(UroToday.com) The 2023 European Association of Urology (EAU) annual congress held in Milan, Italy between March 10th and 13th, 2023 was host to a rapid-fire debate session addressing common problems and controversies in bladder cancer, chaired by Dr. Ashish Kamat.
Following a case presentation by Dr. Jason Efstathiou, Dr. Andrea Necchi was tasked with debating in favor of systemic therapy alone as a sufficient, optimal modality for the management of bladder cancer patients with oligometastatic disease. Conversely, Dr. Aristotelis Bamias was tasked with arguing in favor of local therapy having a role in this setting.
Dr. Efstathiou presented the case of a 56-year-old male with oligometastatic urothelial bladder cancer with a sacro-iliac metastatic lesion. At this point, the discussion was turned over to Dr. Necchi to argue in favor of systemic therapy alone for this patient.

One of the major issues currently remains the lack of a consensus definition for ‘oligometastatic’ urothelial carcinoma, and this has important implications for modern prognostic allocation and treatment recommendations. The AJCC 7th edition Stage IV definition includes patients with:
- T4bN0M0
- TanyN1-3M0
- TanyNanyM1
Dr. Necchi advocated for the use of the term ‘first line’ therapy as a more appropriate term than ‘neoadjuvant’ as this latter term implies that local therapy is expected and planned. All platinum-eligible patients are suited for primary chemotherapy. What remains unknown is whether oligometastatic urothelial carcinoma harbors biologic features/mutations that are distinct to those seen in patients with Muscle Invasive Bladder ancer (MIBC) or visceral metastases. However, newer systemic therapy combinations and/or sequencing regimens have significantly improved outcomes regardless of the disease extent.
