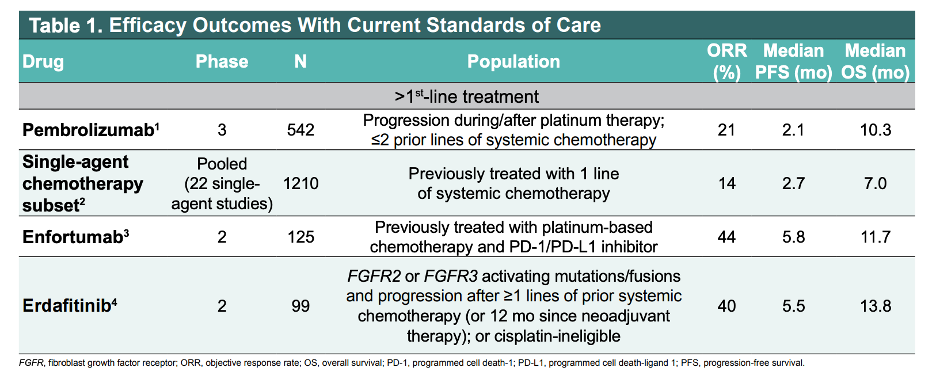
Antibody drug conjugates that are targeted to a highly expressed tumor protein and conjugated to a chemotherapy payload, such as enfortumab vedotin, are emerging as effective therapies for subsequent lines of treatment. The efficacy outcomes of the antibody drug conjugate enfortumab relative to other standards of care in the later lines of therapy are shown below.
Sacitizumuab govitecan (SG) is an antibody drug conjugate targeted to the surface protein Trop2 and is conjugated to the irinotecan metabolite SN38 as payload via a hydrolyzable linker that does not require antibody internalization in order to deliver the payload to tumor cells and the microenvironment. SG has demonstrated clinical activity in pre-treated patients with metastatic urothelial carcinoma (mUC). For example, patients within the mUC cohort of IMMU-132-01 trial had a 31% objective response rate with median duration of response of 12.9 months with SG monotherapy. Two other cohorts within the TROPHY-U-O1 clinical trial confirmed this activity. Interim data from cohort 1, consisting of 35 patients who had progressed on platinum and immune checkpoint blockade, demonstrated an objective response rate of 29%. Preliminary data from cohort 2, consisting of patients ineligible for cisplatin therapy, has shown an objective response rate of 28%. The major side effects of SG therapy have been neutropenia and diarrhea. Based on this data, SG has a fast-track designation for mUC therapy from the FDA.
In this poster, the authors present cohort 3 of the TROPHY-U-O1 open label phase 2 trial of SG. Cohort three will assess the response rate, safety, and other key secondary outcomes of SG in combination with pembrolizumab in up to 61 patients who have not received prior immunotherapy. The schema of the trial is shown below in the context of other TROPHY-U-O1 cohorts.

Patients with advanced urothelial carcinoma who either have disease recurrence less than 12 months after completing neoadjuvant or adjuvant platinum therapy or progressed after platinum therapy in the metastatic setting are eligible. They must have creatinine clearance greated than 30 mL/min, measurable disease, and good ECOG performance status. Prior irinotecan, prior immunotherapy, and active CNS metastases are key exclusion criteria. Enrollment for this study began in March 2020, with a planned 40 sites throughout the United States and France.
Presented by: Petros Grivas, MD, Associate Professor, Clinical Director of Genitourinary Cancers Program, University of Washington, Associate Member, Clinical Research Division, Fred Hutchinson Cancer Research Center
Related Content:
ESMO 2020: The TROPHY-U-01 Study- Sacituzumab Govitecan for Advanced Urothelial Carcinoma - Petros Grivas

