
To generate biomarker data, baseline archival tissue was profiled using the Myriad myChoice homologous recombination deficiency test. This assay generates a genomic instability score (GIS) based on the detected presence of copy number alterations consistent with a deficiency in homologous recombination DNA repair. This test was considered positive (presence of GIS) if an aggregate score from the three inputs (large-scale transitions, telomeric allelic imbalance, and loss of heterozygosity) was 33 or higher. Circulating tumor DNA was also sequenced from baseline blood samples looking for alterations in 16 core DNA damage response genes (ATM, ATR, BARD1, BRCA2, BRIP1, CHEK1, CHEK2, FANCL, PALB2, PP2R2A, RAD51B, RAD51C, RAD51D, RAD54L). A sample was classified as DNA damage repair deficiency positive (DDR+) if an alteration was detected in these genes. The authors then looked for associations between GIS or DDR+ and clinical response to pamiparib and temozolomide.
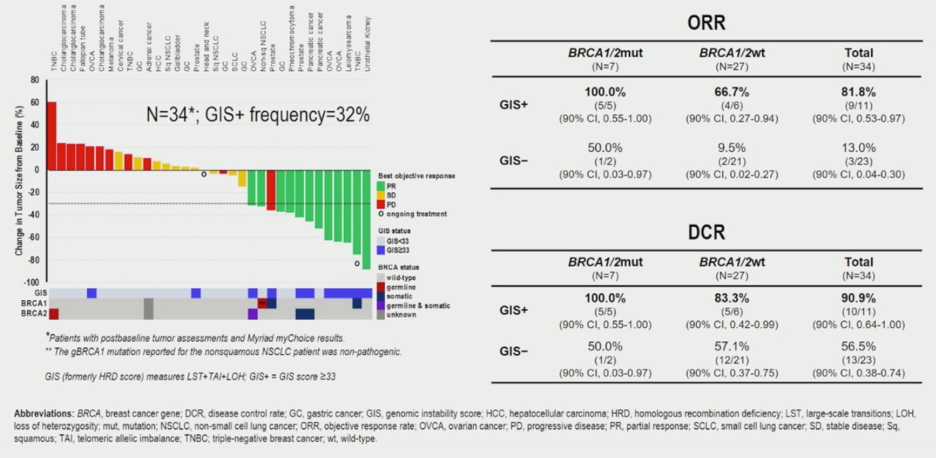
Of 34 samples analyzed for GIS, 32% of tumors were GIS positive (histologies shown above waterfall plot). GIS positivity was associated with higher overall response and disease control rate relative to GIS negative patients in both BRCA wildtype and BRCA mutant patients. DDR+ patients had a higher overall response and disease control relative to DDR- patients only if the DDR mutation was in BRCA1 or BRCA2.

Overall, these data suggest that GIS may be a robust biomarker across multiple tumor types for a response to PARP inhibitor therapy. Mutations in specific DDR genes beyond BRCA1/2 do not clearly associate with differential response to therapy. This trial is now opening a new cohort to explore the efficacy of combination ramiparib and temozolomide in GIS+ NSCLC, head and neck tumors, esophageal cancers and soft tissue sarcomas.
Presented by: Emiliano Calvo, MD, PhD, Medical Oncologist and Director of Clinical Research at the Centro Integral Oncologico Clara Campal, Hospital Universitario HM Sanchinarro
Written by: Alok Tewari, MD, PhD, Medical Oncologist at the Dana-Farber Cancer Institute, at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020

