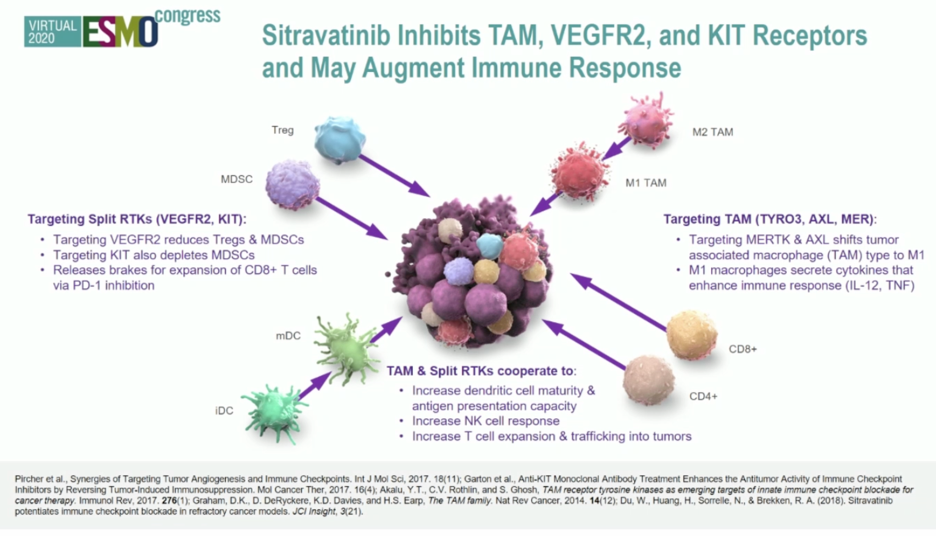
Based on early clinical data suggesting sitravatinib may increase the anti-tumor activity of the anti-PD-1 antibody nivolumab, a phase 2 study of this agent combined with immune checkpoint blockade in advanced urothelial carcinoma was established. In this presentation, Dr. Msaouel presented data from cohort 5, which enrolled patients who had previously received platinum chemotherapy but not anti-PD-1 axis therapy and treated them with combination sitravatinib and nivolumab. The trial schema is shown below:

Patient disposition and demographics at the time of data cutoff on July 30, 2020 is shown below. Notably, 90% of patients had visceral disease including 36% of patients with liver metastases.

The objective response rate was 37%, with 1 complete response.

Median follow-up has been short, 8.7 months, so longer follow-up will be required to fully assess overall survival data. Swimmer’s plots for patient clinical trajectory are illustrated below.

The most common adverse events reported were fatigue, diarrhea, decreased appetite, dysphonia, elevated ALT, and hypertension. The most common grade ¾ toxicity was hypertension. A full listing of frequent treatment related adverse events is shown.

In summary, the combination of sitravatinib and nivoluman shows clinical activity in platinum refractory advanced urothelial carcinoma patients that have not previously been treated with immune checkpoint blockade. The adverse event profile is generally consistent with tyrosine kinase inhibitor therapy. Evaluation of this drug combination across various patient cohorts is ongoing under clinical trial NCT03606174
Presented by: Pavlos Msaouel, MD, PhD, Assistant Professor in the Department of Genitourinary Medical Oncology, Division of Cancer Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX
Written by: Alok Tewari, MD, PhD, Medical Oncologist at the Dana-Farber Cancer Institute at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020

