(UroToday.com) Collecting duct renal cell carcinoma is a rare and aggressive form of kidney cancer that is often misdiagnosed, lacks standard treatment protocols, and is poorly characterized at the molecular level. There are at least two molecular subtypes that have recently been uncovered, based on RNA expression of cell signaling, metabolic and immune-related genes.1
In this presentation, Dr. Giuseppe Procopia presented results from a single-arm phase 2 trial exploring the utility of the multi-tyrosine kinase inhibitor cabozantinib for patients with previously untreated metastatic collecting duct carcinoma (mCDC). A total of 23 patients were enrolled on trial, with the primary endpoint of objective response rate.

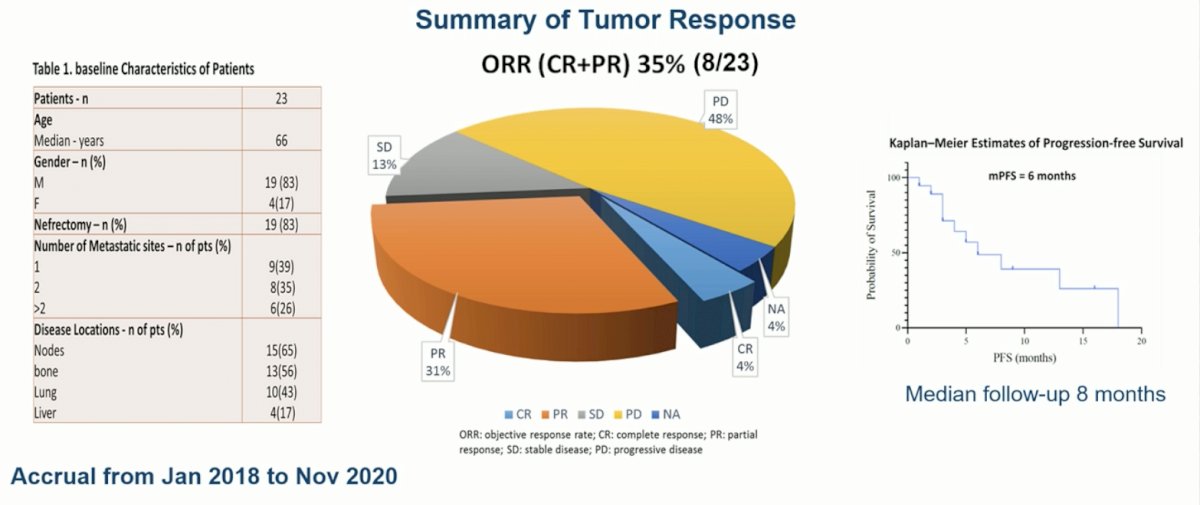
Of the 23 patients enrolled, the majority were male and had previously undergone nephrectomy. The objective response rate was 35%, with 8 patients achieving either a complete or partial response with cabozantinib treatment. This corresponded to a median progression-free survival of 6 months. Almost half of the patients had progressive disease despite treatment at the first disease evaluation timepoint.
No new safety signals were detected with cabozantinib treatment relative to prior studies. A majority of patients had grade 1-2 side effects such as fatigue, hypothyroidism, stomatitis, and anorexia. Four patients required dose reduction due to adverse events.
In an exploratory analysis, 21 out of 23 patients had successful tumor DNA sequencing performed. All tumors were microsatellite stable, and no association between tumor mutational burden and response to cabozantinib was observed. Mutations in FLNA and TGFBR2 were exclusively observed in responders (blue colored bar in graphic below).

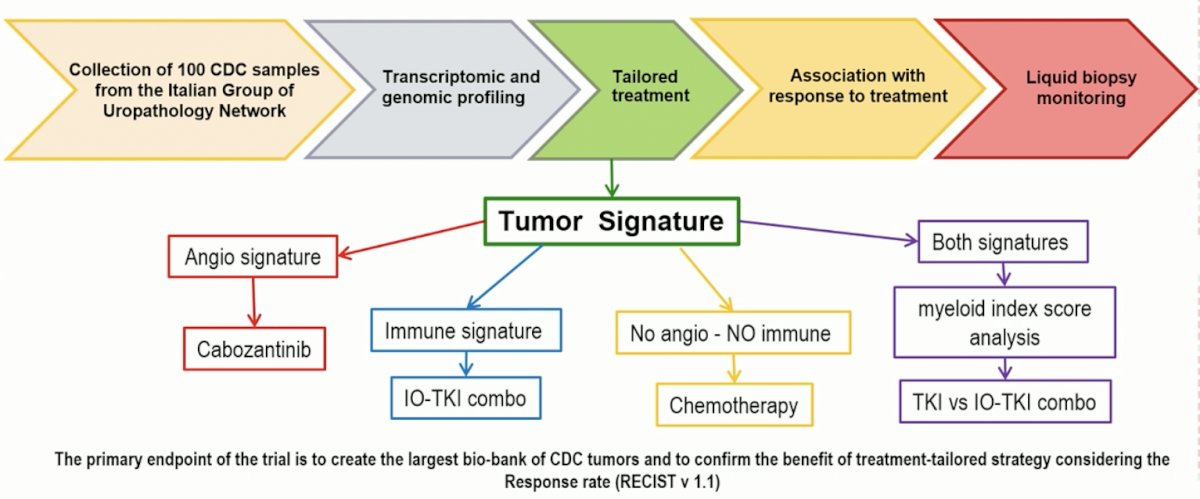
Dr. Procopio concluded that cabozantinib showed promising efficacy and reasonable tolerability in mCDC patients. More mature results with longer follow-up, stratified by molecular analysis of tumor DNA and analysis of peripheral blood mononuclear cells, will be presented in the future. He also mentioned the next planned study in mCDC, which is shown below.
Presented by: Giuseppe Procopio, Department of Medical Oncology, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, Italy
Written by: Alok Tewari, MD, PhD – Genitourinary Medical Oncologist, Instructor in Medicine, Dana-Farber Cancer Institute, Harvard Medical School, Twitter: @aloktewar during the 2021 European Society for Medical Oncology (ESMO) Annual Congress 2021, Thursday, Sep 16, 2021 – Tuesday, Sep 21, 2021.

