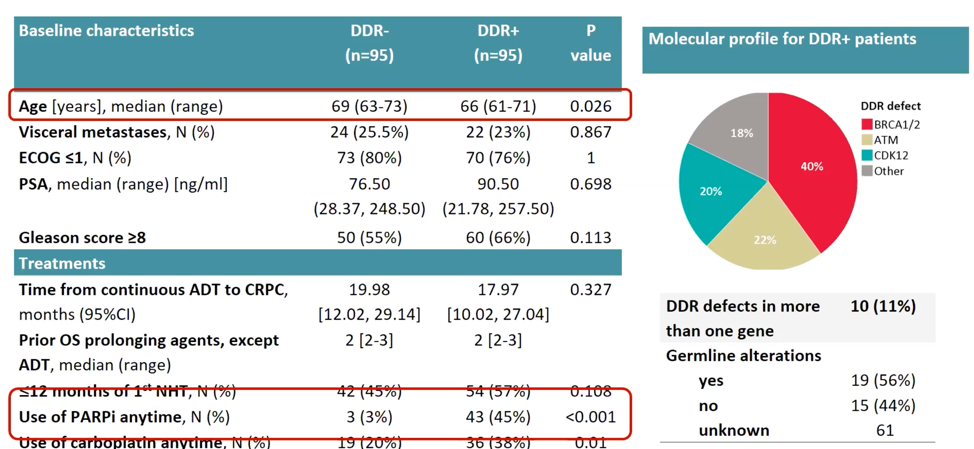
To address this question, the authors performed a multi-center retrospective study of mCRPC patients who received cabazitaxel and had molecular profiling performed that assayed at least BRCA1, BRCA2, CDK12, ATM, CHEK1, CHEK2, FANCA, FANCL, PALB2, RAD51, and BRIP1 genes for alterations. A total of 190 patients were identified, 95 of which had an alteration in a DDR gene (DDR+), and 95 of which did not (DDR-). The clinical characteristics and molecular profiles of the patients in the study are shown below. DDR+ patients were younger than DDR- patients by approximately 3 years, and DDR+ patients more commonly received PARP inhibitor or carboplatin therapy. The most common DDR alteration discovered was in either BRCA1 or BRCA2. A subset of these alterations was germline, and 19 of the 34 patients with available germline patients having a germline DDR alteration.
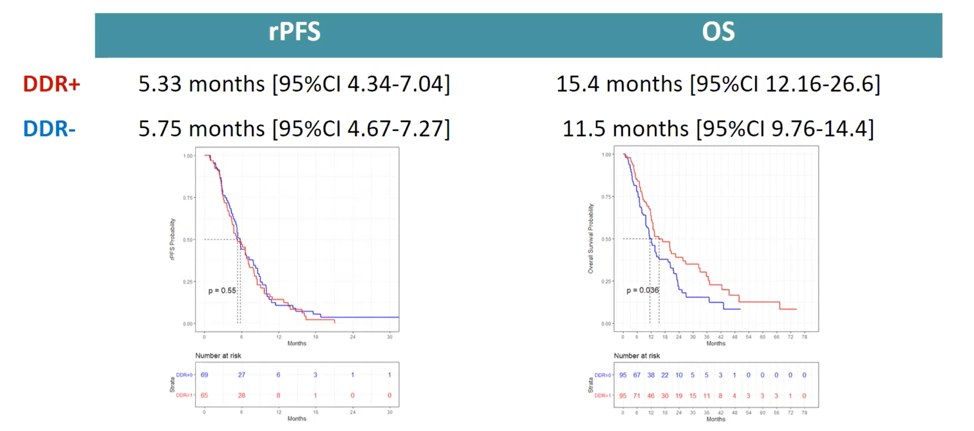
No difference in PSA decline was observed between DDR+ and DDR- patients treated with cabazitaxel. No difference in radiographic progression-free survival was observed between DDR+ and DDR- patients. However, DDR+ patients receiving cabazitaxel had a longer median overall survival (15.4 months) relative to DDR- patients (11.5 months), which could be explained by DDR+ patients deriving benefit from other therapies such as PARP inhibitors or carboplatin.
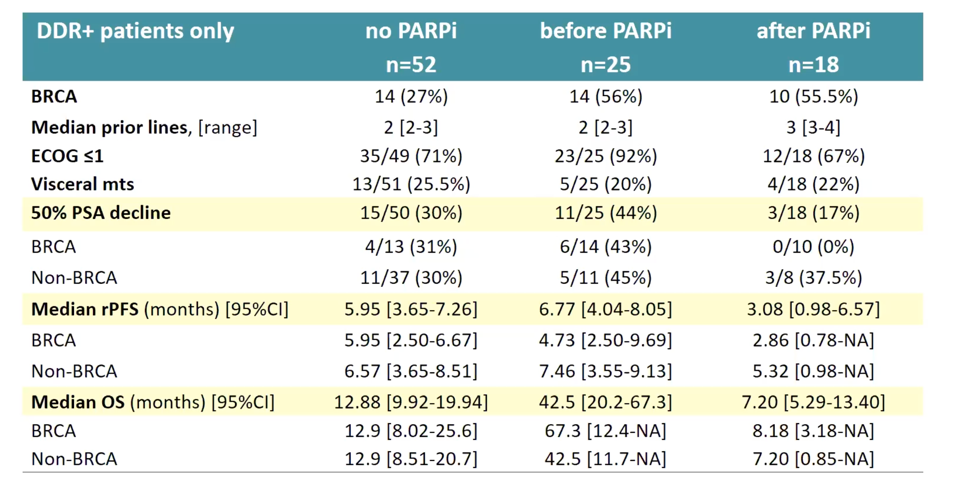
The authors divided the DDR+ patient population into those who received PARP inhibitor therapy or those that did not and examined the efficacy of cabazitaxel in those that did not receive PARPi versus those that received cabazitaxel before PARPi and those that received cabazitaxel after PARPi. Though limited in its generalizability due to small numbers, there is some suggestion that cabazitaxel may be less effective if given after PARPi. This finding will require further validation.
In summary, this study supports the efficacy of cabazitaxel regardless of DDR gene status but raises the question of the optimal timing of cabazitaxel administration relative to PARP inhibitor treatment for men eligible for both treatments.
Presented by: Mihaela Aldea, MD, PhD, Medical Oncologist at the Institut Gustave Roussy, Villejuif, France
Written by: Alok Tewari, MD, Ph.D., Medical Oncologist at the Dana-Farber Cancer Institute, at the 2020 European Society for Medical Oncology Virtual Congress (#ESMO20), September 19th-September 21st, 2020.
Related Content:

