(UroToday.com) Michael Leapman and colleagues examine the association between the use of prostate MRI and tissue-based genomic tests on the impact of clinical management in the real-world, using SEER-Medicare data.
As the authors note, prostate magnetic resonance imaging (MRI) and tissue-based gene expression (genomic) tests improve local staging and estimates of disease prognosis and have been incorporated into national and international guidelines. However, the impact of their use in clinical management is not well understood.
Prior work from his group and other ecological analyses have demonstrated that the use of testing may be associated with increased observation (vs. treatment) and lower positive surgical margin rates. On the other hand, testing may also increase the intensity of treatment without clear benefit – treatment of low-risk cancer, non-nerve sparing RP, added ADT to XRT or pelvic LND at the time of RP.
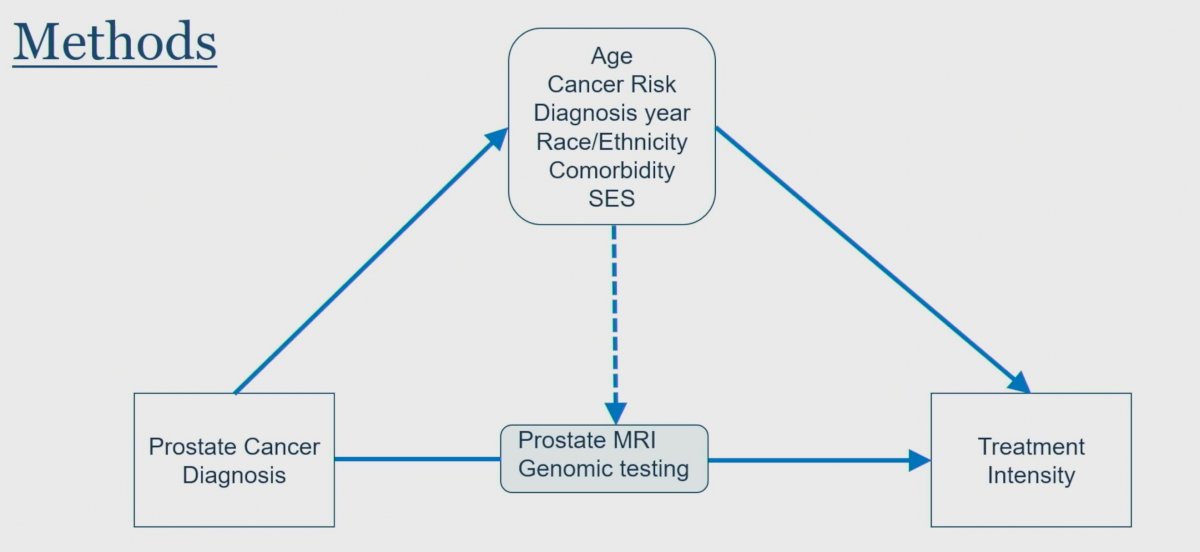
The authors completed a retrospective cohort study using SEER-Medicare, with a specific focus on clinically localized prostate cancer diagnosed between 2013-2017 who were treated with prostate radiation (XRT) or radical prostatectomy (RP). They identified prostate MRI and genomic tests (Decipher, Prolaris, Oncotype) using specific administrative claims codes.
Their primary study outcome was treatment intensification, which they defined as any use of androgen deprivation therapy (ADT) for prostate XRT or pelvic lymph node dissection (PLND) for RP, respectively. They also assessed the associations between patient-level receipt of prostate MRI and genomic testing and treatment intensification via multivariable logistic regressions, adjusting for clinical and sociodemographic factors, and further stratifying the analyses by D’Amico clinical risk status.
* However, it should be noted that they presume treatment intensification was due to these tests, but in this cohort study, it will never be known physician decision making – physicians may have opted for ADT or PLND regardless of these tests. There are other factors that may influence the decision to get the MRI or genomic test as well. They acknowledged this as well.
They identified 36,191 patients who met inclusion criteria.
In the overall cohort, receipt of prostate MRI was associated with increased odds of treatment, while genomic testing was associated with decreased odds of treatment.

Broken down by risk strata:
- Low-risk: MRI and genomic testing associated with decreased odds of treatment (HR 0.75 and 0.67, respectively)
- Intermediate Risk and High-risk: MRI was associated with increased risk (HR 1.20 and 1.37 respectively) while genomic testing was associated with decreased odds of treatment (HR 0.40 and 0.45, respectively)
In the overall cohort, receipt of prostate MRI was associated with increased odds of treatment intensification, while genomic testing was not associated with treatment intensification.
This carried through to all risk strata:


By specific treatments, prostate MRI was associated with increased odds of PLND (OR 2.54, 95% CI 2.24-2.87) and ADT (OR 1.37, 95% CI 1.26-1.48) across all risk strata, while genomic testing was associated with decreased odds of PLND (OR 0.67, 95% CI 0.54-0.83) and ADT (OR 0.55, 95% CI 0.41-0.76). This analysis is limited by the absence of MRI and genomic testing results.
Based on this, the authors concluded that there were distinct patterns of management associated with prostate MRI and genomic testing in localized prostate cancer. These findings suggest the need for additional information about the independent role of diagnostic technologies in decision-making.
Presented by: Michael Leapman, MD, Yale School of MedicineWritten by: Thenappan (Thenu) Chandrasekar, MD – Urologic Oncologist, Associate Professor of Urology, University of California, Davis @tchandra_uromd @UCDavisUrology on Twitter during the 2023 American Urological Association (AUA) Annual Meeting, Chicago, IL, April 27 – May 1, 2023

