In this presentation, the authors report an interim analysis of baseline characteristics, bone-pain related quality of life, and safety outcomes of patients who received at least one dose of Radium 223. In this analysis, patients were grouped on the extent of disease (EOD) at baseline, with two subgroups defined: EOD 1-2 (<=20 metastases) and EOD 3-4 (>20 metastases super-scan). Pain response was determined by the worst pain item on the Brief Pain Inventory – Short Form (BPI-SF) patient questionnaire. Clinically meaningful pain response was defined as an improvement of two points from the baseline BPI-SF worst pain score at any post-baseline assessment.
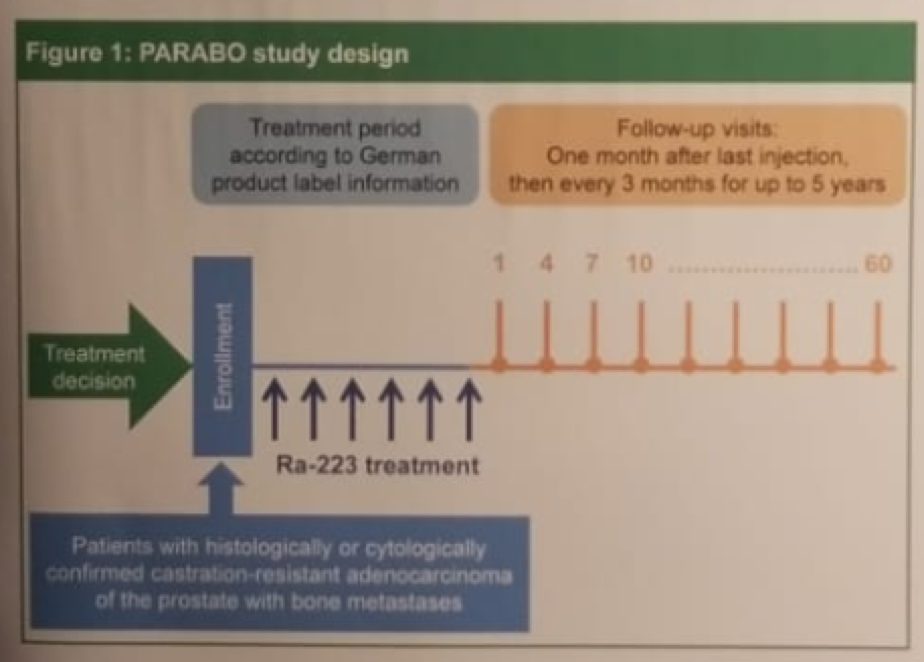
Figure 1 – PARABO study design:
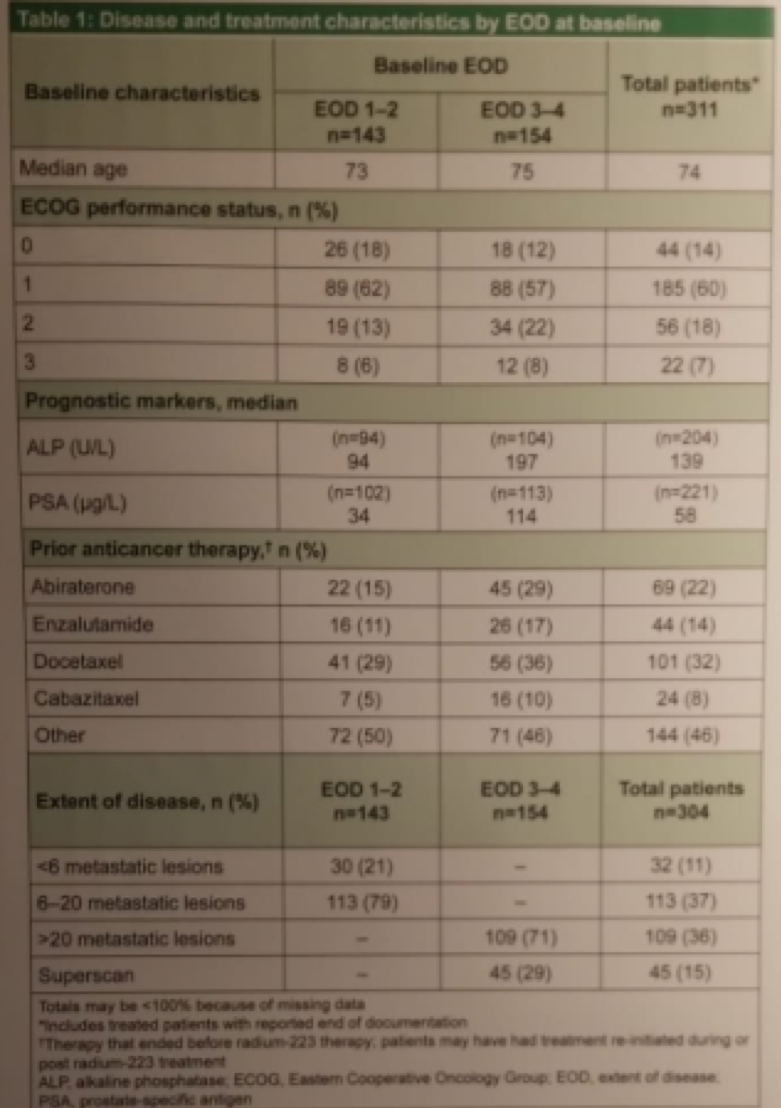
The results showed that patients with EOD 3-4 had a trend towards poorer ECOG performance status at baseline, and higher prior anticancer therapy use compared with the EOD 1-2 group. Moreover, PSA and alkaline phosphatase levels at baseline were considerably higher for EOD 3-4 patients than for EOD 1-2 patients (Table 1).
Table 1: Disease and treatment characteristics:
Nearly half of all patients used opioids (49%). Mean BPI-SF worst pain scores indicated a trend towards reduction in pain as treatment cycles increased (Figure 2). Most patients (60%) reported ≧1 pain response during observation (Table 2). A higher proportion of patients reported ≧1 pain response in the group that did not use opioids compared with the group that used opioids (65% vs. 55%).
Figure 2 – Change in BPI-SF score in patients treated with Radium – 223:

Table 2- Pain response during observation:

The most common treatment-related adverse effect was anemia, which was observed in 9% of patients, as seen in Table 3. The rates of fractures in the total population was very low (3%).
Table 3 – Treatment-emergent adverse effects:

In conclusion, patients reported a reduction in pain, regardless of EOD status, following Radium-223 treatment. At baseline, bone pain-related quality of life was worst in patients with opioid use, irrespective of EOD status. Radium-223 mediated pain reduction and bone pain-related quality of life improvement appear independent of EOD status. Lastly, the rate of fractures was low for both EOD groups.
Presented by: Cosimo De Nunzio, MD, Sapienza University of Rome, Sant'Andrea Hospital, Department of Urology, Rome, Italy
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, Twitter: @GoldbergHanan, at the 34th European Association of Urology (EAU 2019) #EAU19, conference in Barcelona, Spain, March 15-19, 2019.
References:

