Figure 1 – Imvigor130 study design:
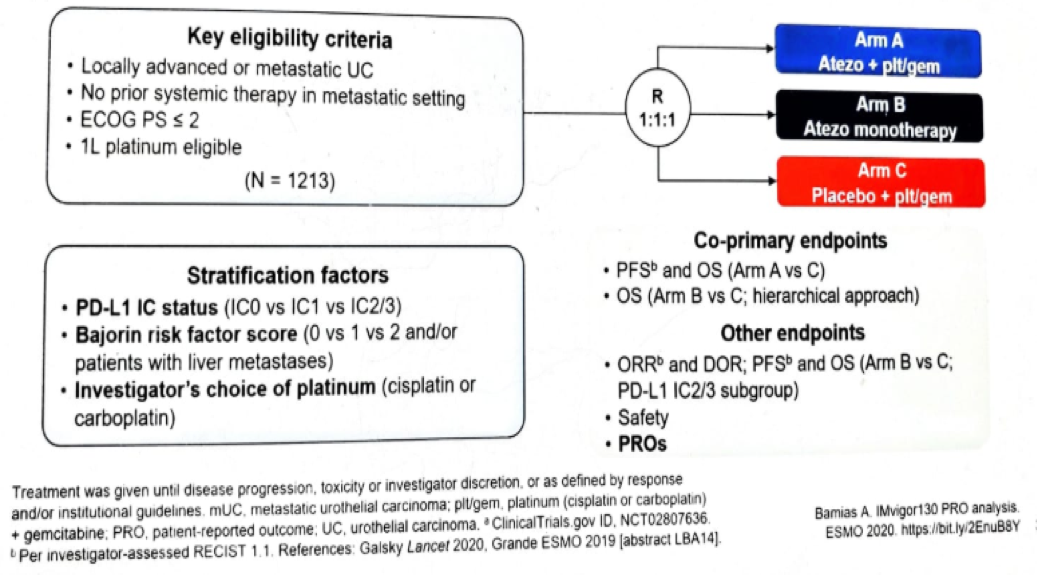
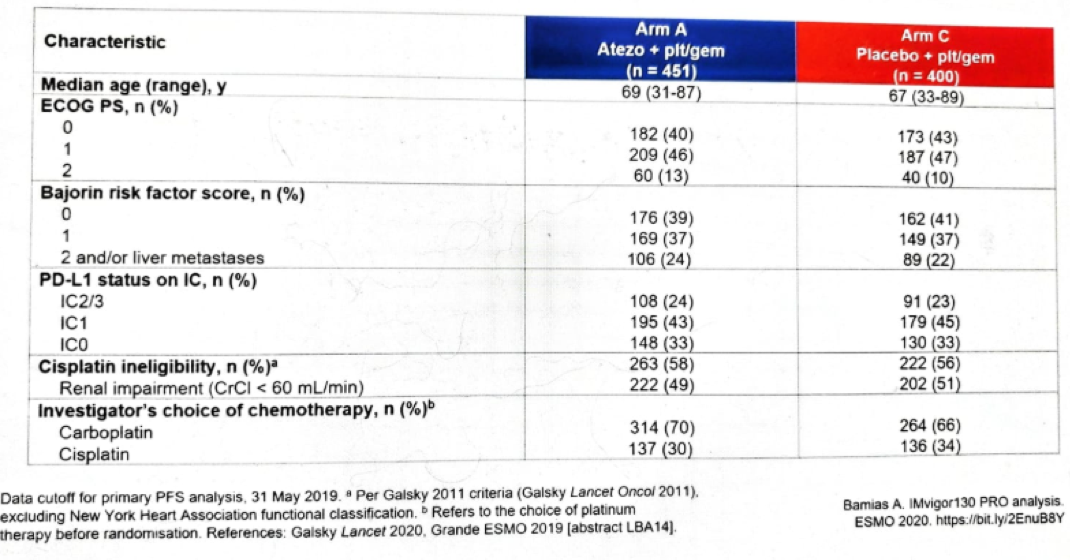
Figure 2 – Baseline characteristics:
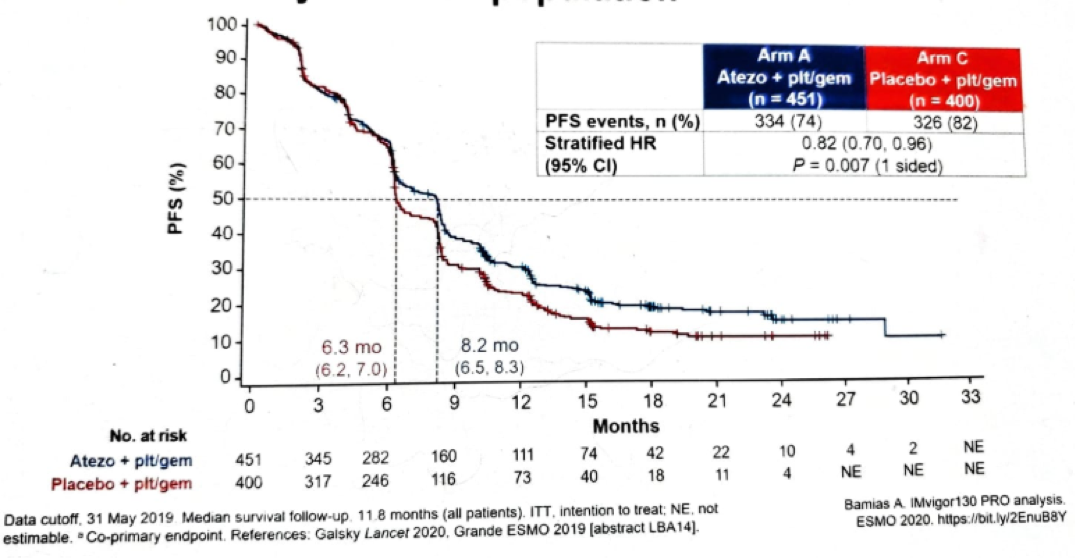
The final progression-free survival (PFS) in the intention to treat (ITT) population is shown in figure 3, demonstrating a hazard ratio (HR) in favor of the combination therapy of atezolizumab and chemotherapy compared to chemotherapy + placebo of 0.82 (95% CI 0.8-0.96), p=0.007, with a median PFS of 8.2 months compared to 6.3 months in the chemotherapy + placebo group. This finding is quite significant as it is the first immune checkpoint inhibitor study to demonstrate an improvement in PFS over the standard of care for first-line therapy in mUC.
Figure 3 – Final progression-free survival in the ITT population:
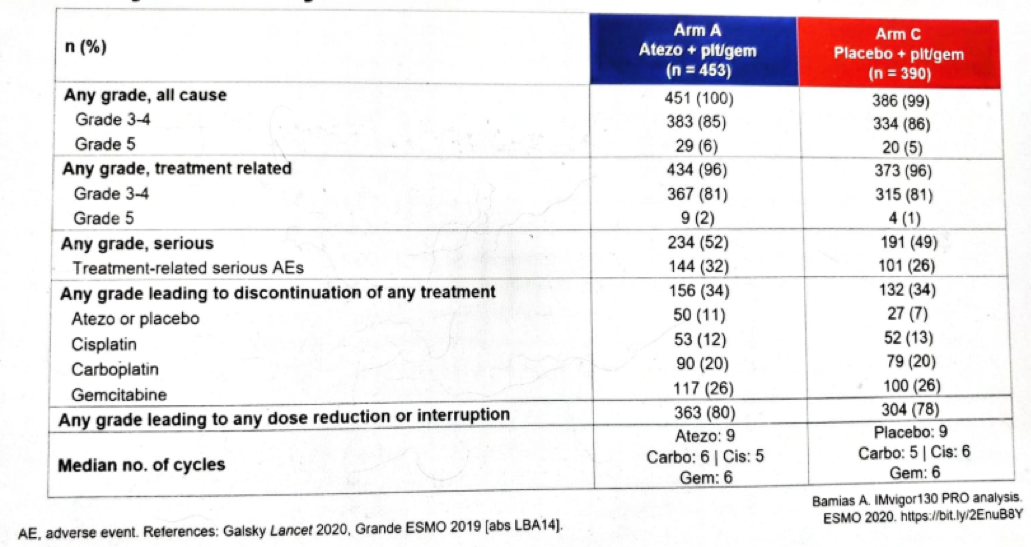
Next, Dr. Bamias showed the safety and adverse events data (Figure 4), showing no significant difference between the treatment arms.
Figure 4 – Safety summary:
One of the unique features of this trial is that it also provided data on patient-reported outcomes (PRO). PROs assessing the impact of disease and treatment on patient function and quality of life were evaluated to provide a comprehensive assessment of the patient treatment experience in both treatment arms. The instrument used, and assessment schedule that was used is shown in figure 5. The PROs were specified as secondary and exploratory endpoints without type 1 error control.
The PROs at baseline and the compliance rates are shown in figure 6. This shows that baseline scores were comparable between arms. Compliance rates were 86% at baseline and remained >=70% until week 111 in patients treated with atezolizumab and chemotherapy, and until week 66 in patients treated with chemotherapy + placebo. At week 51, approximately 23% of patients remained on study treatment and were eligible for PRO assessments.
