(UroToday.com) Immune checkpoint blockade therapy by blocking PD-1/PD-L1 axis signaling is approved in advanced urothelial cancers in a few settings: (1) first-line therapy for platinum-ineligible patients, (2) first-line therapy in cisplatin-ineligible patients with high PD-L1 expression, (3) maintenance therapy for patients who do not progress with first-line chemotherapy, and (4) as subsequent therapy for patients progressing after cisplatin-based first-line therapy. Approvals for settings #1 and #2 is based on single-arm phase 2 data, with setting #3 based on the JAVELIN-100 Bladder randomized control trial. Durvalumab, an anti-PD-L1 antibody, is one of the immunotherapy agents approved for setting #4.
Given the suggestion that combination anti-PD-L1 therapy (durvalumab) and anti-CTLA4 therapy (tremelimumab) has activity in platinum-refractory advanced urothelial carcinoma regardless of PD-L1 status, the DANUBE study was designed as a randomized phase 3 study to investigate durvalumab monotherapy versus durvalumab + tremelimumab versus platinum-based chemotherapy in the first-line setting. The schema of the trial is below. Importantly, based on prior data, the primary endpoint for the combination immunotherapy arm was overall survival (OS) in the intention-to-treat population. The primary endpoint in the durvalumab monotherapy arm was overall survival in the PD-L1 positive population. PD-L1 status was measured by either tumor or immune cell positivity.
The statistical analysis plan entailed a split overall alpha between the durvalumab monotherapy comparison to chemotherapy and combination immunotherapy comparison to chemotherapy.
Baseline characteristics were generally well balanced between all treatment groups. 60% of patients in all arms had high PD-L1 expression.

There was no overall survival benefit for durvalumab monotherapy in the PD-L1 high population relative to chemotherapy (HR 0.89, 0.71 – 1.11, p = 0.3039).

There was also no overall survival benefit with combination durvalumab and tremelimumab therapy compared to chemotherapy in the intention to treat population, with a median OS of 15.1 months from combination immunotherapy and 12.1 months with chemotherapy (HR 0.85, 0.72-1.02, p = 0.0751).

As shown in the chart below, in the intention to treat population, chemotherapy had the highest objective response rate (ORR) rate. Interestingly, in the PD-L1 high subgroup (exploratory analysis), the combination immunotherapy group had an ORR comparable to chemotherapy.
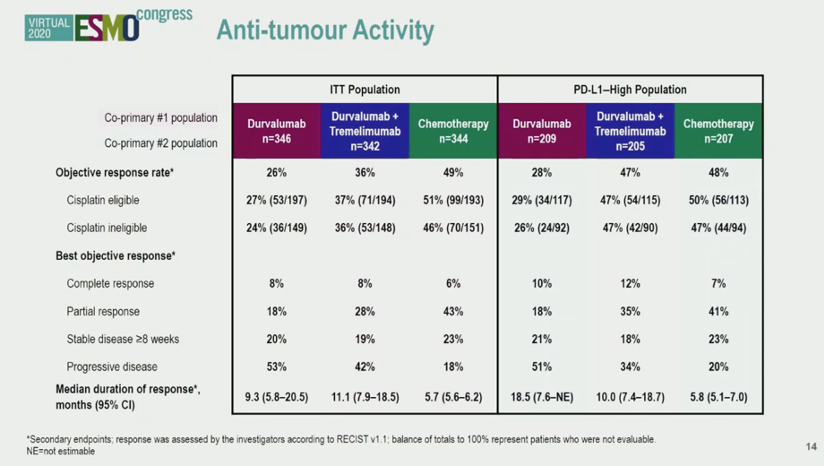
As an exploratory secondary endpoint, combination immunotherapy in PD-L1 high patients resulted in a median overall survival of 17.9 months compared to 12.1 months with chemotherapy alone (HR 0.74, 0.59-0.93).

Equivalent rates of patients in each treatment arm went on to receive anti-cancer therapy.

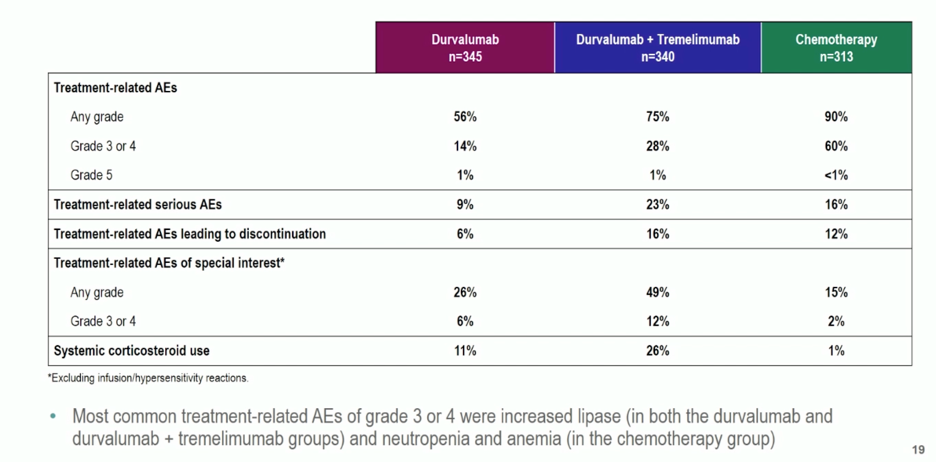
Durvalumab monotherapy or combination immunotherapy did not have higher rates of treatment-related adverse events, though more patients receiving combination immunotherapy discontinued treatment due to adverse events. The most common grade 3+ adverse events in the immunotherapy arms was lipase elevation.
In summary, the DANUBE trial was negative for its co-primary endpoints of overall survival in (1) durvalumab monotherapy versus chemotherapy in the PD-L1 high patient population and (2) combination durvalumab and tremelimumab versus chemotherapy in the intention-to-treat population.
The intriguing results from the secondary endpoint analysis of combination immunotherapy in the PD-L1 high population warrants further investigation. Given the longer duration of response with immunotherapy, identification of which patients are likely to benefit from immunotherapy remains a critical question for investigation moving forward.
Presented by: Thomas Powles, MBBS, MRCP, MD, Professor of Genitourinary Oncology, Lead for Solid Tumour Research at Barts Cancer Institute, Director of Barts Cancer Institute, London, United Kingdom
Written by: Alok Tewari, MD, PhD, Medical Oncologist at the Dana-Farber Cancer Institute, at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Related Content:
Read the Invited Discussion on this Topic: ESMO Virtual Congress 2020: Invited Discussant KEYNOTE-361 (LBA23) and DANUBE (697O)
ESMO 2020: Combination Therapies in Locally Advanced or Metastatic Urothelial Carcinoma Bladder Cancer - Josh Meeks