Clearly, we have seen an evolution in the treatment paradigm of patients in the metastatic setting with the emergence of immunotherapy agents, either alone or in combination. What is the evidence from the ‘pre-modern’ era?
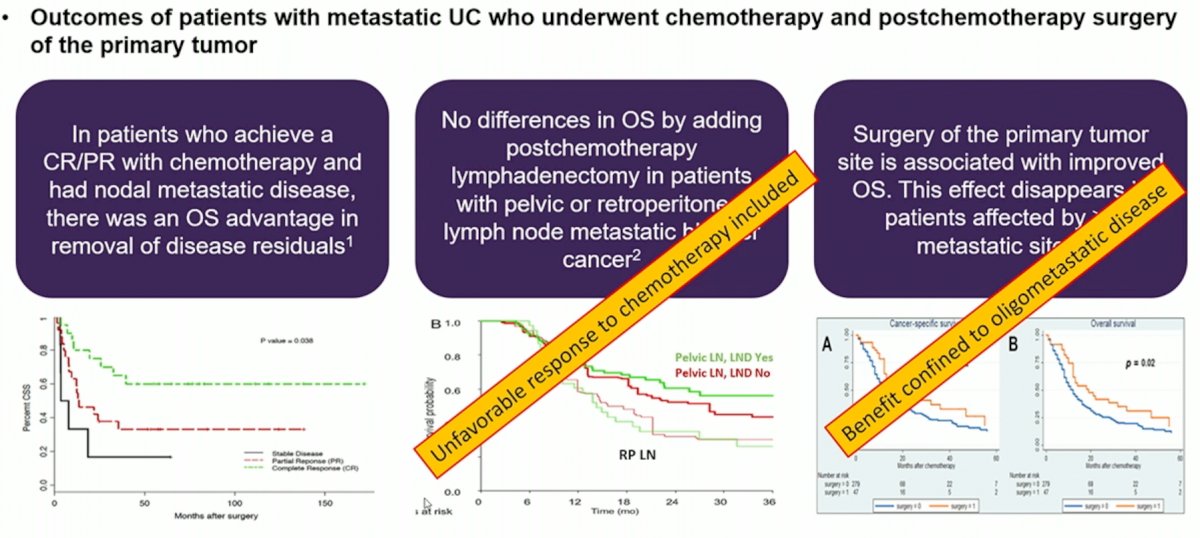
In patients who achieve a complete or partial response with chemotherapy and had nodal metastatic disease only, there was an OS advantage in removal of residual disease.1
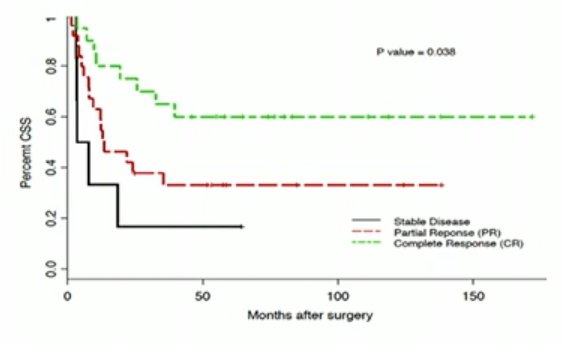
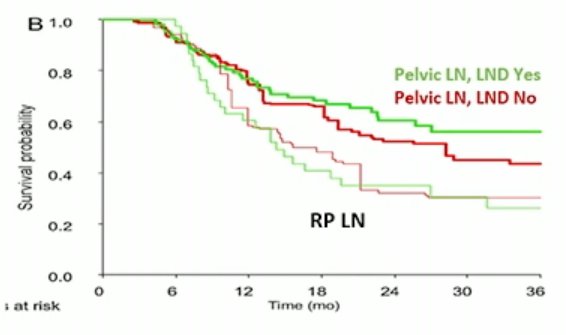
Conversely, there are no OS benefits by adding post-chemotherapy lymphadenectomy in patients with pelvic or retroperitoneal lymph node metastatic bladder cancer.=, albeit this study included patients irrespective of chemotherapy response.2
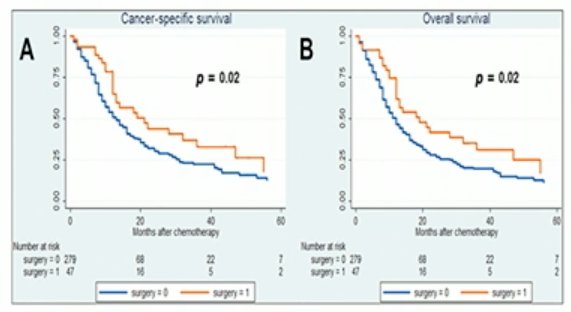
Surgery of the primary tumor site may be associated with improved overall survival. However, this effect disappears in patients affected by 2 or more metastatic sites.3
What about patient outcomes in the modern era? Dr. Necchi highlighted his group’s recent editorial in European Urology that discusses whether it is time to consider eliminating surgery from the treatment paradigm of locally advanced bladder cancer. The evolving reality is that patients who achieve a partial or complete response with immunotherapeutic agents have much improved prognoses compared to historical references. This was demonstrated in the KEYNOTE-361 trial evaluating pembrolizumab addition to chemotherapy, whereby patients achieving a PR/CR at 9 weeks had marked clinical benefits.4

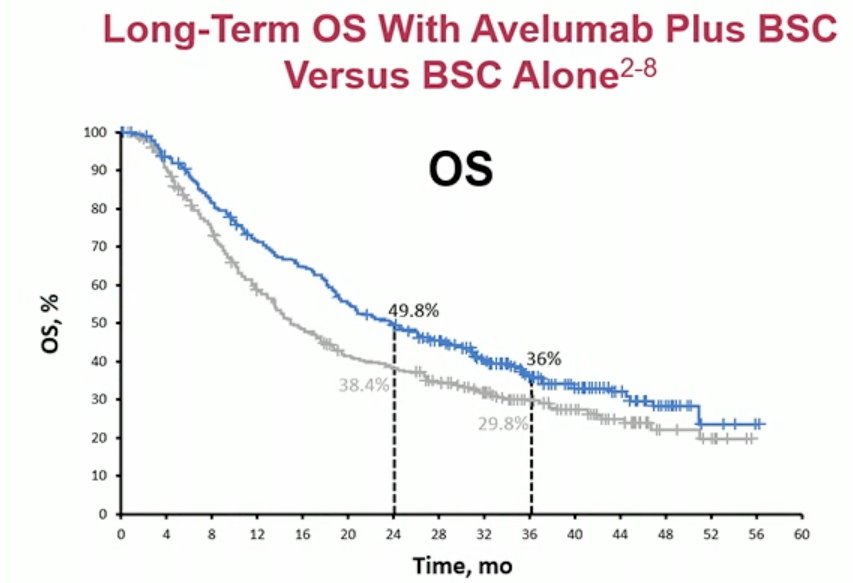
