(UroToday.com) The 2023 European Association of Urology (EAU) annual congress held in Milan, Italy between March 10th and 13th, 2023 was host to a session addressing precision medicine in patients with metastatic castrate-resistant prostate cancer (mCRPC). Dr. Stefano Fanti delivered a state-of-the-art lecture discussing best predictive factors for successful treatment with LuPSMA.
The core principle of theranostics is that we “treat what we can see”. As such, it appears quite intuitive that patients with significant uptake of the tracer on PSMA-PET/CT would derive the most benefit from PSMA-targeted theranostics.

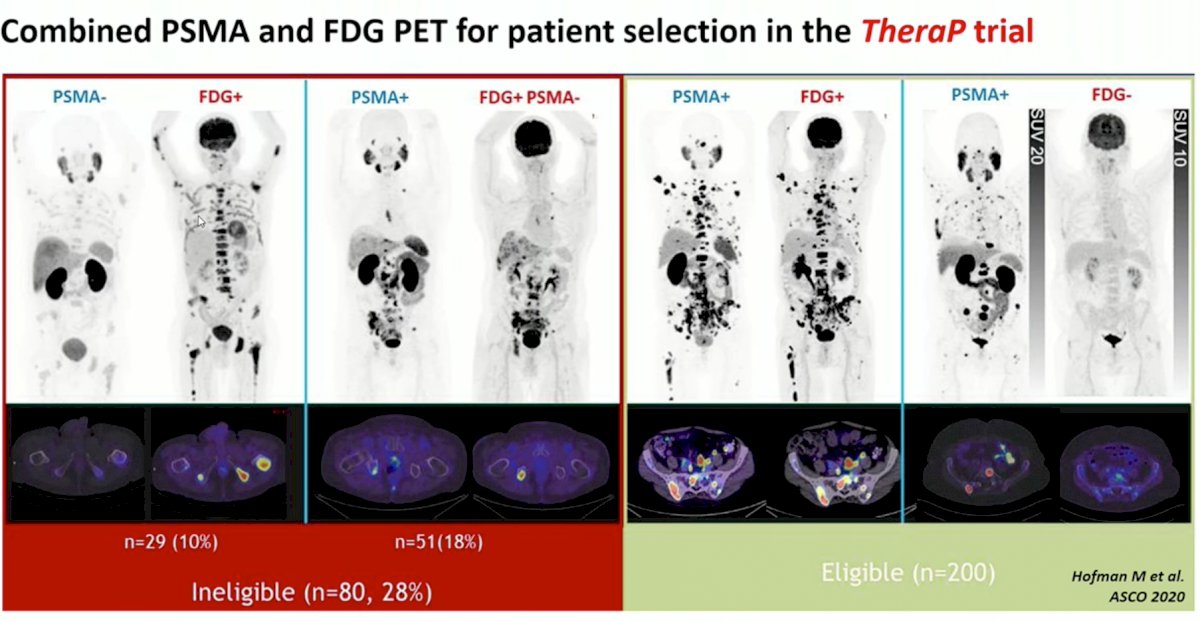
What is currently the biggest problem with theranostics? Dr. Fanti believes it is the non-responders. In the TheraP trial of 177Lu-PSMA-617 versus cabazitaxel in the 3rd line mCRPC setting, we see that a significant proportion of patients in the 177Lu-PSMA-617 did not respond to therapy (denoted in blue in the figure below). This occurred even after patients were enrolled on the basis of a 68Ga-PSMA-11 scan showing high PSMA-expression (at least one site with SUVmax≥20) and an 18F-FDG demonstrating no sites of disease of FDG-positive and PSMA-negative (i.e., discordant) disease.1
