and ongoing responses over time were presented (Figure 2) in all patients and stratified by the PD-L1 expression, where IC0/1 - <5% PD-L1 expression, and IC2/3 - >=5% PD-L1 expression. These results demonstrated a stable overall response rate among all patients in the intention to treat analysis of approximately 23-24%. However, in patients with PD-L1,>=5% the objective response rate was considerably higher than in patients with PD-L1<5%.

Figure 2 – Objective and ongoing responses over time:
Figure 3 demonstrates the complete response rate over time, again showing that in patients with PD-L1>=5% the results are considerably better.
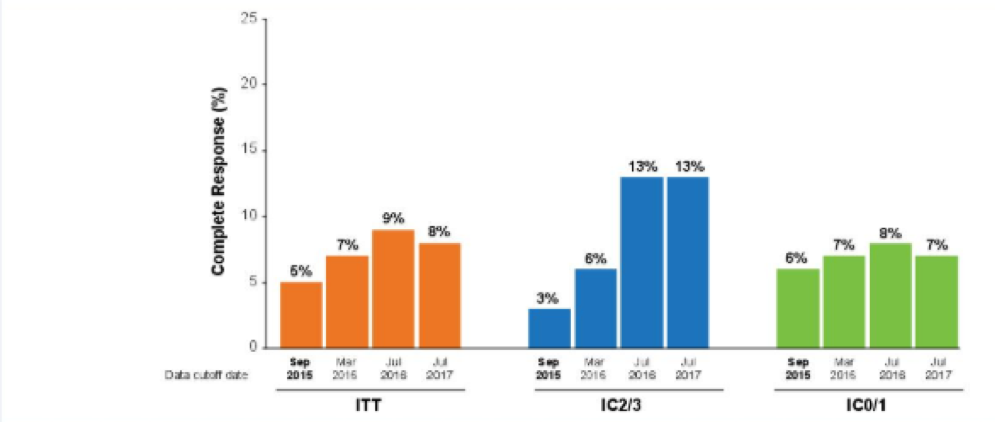
Figure 3- Complete response rates over time:
Many responders experienced long-term responses. Responses lasting more than four months, continue to last until the most recent analysis. Lastly, the authors analyzed the rate of survival over time. At the 2017 cutoff, the two-year overall survival was 41% in the intention to treat analysis patients, 39% in the PD-L1>=5%, and 42% in the PD-L1<5%, as shown in Figure 4.

Figure 4- Overall survival over time:
The authors concluded that evolution of overall response, complete response rates, and overall survival was witnessed with additional follow-up of up to 29 months in the IMvigor210 trial. Late conversions to complete response were observed, particularly in the PD-L1>=5% group of patients. Interestingly, the responses appeared to be durable irrespective of the PD-L1 status, and continued improvement in overall survival was demonstrated since the primary analysis was performed. Lastly, ongoing randomized studies (such as the IMvigor130) are needed to definitively evaluate the long-term benefit of 1st line Atezolizumab with or without chemotherapy in metastatic urothelial carcinoma patients.
Presented by: Durán Ignacio, MD, Spain
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan at the Global Conference on Bladder Cancer 2018 - September 20-21, 2018 Madrid, Spain