Furthermore, based on the results of JAVELIN-100 demonstrating an OS benefit, avelumab has emerged as an FDA-approved agent in the switch-maintenance treatment of locally advanced or metastatic urothelial carcinoma that has not progressed with first-line platinum chemotherapy.5 As such not only are agents in this setting associated with improved outcomes compared to historical references, but newer agents in novel treatment settings are becoming increasingly available.
Additionally, the combination of immunotherapeutic agents (pembrolizumab) and targeted agents (anti-Nectin-4: enfortumab vedotin) has been evaluated in the 1st line cis-ineligible setting for patients with locally advanced or metastatic urothelial carcinoma. EV-103 cohort K assessed the efficacy of enfortumab vedotin monotherapy or in combination with pembrolizumab in previously untreated cisplatin-ineligible patients with locally advanced or metastatic urothelial cancer. Patients in this trial were randomized 1:1 to EV (1.25 mg/kg) as monotherapy on Days 1 and 8 or in combination with pembrolizumab (200 mg) on Day 1 of 3-week cycles (EV-103, NCT03288545). The primary endpoint of this study was confirmed ORR, per RECIST v1.1 by blinded independent central review. 149 patients were enrolled. The confirmed ORR was 64.5% (95%CI 52.7 to 75.1) among patients treated with the combination of EV and pembrolizumab, with a median duration of response not reached. Among those treated with EV monotherapy, the confirmed ORR was 45.2% (95% CI 33.5 TO 57.3) and median DOR was 13.2 months (95% CI 6.1 to 16.0).

Furthermore, modern bladder-sparing strategies in T2-4N0M0 MIBC may impact the subsequent decision-making for N1-3 or M1 patients.
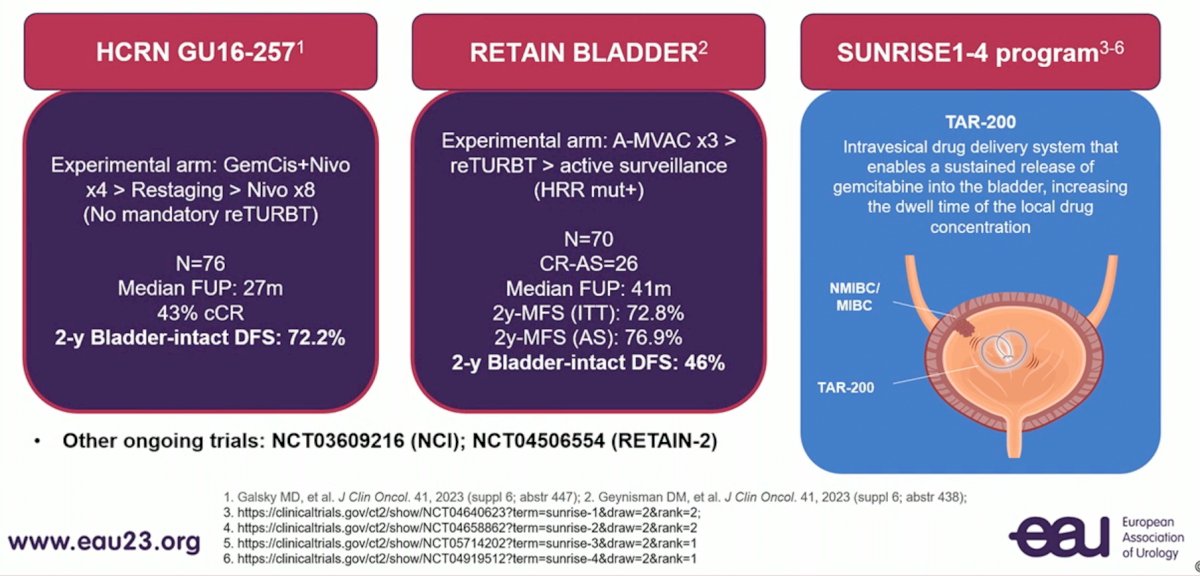
