(UroToday.com) The EUO lecture at the 2021 European Association of Urology (EAU) Section of Oncological Urology (ESOU) virtual annual meeting was given by Dr. Thomas Powles, a medical oncologist from London discussing the systemic treatment of metastatic renal cell carcinoma. There have been exciting advances in the first-line treatment of metastatic renal cell carcinoma over the last 5-10 years, summarized by Dr. Powles in his EUO lecture.
First presented at ESMO in the fall of 2017 and subsequently published in 2018, CheckMate 214 demonstrated an overall survival benefit for first-line nivolumab plus ipilimumab vs sunitinib.1 This trial randomized 1,096 patients to the combination immunotherapy approach of nivolumab plus ipilimumab (550 patients) or sunitinib (546 patients). The majority of patients had intermediate or poor risk disease (n=847). Overall survival was significantly improved in the overall patient population; however, stratified analyses provide more nuanced results. Among the subgroup of patients with intermediate or poor-risk renal cell carcinoma (RCC), treatment with nivolumab plus ipilimumab resulted in significantly improved overall response rate, comparable progression-free survival, and significantly improved overall survival: minimum 17.5 month follow-up: 0.63, 99.8% CI 0.44-0.89; minimum 30 month follow-up: 0.66, 95% CI 0.54-0.80; minimum 42 month follow-up: 0.66, 95% CI 0.55-0.80. In contrast, among patients with favorable-risk disease, progression-free survival and overall response rate were higher among patients who received sunitinib.
Subsequently, Escudier and colleagues have assessed the efficacy of nivolumab and ipilimumab according to the number of IMDC risk factors.2 In keeping with the previously reported differences in the comparative benefit of nivolumab plus ipilimumab versus sunitinib on the basis of risk category (intermediate/poor vs favorable), the authors demonstrated stable objective response rate (ORR) across increasing numbers of IMDC risk factors (from zero to 6) for those who received nivolumab and ipilimumab, while the ORR in patients treated with sunitinib decreased with an increasing number of IMDC risk factors.
According to Dr. Powles, one of the highlights of the nivolumab plus ipilimumab combination therapy is the durable progression-free survival results in the intermediate/poor risk patients (35% at 42 months of follow-up):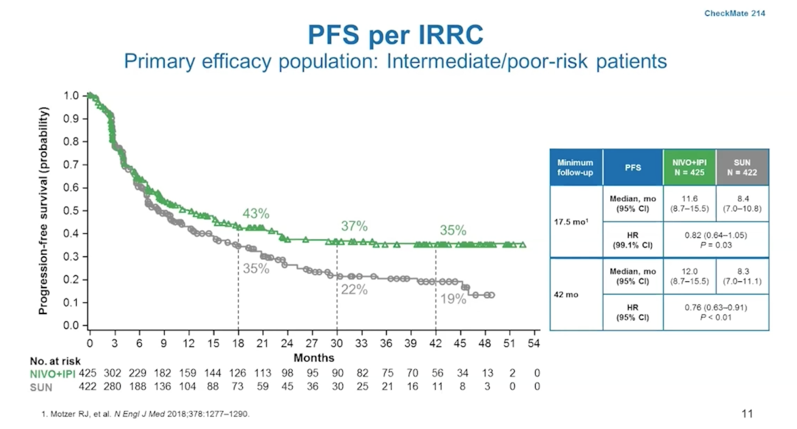
In KEYNOTE-426, 861 patients with metastatic clear cell RCC, predominately with intermediate or poor risk disease, who had not previously received systemic therapy were randomized to pembrolizumab + axitinib or sunitinib and followed for the co-primary endpoints of overall survival and progression free survival.3 While median survival was not reached, patients who received pembrolizumab + axitinib had improved overall survival (HR 0.53, 95% CI 0.38 to 0.74) and progression-free survival (HR 0.69, 95% CI 0.57 to 0.84), as well as overall response rate. These results were consistent across subgroups of demographic characteristics, IMDC risk categories, and PD-L1 expression level.