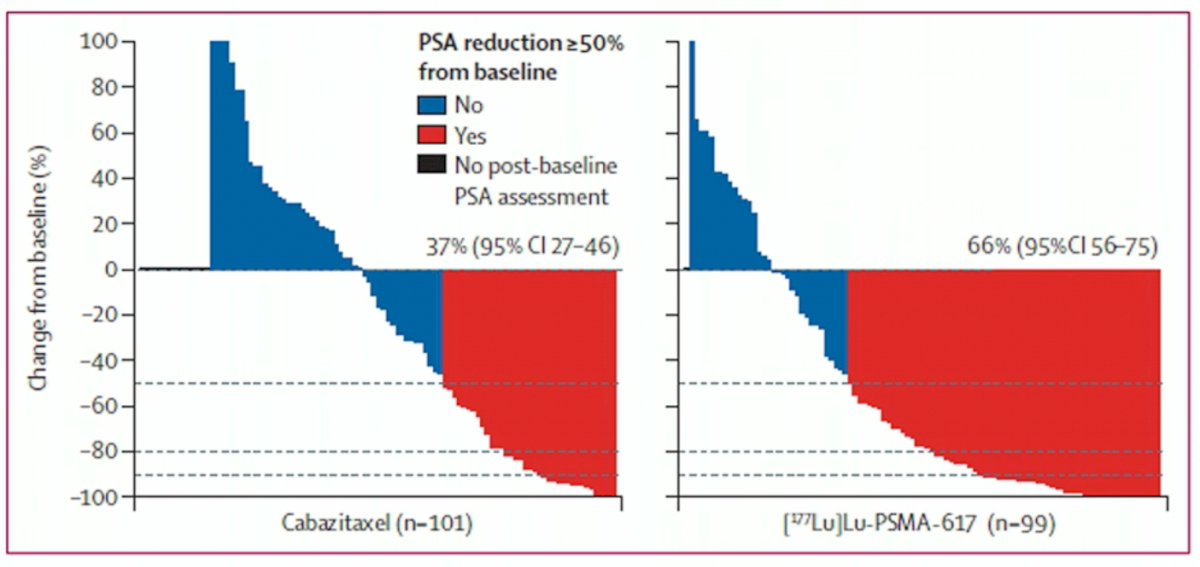
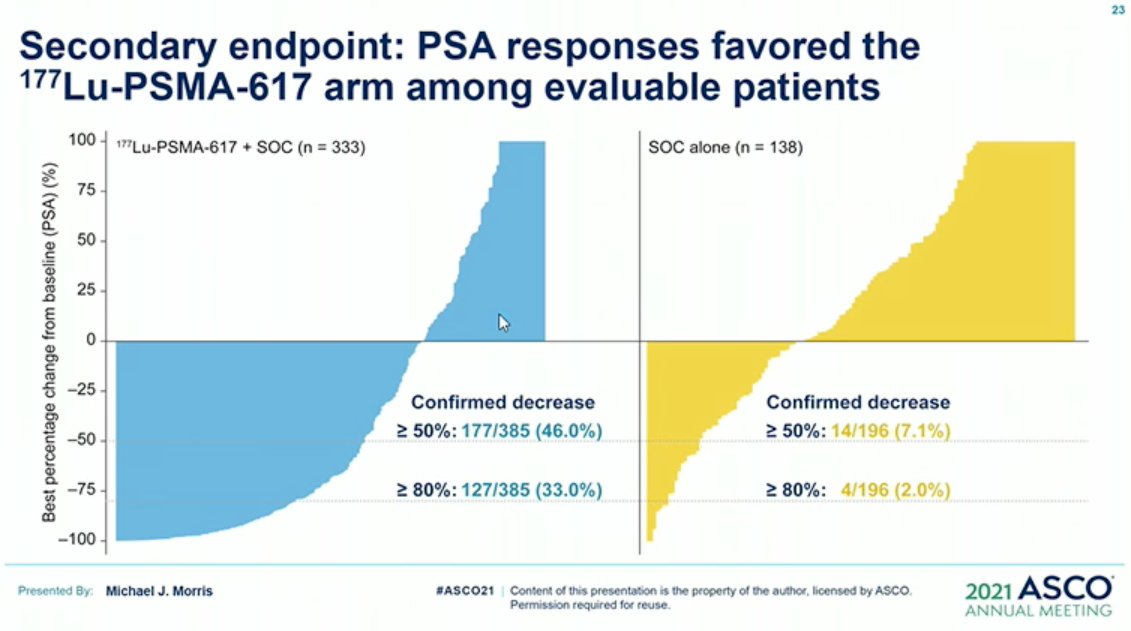
A similar situation was observed in the VISION trial of 177Lu-PSMA-617 versus alternate ARPi in patients with PSMA-positive mCRPC who had previously received treatment with next-generation androgen receptor signaling inhibition (abiraterone, enzalutamide, etc) and one or two prior lines of taxane chemotherapy. Even after applying exclusion criteria (e.g., liver metastasis <2.5 cm) based on CT/MRI findings, a significant proportion of patients in the experimental arm did not derive a clinical benefit from 177Lu-PSMA-617.2
What can we do to reduce the proportion of non-responders? Again, treat what we can see (fundamental principle of theranostics). Clearly, relying on the findings from a positive 68Ga PSMA-11 along with a negative FDG PET, as in the TheraP trial is not sufficient (28% of patients ineligible based on the imaging criteria).
Similarly, as seen in the VISION trial, relying on CT/MRI findings alone, does not minimize the proportion of non-responders.

