Methotrexate + vinblastine + doxorubicin + cisplatin (MVAC) protocol for the treatment of advanced urothelial carcinoma was first introduced in the 1980s. Later the treatment was changed to gemcitabine and cisplatin (GC), after a trial by Von der Masse in the year 2000 demonstrated a similar survival advantage as MVAC, but a better safety profile.1
Later, the concept of increasing the dose density was introduced. The initial results of comparing dose-dense (DD)-MVAC with granulocyte-colony stimulating factor (GCSF) compared to standard MVAC showed that DD was not superior in the median overall survival (p=0.122), despite obtaining a higher objective response rate (62% vs. 50%) and longer progression-free survival (9.1 vs. 8.2 months).2 DD also conferred less neutropenia and febrile neutropenia episodes than standard MVAC.
As time progressed, researchers attempted to see whether the addition of other drugs can improve outcomes. GCSF addition was shown to ameliorate the toxicity significantly. Docetaxel demonstrated lower efficacy compared to MVAC. The triplet of Paclitaxel, Cisplatin, and Gemcitabine provided a higher response rate (55.5%) when compared to GC (43.6%), but overall survival was not different (15.8 vs. 12.7 months, p=0.75).
Dr. Duran moved on to discuss the population of patients that are unfit for 1st line cisplatin-based chemotherapy. Approximately 30-50% of patients will not be able to receive cisplatin chemotherapy due to different reasons. Therefore, other drug combinations were tried in these patients. This included gemcitabine and carboplatin, and the combination of vinflunine and gemcitabine, but these did not demonstrate any survival advantage worth pursuing.
Currently, GC is the first line therapy recommended for cisplatin eligible patients, followed by DD-MVAC. In Cisplatin ineligible patients, gemcitabine and carboplatin are recommended or Atezolizumab or Pembrolizumab (for patients whose tumors express high PD-L1 levels, or those not eligible for any platinum-containing chemotherapy despite their PD-L1 expression).
More recently, there have been some studies challenging the role of chemotherapy as the first line of therapy in patients by comparing its performance to the use of immune checkpoint inhibitors, or some combination of them. Pembrolizumab alone as 1st line therapy in patients with advanced bladder cancer, who have never received or were not eligible for cisplatin-based chemotherapy, demonstrated to have an overall response rate of 28.9%, with a complete response rate of 8.1% and a 20.8% partial response rate.3 The median duration of response was not reached, and the median overall survival was 11.5 months, with a 12 months overall survival rate of 47.5%. In a similar study population, Atezolizumab monotherapy was examined as well, demonstrating an overall response rate of 24% and complete response rate of 8%, with the median overall survival of 15.9 months, and a median durable response rate not yet reached.4
There are currently several randomized trials comparing various immune checkpoint inhibitors in the first line setting in metastatic patients who did not receive cisplatin chemotherapy or were not eligible for it, as detailed in Table 1.
Table 1 – Ongoing randomized immune checkpoint inhibitor trials assessing their role in the setting of first-line therapy for advanced bladder cancer:

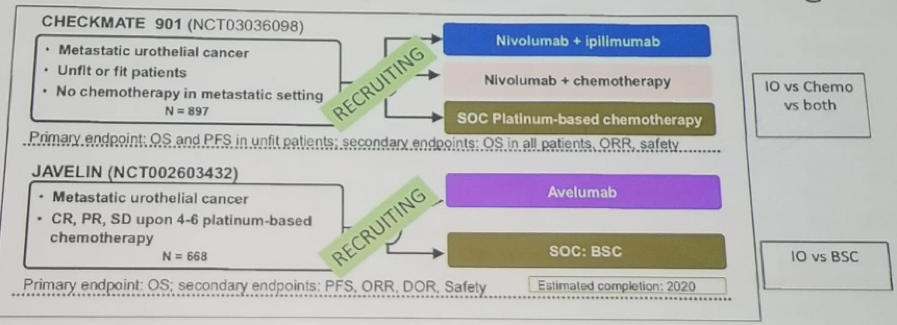
It is important to note that earlier this year, the FDA issued a warning coming from the data monitoring committees of the KEYNOTE-361 and IMvigor 130 trials, described above in table 1, assessing the role of Pembrolizumab and Atezolizumab, respectively as monotherapy. This warning stated that both drugs demonstrated decreased survival in patients with low expression of PD-L1, leading to the assumption that a high level of PD-L1 expression could be a requisite for benefiting from immune checkpoint inhibitors as monotherapy.
Summarizing the topic of 1st line treatment, Dr. Duran state that chemotherapy remains the preferred option for systemic treatment in 1st line metastatic urothelial carcinoma patients who are cisplatin eligible. Immunotherapy should be currently restricted only to patients who are not eligible for cisplatin-containing chemotherapy, and whose tumors express PD-L1. The ongoing randomized trials will hopefully shed some more light on this issue soon.
The next topic briefly discussed by Dr. Duran was the setting of second-line therapy. There are several studies assessing this setting. However, most are undersized single-agent studies with scarce success rates. The overall response rate ranges between 0-28%, with median overall survival of 7-8 months. The few randomized studies demonstrated and overall survival 0f 6.9 months for the Vinflunine medication, but overall survival was not shown to be better than standard treatment in the intention to treat analysis. There might be some hope with the use of docetaxel and anti VEGF2, but more data is required. Currently, 2nd line immune checkpoint inhibitors have superseded and replaced chemotherapy in the second line setting as the preferred approach. However, we still lack the predictive biomarkers to help us select the patients most likely to benefit from this treatment of immune checkpoint inhibitors. Lastly, chemotherapy continues to have a role in the treatment after immune checkpoint inhibitors (both in 2nd line and 3rd line), and there does not seem to be any cross-resistance.
Dr. Duran concluded his elegant talk with a nice algorithm demonstrating where chemotherapy should be considered, according to his opinion, in the setting of metastatic urothelial carcinoma patients (Figure 1).
Figure 1 – Dr. Duran’s opinion on where Chemotherapy should be considered in the setting of advanced urothelial carcinoma:

Presented By: Ignacio Duran, Madrid, Spain
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan at the Global Conference on Bladder Cancer 2018 - September 20-21, 2018 Madrid, Spain
References:
1. Von der Masse H et al. J Clin Oncol 2000
2. Sternberg CN et al. J Clin Oncol 2001
3. Vuky J et al. J Clin Oncol 2018
4. Balar AV et al. Lancet Oncol 2016
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan at the Global Conference on Bladder Cancer 2018 - September 20-21, 2018 Madrid, Spain
References:
1. Von der Masse H et al. J Clin Oncol 2000
2. Sternberg CN et al. J Clin Oncol 2001
3. Vuky J et al. J Clin Oncol 2018
4. Balar AV et al. Lancet Oncol 2016