First presented at ESMO 2020, the CheckMate-9ER trial is a phase III RCT comparing nivolumab plus cabozantinib to sunitinib, in the first-line treatment of patients with advanced or metastatic renal cell carcinoma. Included patients had previously untreated, histologically confirmed advanced or metastatic renal cell carcinoma with a clear cell component and were randomized in a 1:1 fashion to receive nivolumab 240mg IV q2 weeks plus cabozantinib 40mg PO daily or sunitinib 50mg PO daily for 4 weeks in repeating 6 week cycles, with randomization was stratified by IMDC risk score, tumor PD-L1 expression, and region. The primary outcome was progression-free survival with overall survival, objective response rate, and toxicity comprising important secondary outcomes.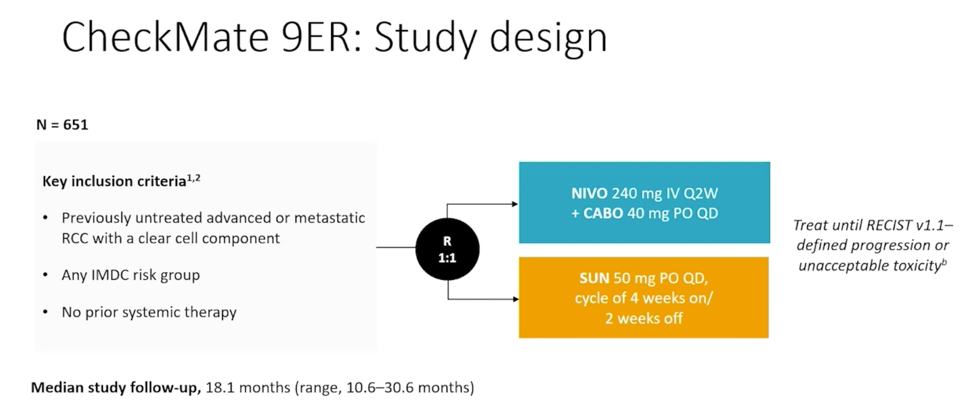
Among 651 randomized patients (22.6% favorable-risk disease, 57.6% intermediate-risk disease, and 19.7% poor-risk disease), 323 received nivolumab + cabozantinib and 328 received sunitinib. Over a median follow-up of 18 months, median progression-free survival was significantly longer among those randomized to nivolumab + cabozantinib (16.6 months) than those randomized to sunitinib (8.3 months), with a relative difference of 49% (HR 0.51, 95% CI 0.41 to 0.64) as was overall survival (medians not reached; HR 0.60, 98.89% CI 0.40 to 0.89):
Notably, these benefits were seen consistently across pre-specified subgroups defined according to IMDC risk categories and PD-L1 expression. Of particular interest to Dr. Powles is the vast improvement in health-related quality of life for patients receiving nivolumab plus cabozantinib compared to the quality of life deterioration among patients receiving sunitinib:
The JAVELIN Renal 101 randomized 886 patients to avelumab + axitinib or sunitinib.4 The majority of patients had IMDC intermediate or poor risk disease. In this analysis, the primary endpoints were progression-free survival and overall survival in patients with PD-L1 positive tumors, of which 560 of the 886 patients had PD-L1 positive tumors. Among the PD-L1 positive subgroup, progression-free survival (HR 0.61, 95% CI 0.47 to 0.79) was improved in patients receiving avelumab + axitinib compared to sunitinib while overall survival did not significantly differ (HR 0.82, 95% CI 0.53 to 1.28). In the overall study population, progression-free survival was similarly improved, as compared to the PD-L1 positive population (HR 0.69, 95% CI 0.56 to 0.84). Although the JAVELIN Renal 101 study did not have an OS benefit, the PFS gene signature has been of interest moving forward in this disease space:
Regarding the second line of therapy, Dr. Powles notes that it is essentially a dealer’s choice, however the goal is to give the patient a VEGF targeted therapy that they have not previously received.
Dr. Powles concluded with the following take-home messages from his presentation:
- Treatment choice exists and there are attractive points for all three regimens
- There are more similarities than differences between pembrolizumab plus axitinib and nivolumab plus cabozantinib
- The pendulum will likely swing toward VEGF TKI plus IO and away from IO plus IO
- Good clinical care is more important than treatment choice
Presented by: Thomas Powles, MBBS, MRCP, MD, Professor of Genitourinary Oncology, Lead for Solid Tumour Research at Barts Cancer Institute, Director of Barts Cancer Institute, London UK.
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md during the 18th Meeting of the EAU Section of Oncological Urology (ESOU21), January 29-31, 2021
References:
1. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carinoma. N Engl J Med2018;378(14):1277-1290.
2. Escudier B, Motzer RJ, Tannir NM, et al. Efficacy of Nivolumab plus Ipilimumab According to Number of IMDC Risk Factors in CheckMate 214. Eur Urol. 2020 Apr;77(4):449-453.
3. Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380(12):1116-1127.
4. Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380(12):1103-1115.
Related Content
Watch: ESMO 2020: CheckMate-9ER Study of Nivolumab Combined With Cabozantinib Vs. Sunitinib in Participants With Previously Untreated mRCC - Toni Choueiri