Current commonly accepted eligibility criteria for 177Lu-PSMA therapy, include:
1. PSMA uptake above the liver in the majority of the lesions (small lesions excluded)
2. Absence of PSMA-negative lesions >1 cm (liver, lung, lymph nodes)
Conversely, predictive factors of response to 177Lu-PSMA therapy, include:
1. High whole-body SUVmean
2. Homogenous target expression
3. Low whole body tumor volume
Post-hoc analysis from TheraP demonstrated that the odds of a PSA response to 177Lu-PSMA-617 versus cabazitaxel were significantly higher for men with SUVmean of 10 or higher compared with those with SUVmean of less than 10 (odds ratio [OR]: 12.19 [95% CI: 3.42-58.76] vs 2.22 [1.11-4.51].3

Furthermore, patients with a lower PSMA-PET whole body tumor volume (prognostic variable) have superior survival outcomes compared to those with higher volume of PSMA-positive disease:

Based on results from the VISION trial, Gafita et al. developed a 17-variable nomogram that predicts outcome after LuPSMA therapy. This can be easily used as a decision aid tool to help counsel patients on expected response/prognosis. Dr. Fanti emphasized that it is important that we use such tools as decision aids, not treatment selection tools (i.e. do not choose patients solely based on the results of such nomograms).4

Can we use interim PET results as a prognostic/predictive tool? Calais et al. form UCLA have endorsed the use of an interim PET after 3 cycles of 177Lu-PSMA-617. Dr. Fanti argued however that if an interim PET is to be used to assess response, 3 cycles is probably too late to do so and patients should probably be assessed earlier.
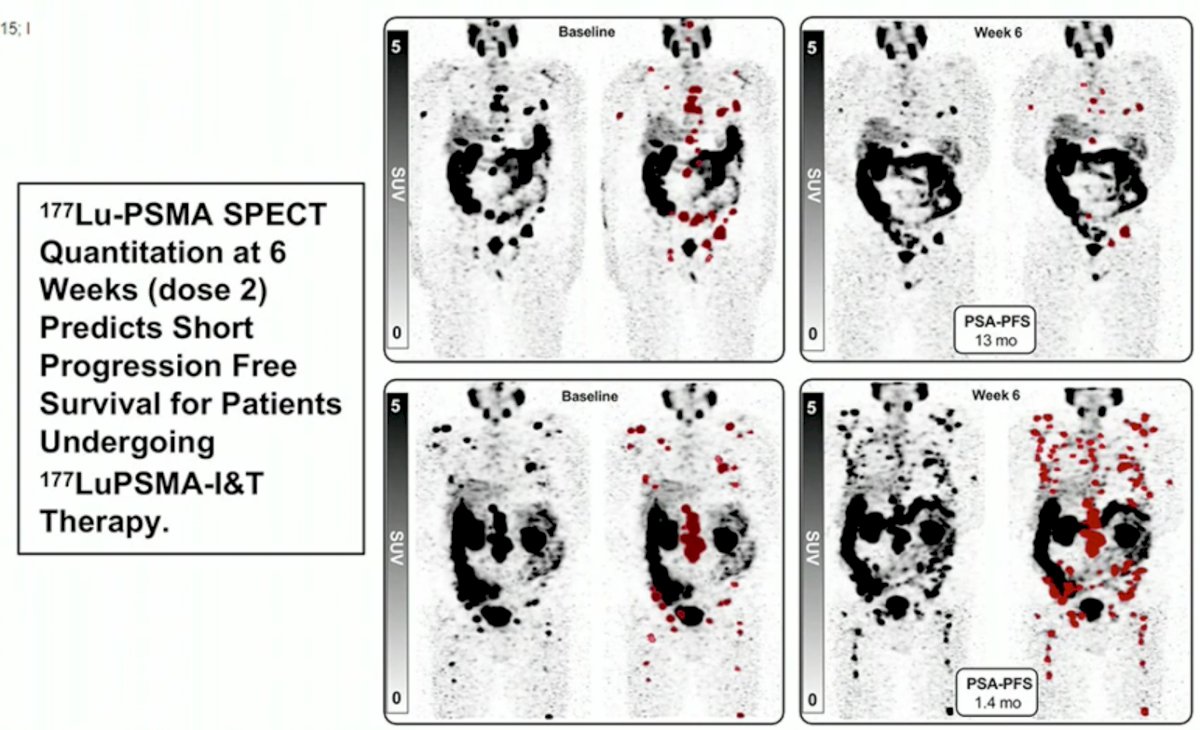
Conversely, performance of 177Lu-PSMA SPECT quantitation at 6 weeks (dose 2) predicts short PFS for patients undergoing 177LuPSMA-I&T therapy:
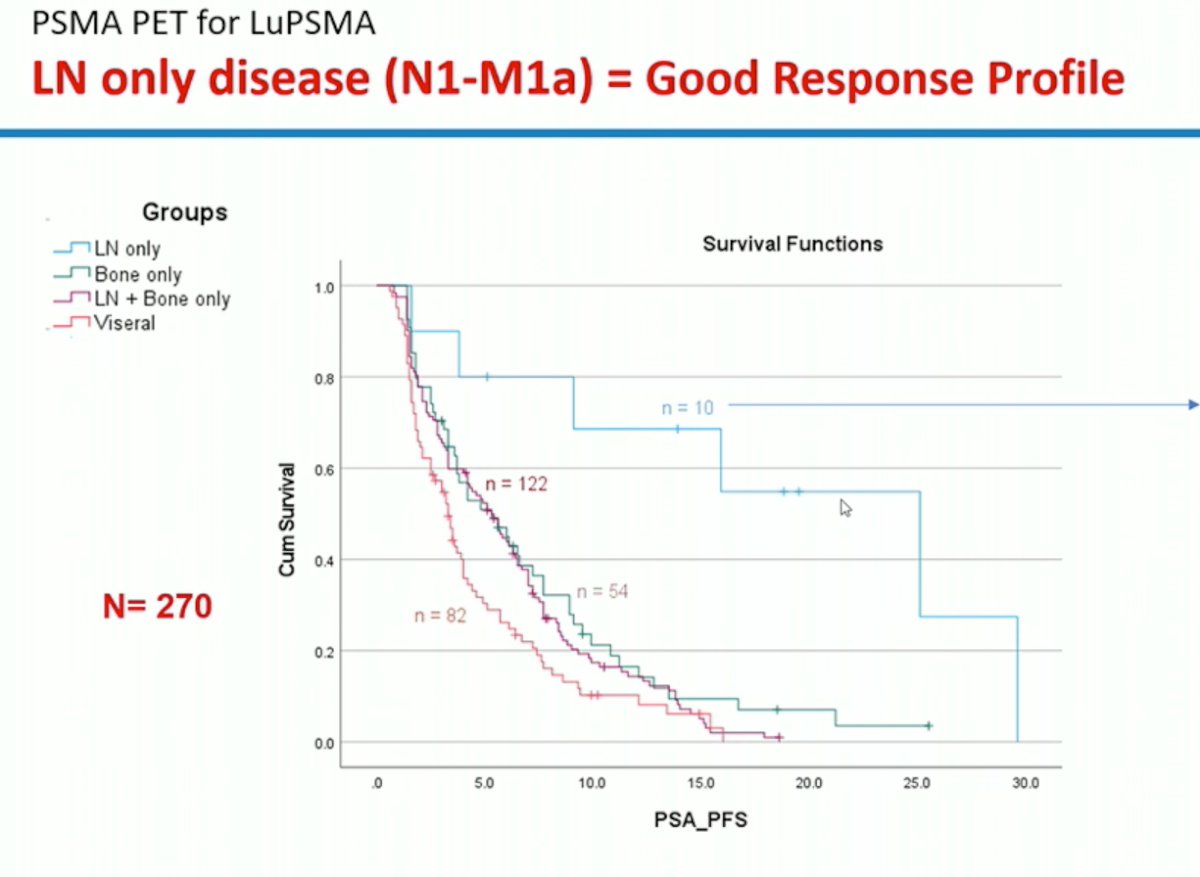
How do we maximize the proportion of patients likely to benefit from 177Lu-PSMA theranostics in real practice? While PSMA-PET findings are useful as prognostic/predictive biomarkers, we cannot forget about routine clinical factors that risk stratify our patients and help guide treatment decisions routinely. As seen with other treatments in both the mHSPC and mCRPC disease settings, patients with LN only disease have a good response profile with 177Lu-PSMA therapy.
Other factors include the Gleason Score, improved performance status, younger age, etc. as outlined in the table below:

In summary, Dr. Fanti emphasized:
- Use nomograms as an outcomes predictor, not a treatment selector
- Treat what you see (basic principle of theranostics)
Presented by: Dr. Stefano Fanti, MD, Professor, Department of Experimental, Diagnostic and Specialty Medicine, Universita di Bologna, Bologna, Italy
Written by: Rashid K. Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 European Association of Urology (EAU) 38th annual congress held in Milan, Italy between March 10th and 13th, 2023
References- Hofman, et al. Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet 2021. 797-804.
- Sartor, et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med 2021. 385:1091-1103.
- Buteau JP, et al. Continuing enzalutamide with docetaxel in castration-resistant prostate cancer. Lancet Oncol 2021. 1345-1347.
- Gafita A, et al. Nomograms to predict outcomes after 177Lu-PSMA therapy in men with metastatic castration-resistant prostate cancer: an international, multicentre, retrospective study. Lancet Oncol 2021. 1115-1125.