Figure 5 – Patient-reported outcomes and assessment schedule:

Next, Dr. Bamias focused on the time to deterioration in different aspects, including role function (figure 7), physical function (Figure 8), emotional function (Figure 9), social function (Figure 10), and cognitive function (Figure 11).
Figure 6 – PROs at baseline:
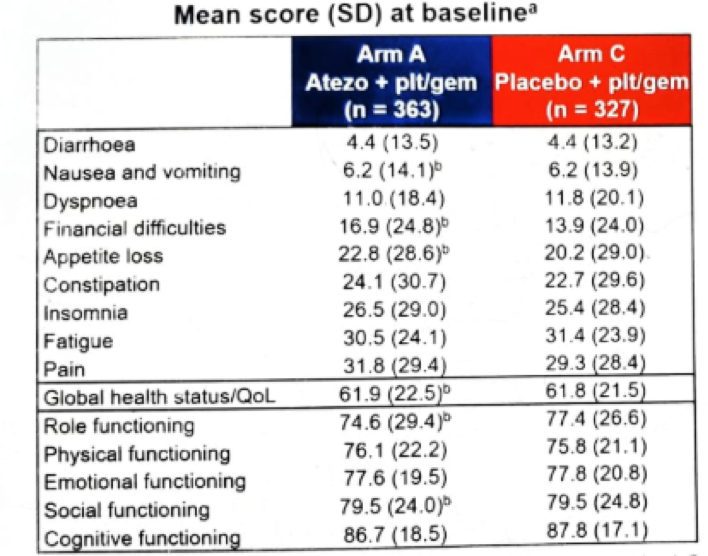
Figure 7 – Role function time to deterioration:
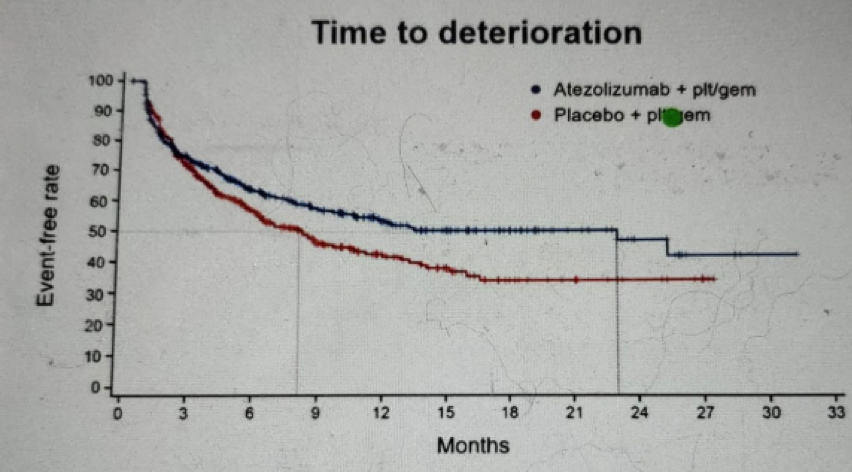
Figure 8 – Physical function time to deterioration:
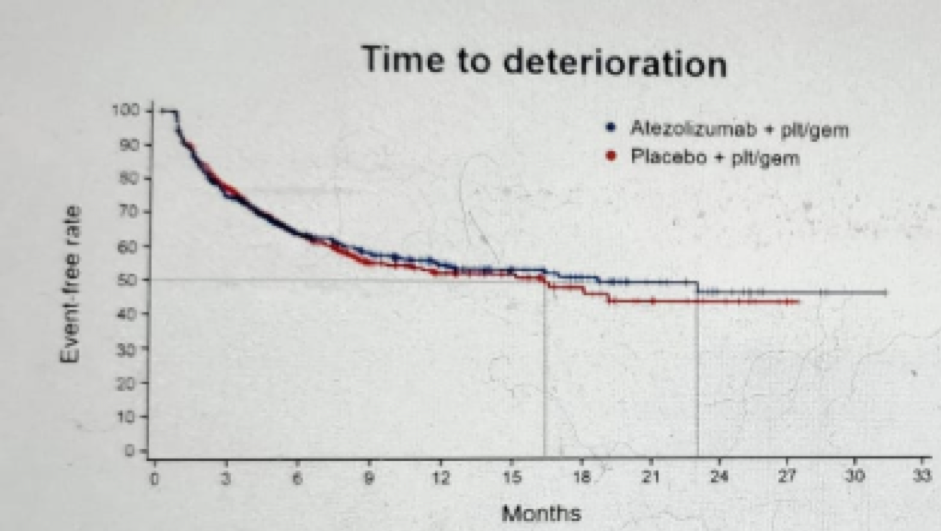
Figure 9 – Emotional function time to deterioration:
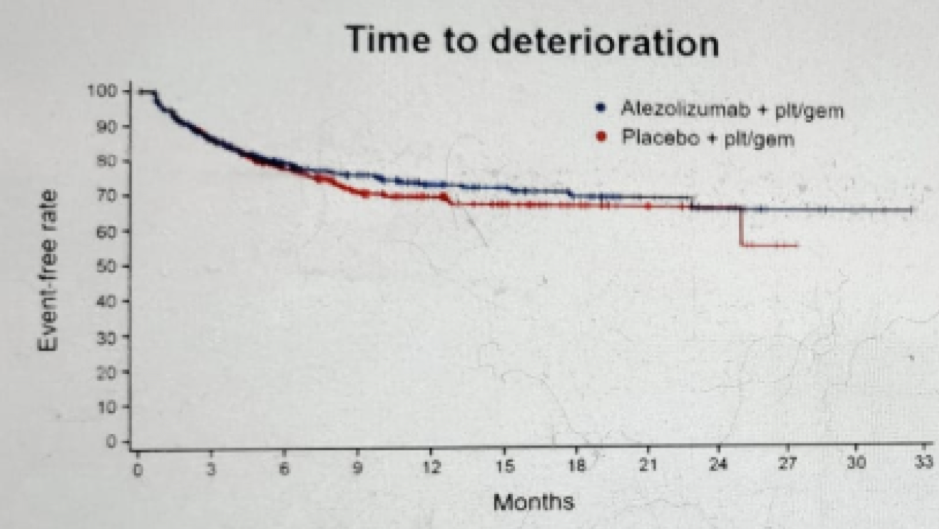
Figure 10 – Social function time to deterioration:
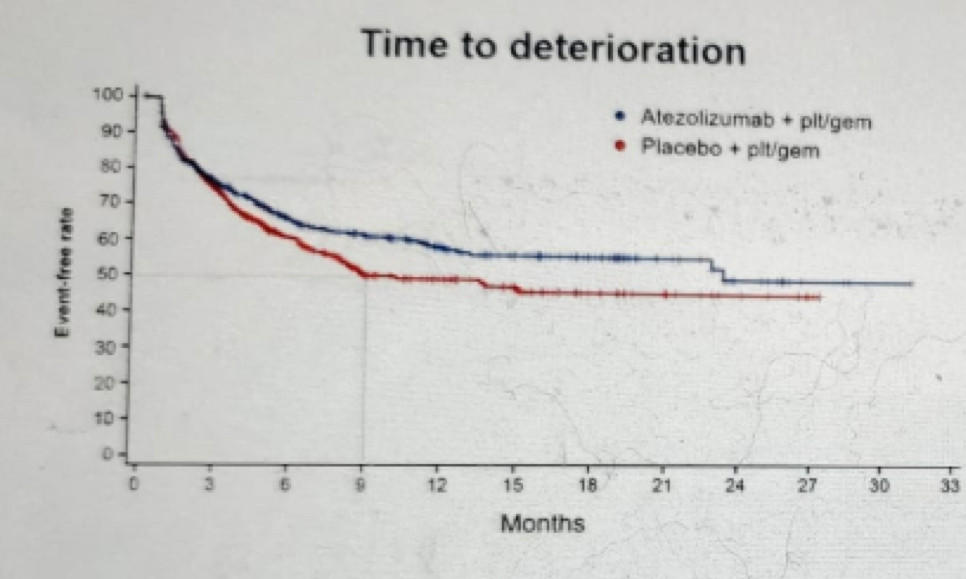
Figure 11 – Cognitive function time to deterioration:
In summary, IMvigor130 met its coprimary endpoint of PFS and showed a benefit for atezolizumab + chemotherapy vs. placebo + chemotherapy in the ITT population. Patients in both treatment arms reported similar nominal changes in most symptoms and function/quality of life domains, suggesting positive impacts overall. Analyses of the PROs showed that the addition of atezolizumab to platinum-based chemotherapy resulted in PFS improvement without compromising patients’ function or quality of life. These PRO results, coupled with the prolonged PFS and satisfactory safety profile, support the favorable benefit-risk profile of atezolizumab + platinum-based chemotherapy as an important new treatment option for patients with untreated mUC.
Presented by: Aristotelis Bamias, MD, National & Kapodistrian University of Athens, Athens, Greece
Written by: Hanan Goldberg, MD, MSc., Assistant Professor of Urology, SUNY Upstate Medical University, Syracuse, NY, USA, Twitter: @GoldbergHanan,