Dr. Necchi concluded with the following comments regarding filling the management gaps within bladder-sparing treatment over the next decade:
- New effective therapy options are available, and the optimal positioning of radical surgery is uncertain: accumulating data from ongoing studies may be beneficial
- Multidisciplinary discussion stands at the forefront of such tremendous achievements
- Shared decision-making still enables patients to engage and find the right treatment for them
- Safety of newer therapeutic options long-term is still a concern
At this point, the debate was turned over to Dr. Bamias to argue in favor of local therapy in the setting of oligometastatic disease. Dr. Bamias argued that oligometastatic cancer is a disease state along a continuum from localized to systemically, overtly metastatic disease, and thus requires a tailored diagnostic and therapeutic approach. This concept is not unique to bladder cancer and has been described in many other disease states, including lung cancer.

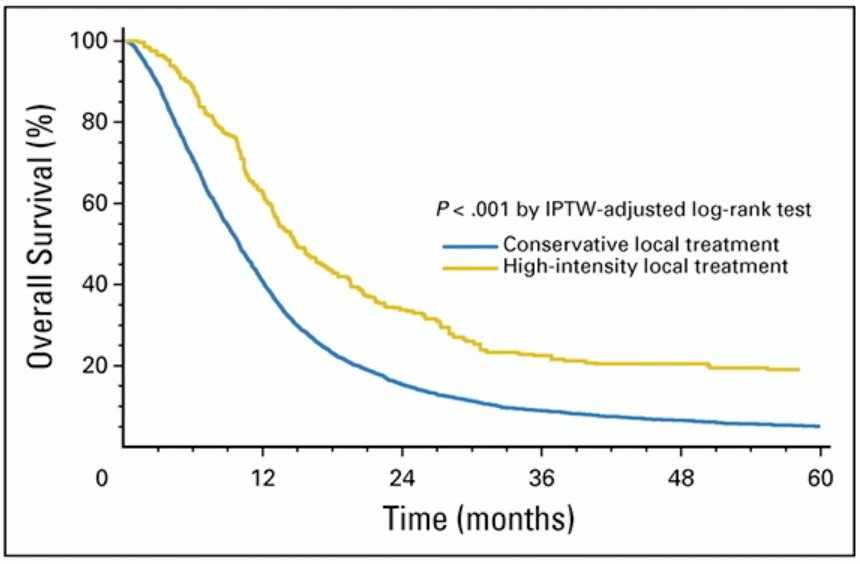
Dr. Bamian highlighted the results of a propensity score-weighted analysis from the National Cancer Database that evaluated the efficacy of high-intensity local treatment for metastatic urothelial carcinoma of the bladder. In this analysis, the investigators demonstrated that treatment was associated with significant improvements in overall survival.6
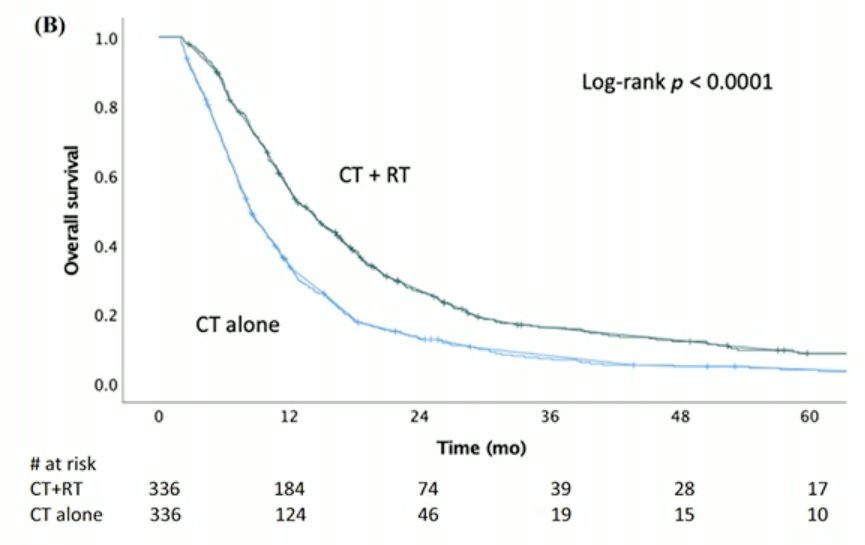
This was similarly demonstrated in another propensity score-matched analysis of patients with metastatic urothelial carcinoma receiving systemic chemotherapy, whereby addition of local radiation therapy to the primary bladder tumor was associated with significant improvements in overall survival.7 As such, there appears to be some evidence that local consolidation in well-selected patients receiving systemic therapy may be associated with survival benefits.
Dr. Bamias next highlighted the same 3 studies previously addressed by Dr. Necchi earlier in the debate that highlight a benefit for surgical resection in well-selected patients with limited metastatic burden. He argued that in the 2nd study below (Necchi et al. Eur Urol Focus 2019), no benefit was observed by consolidating with post-chemotherapy LND in this patient cohort, as patients with unfavorable response to chemotherapy, and thus inherently unlikely to benefit from surgery, were included in this analysis.
In addition to local therapy to the bladder, as has been seen in many other disease sites, there is an emerging role for metastasis directed therapy with stereotactic body radiation therapy, as demonstrated by the ongoing trials summarized in the table below:
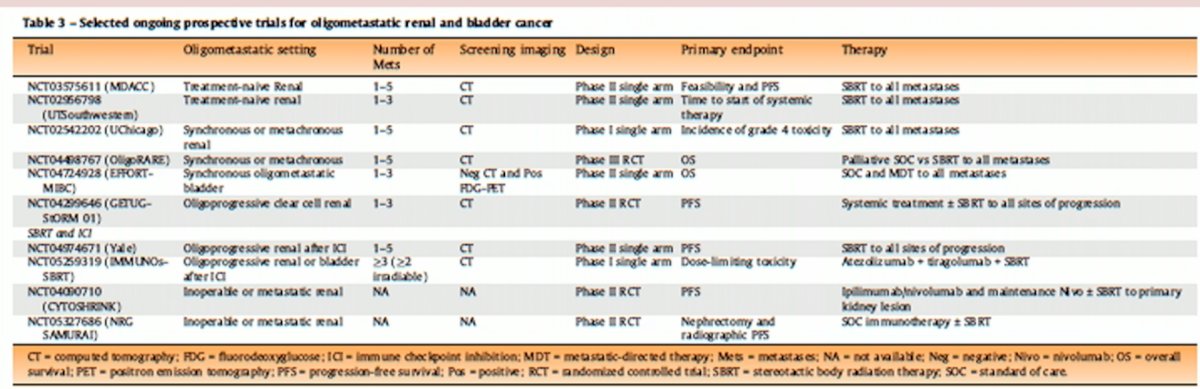
Dr. Bamias argued that we should not dismiss the concept of oligometastatic disease in bladder cancer, and instead we should intensify research in this field. Lack of data does not make a concept redundant, with oligometastatic disease a distinct entity in other cancers. As such, why should this be different in bladder cancer? This concept was highlighted in the recent consensus meeting statements regarding the role of treatment of curative intent in oligometastatic disease:
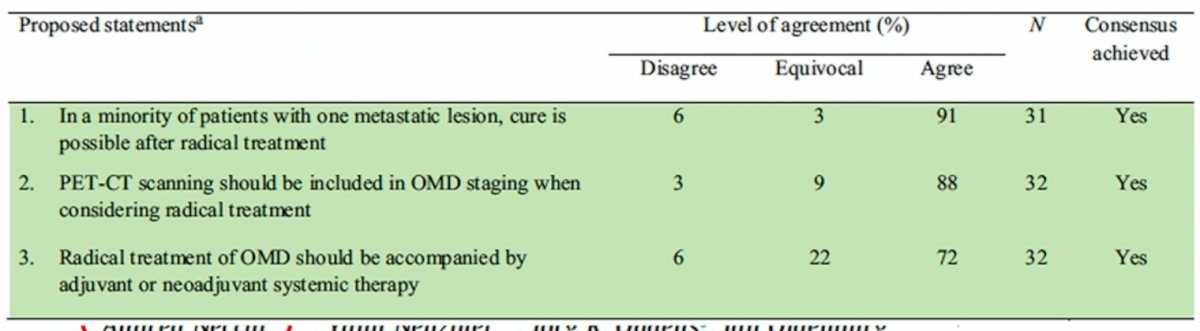
In summary, focused clinical and translational research in oligometastatic bladder cancer represents an unmet need. Systemic treatment remains the most vital but not the only component of the optimal management of oligometastatic bladder cancer. Metastases-directed therapy should be considered in patients with oligometastatic bladder cancer and favorable response to systemic therapy.
Presented by:- Professor Andrea Necchi, MD, Director of GU Medical Oncology, San Raffaele Hospital and Scientific Institute, Milan, Italy
- Dr. Aristotelis Bamias, MD, PhD, Professor at the Medical School, University of Athens, Greece and Director of the 2nd Propaedeutic Dept of Internal Medicine, ATTIKON University Hospital, Athens, Greece
Written by: Rashid K. Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 European Association of Urology (EAU) Annual Meeting, Milan, IT, Fri, Mar 10 – Mon, Mar 13, 2023.
References:- Necchi et al. Panitumumab Treatment for Advanced Penile Squamous Cell Carcinoma When Surgery and Chemotherapy Have Failed. Clin Genitourin Cancer 2015. (3):231-6.
- Necchi et al. Comparison of Fibroblast Growth-factor Receptor Gene Alterations at the DNA versus Messenger RNA Level in Advanced Urothelial Cancer: Insights for Clinical Research. Eur Urol Focus 2019. 5(4):689-692.
- Moschini et al. Efficacy of Surgery in the Primary Tumor Site for Metastatic Urothelial Cancer: Analysis of an International, Multicenter, Multidisciplinary Database. Eur Urol Oncol 2020. 3(1):94-101.
- Powles et al. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. Lancet Oncol 2021. 7:931-945.
- Powles T et al. Avelumab Maintenance Therapy for Advanced or Metastatic Urothelial Carcinoma. N Engl J Med 2020. 383:1218-1230.
- Seisen et al. Efficacy of High-Intensity Local Treatment for Metastatic Urothelial Carcinoma of the Bladder: A Propensity Score-Weighted Analysis From the National Cancer Data Base. J Clin Oncol 2016. 34(29):3529-3536.
- Fischer-Valuck BW et al. Clin Genitourin Cancer 2021.

