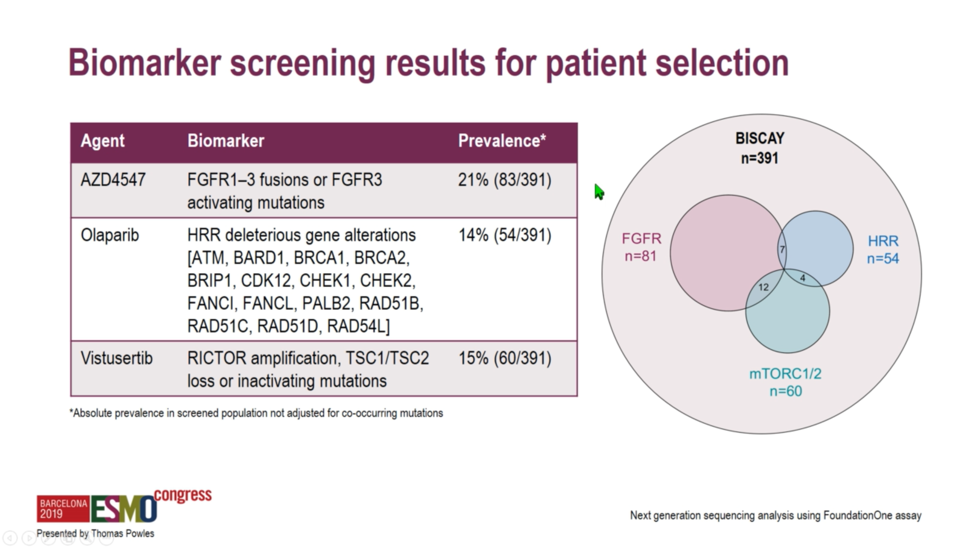
 Patients were assigned to treatment arms on the basis of various biomarkers as assessed by the Foundation Medicine assay. Patients with activating FGFR1, FGFR2, of FGFR3 mutations or fusions were assigned to the FGFR inhibitor AZD4547 with or without the PD-L1 antibody durvalumab. Patients with ATM, BRCA1, BRCA2, or otherwise harboring deleterious mutations in a number of homologous recombination repair enzymes were assigned to receive olaparib with durvalumab. A partially-selected arm with RICTOR amplification or deleterious mutations in TSC1 or TSC2 was assigned to vistusertib plus durvalumab, and finally, an unselected arm was assigned to durvalumab monotherapy to serve as a benchmark against which to interpret response rates in the other arms.
Patients were assigned to treatment arms on the basis of various biomarkers as assessed by the Foundation Medicine assay. Patients with activating FGFR1, FGFR2, of FGFR3 mutations or fusions were assigned to the FGFR inhibitor AZD4547 with or without the PD-L1 antibody durvalumab. Patients with ATM, BRCA1, BRCA2, or otherwise harboring deleterious mutations in a number of homologous recombination repair enzymes were assigned to receive olaparib with durvalumab. A partially-selected arm with RICTOR amplification or deleterious mutations in TSC1 or TSC2 was assigned to vistusertib plus durvalumab, and finally, an unselected arm was assigned to durvalumab monotherapy to serve as a benchmark against which to interpret response rates in the other arms. 
All patients were required to have locally advanced or metastatic urothelial carcinoma and have progressed on at least one prior line of platinum-based chemotherapy. Biomarker analysis required archival tissue from within three years of study enrollment. Of 391 patients screened, 81 were assigned to the FGFR arm, 60 the mTORC1/2 arm, and 54 to the homologous recombination repair deficiency arm.
 A 50% objective response rate was set as a threshold for the ongoing study of select immune-oncology combinations. Unfortunately, investigators found that none of the study arms reached this threshold, with response rates ranging from 20.0% with AZD4547 monotherapy to 35.7% with olaparib plus durvalumab. Interestingly, PD-L1 expression was lowest at 23%-25% among the FGFR-altered tumors, perhaps explaining why durvalumab plus AZD4547 only achieved a 28.6% response rate. While the combination of the PARP inhibitor olaparib and durvalumab achieved the highest numerical response rate at 35.7%, these homologous recombination repair-deficient tumors also exhibited the highest tumor mutational burden of any module, which perhaps explains the modest increase in response rate relative to the 27.6% rate observed with durvalumab monotherapy.
A 50% objective response rate was set as a threshold for the ongoing study of select immune-oncology combinations. Unfortunately, investigators found that none of the study arms reached this threshold, with response rates ranging from 20.0% with AZD4547 monotherapy to 35.7% with olaparib plus durvalumab. Interestingly, PD-L1 expression was lowest at 23%-25% among the FGFR-altered tumors, perhaps explaining why durvalumab plus AZD4547 only achieved a 28.6% response rate. While the combination of the PARP inhibitor olaparib and durvalumab achieved the highest numerical response rate at 35.7%, these homologous recombination repair-deficient tumors also exhibited the highest tumor mutational burden of any module, which perhaps explains the modest increase in response rate relative to the 27.6% rate observed with durvalumab monotherapy.  No striking differences between progression-free or overall survival were observed in the randomized arm of AZD4547 with or without durvalumab, suggesting that there is no meaningful difference in response durability associated with the combination.
No striking differences between progression-free or overall survival were observed in the randomized arm of AZD4547 with or without durvalumab, suggesting that there is no meaningful difference in response durability associated with the combination. 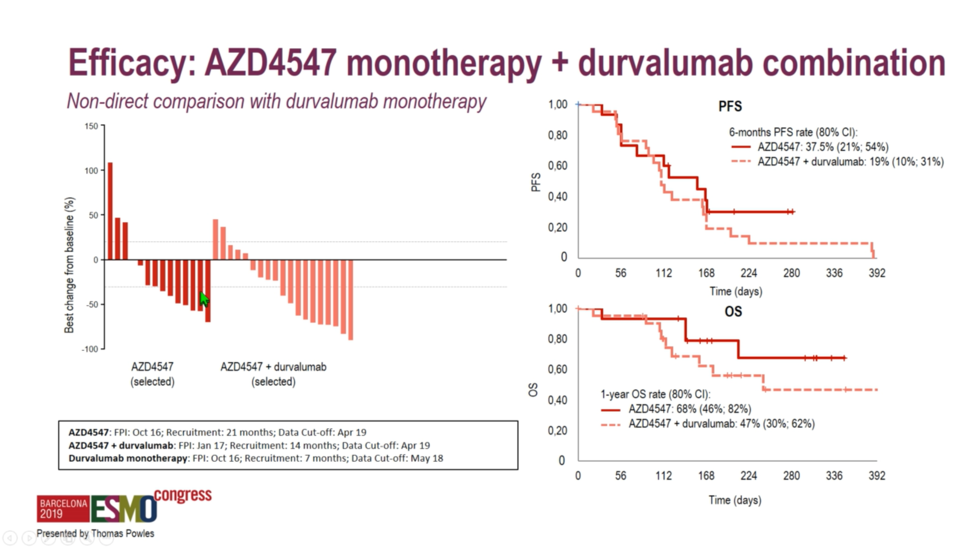 When benchmarked against durvalumab monotherapy, the homologous recombination repair deficiency arm and the mTOR targeting arm did not exhibit markedly different survival curves relative to the durvalumab arm. However, it should be noted that direct comparisons between the combination arms and the durvalumab monotherapy arm cannot be made, as patients were selected on the basis of biomarkers for the combination arms and were not randomized.
When benchmarked against durvalumab monotherapy, the homologous recombination repair deficiency arm and the mTOR targeting arm did not exhibit markedly different survival curves relative to the durvalumab arm. However, it should be noted that direct comparisons between the combination arms and the durvalumab monotherapy arm cannot be made, as patients were selected on the basis of biomarkers for the combination arms and were not randomized.
 With respect to safety, grade 3 and grade 4 treatment-related adverse events occurred in 10.3% of the durvalumab monotherapy arm, and each of the combination arms exhibited slightly higher rates of grade ≥3 adverse events, which were highest in the AZD4547 combination arm at 47.6 percent, which led to discontinuation or dose reduction in 81% of patients in the AZD4547 plus durvalumab arm, perhaps suggesting that the combination would have been more efficacious should higher doses been deliverable.
With respect to safety, grade 3 and grade 4 treatment-related adverse events occurred in 10.3% of the durvalumab monotherapy arm, and each of the combination arms exhibited slightly higher rates of grade ≥3 adverse events, which were highest in the AZD4547 combination arm at 47.6 percent, which led to discontinuation or dose reduction in 81% of patients in the AZD4547 plus durvalumab arm, perhaps suggesting that the combination would have been more efficacious should higher doses been deliverable.  In summary, BISCAY was a novel biomarker-directed combination immune-oncology trial in platinum-refractory metastatic urothelial cancer. This small trial was designed to detect large magnitude clinical differences associated with the combination of PD-L1 blockade and biomarker-selected targeted therapy. Unfortunately, response rates to anti-PD-L1 therapy were not markedly amplified by the combination with targeted agents. Future work will explore how to best combine targeted therapy with checkpoint blockade to further improve response rates in this area of high unmet need.
In summary, BISCAY was a novel biomarker-directed combination immune-oncology trial in platinum-refractory metastatic urothelial cancer. This small trial was designed to detect large magnitude clinical differences associated with the combination of PD-L1 blockade and biomarker-selected targeted therapy. Unfortunately, response rates to anti-PD-L1 therapy were not markedly amplified by the combination with targeted agents. Future work will explore how to best combine targeted therapy with checkpoint blockade to further improve response rates in this area of high unmet need.Presented by: Thomas Powles, MBBS, MRCP, MD, Professor of Genitourinary Oncology, Lead for Solid Tumour Research at Barts Cancer Institute, Director of Barts Cancer Institute, London UK
Written by: Michael Lattanzi, MD, Medical Oncology Fellow, Memorial Sloan Kettering Cancer Center, @MikeLattanzi, at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept - 1 Oct 2019 in Barcelona, Spain

