First, he started by reviewing active surveillance. Active surveillance (AS) is a treatment option for low-risk prostate cancer per the EAU and AUA guidelines. It aims to avoid the side effects of unnecessary invasive therapy while maintaining the opportunity for cure if there is subsequent progression. Unfortunately, screening for prostate cancer contributes to an unavoidable detection of low-risk prostate cancer (overdiagnosis), resulting in a large proportion of men being on active surveillance. Fear of prostate cancer and uncertainty are the primary drivers for prostate cancer screening.
Once diagnosed with low-risk prostate cancer and put on active surveillance, men can carry a heavy burden. The focus of the presentation is on anxiety and quality of life. Anxiety is a feeling of unease, such as worry or fear, that can be mild or severe. It can range from normal stress to pathologic condition and also from incidental to continuous – to the point it can affect daily life.
It is the main symptom of many conditions, including panic disorder, phobias, PTSD, social anxiety disorder
Why is measuring anxiety relevant?
- It has a direct impact on patients quality of life (reduces QOL measures)
- It influences shared decision-making and outcomes of diseases 25% of urologic patients experience anxiety, enhancing decisional conflict
- It reduces treatment compliance ~50% treatment dropout in patients with anxiety disorders
- It induces unwanted interventions (medical, other) ~26% of individuals with anxiety obtained extra medications
How do we quantify QoL and anxiety?
There are numerous validated metrics including:
- EQ-5D-5L (generic health)
- EORTC-QLQ-30 (cancer related QoL)
- EPIC 26 (expanded Prostate cancer index)
- RAND-36 (includes a domain on emotional wellbeing)
He then noted that there are numerous controversial opinions in AS:
- Living with untreated cancer leads to anxiety (on one end of the spectrum) --- but on the other end, AS may be associated with lower levels of anxiety compared to curative treatment due to loss of physical functions
- Diagnostic testing can either induce OR decrease stress, depending on the patient
There are a few studies that have addressed these questions, which he then summarizes briefly:
- Cross-sectional snap shot studies – EUPROMS (2020)
- Longitidunal studies: REDEEM (2021), HAROW (2020), Finnish PRIAS (2021), ProtecT (2016)
- Psychological Personality Studies: PRIAS cohorts
- Interventional studies – there aren’t any!
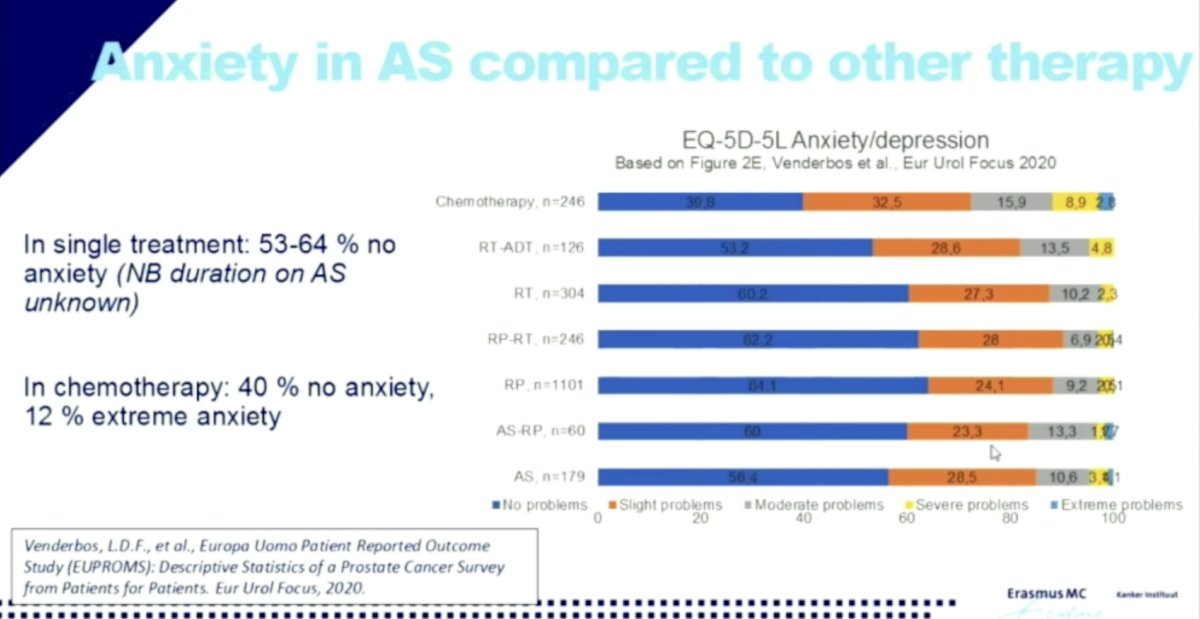
EUPROMS1
In this cross-sectional survey among PCa patients currently receiving or having received treatment, when they specifically assessed anxiety based on therapy type, AS was no different than the other treatment modalities:
REDEEM2
In their retrospective analysis of all 302 subjects from the Reduction by Dutasteride of clinical progression Events in Expectant Management (REDEEM) study, Naha et al. found that high level of anxiety was ~5% and anxiety was not correlated with treatment shift.
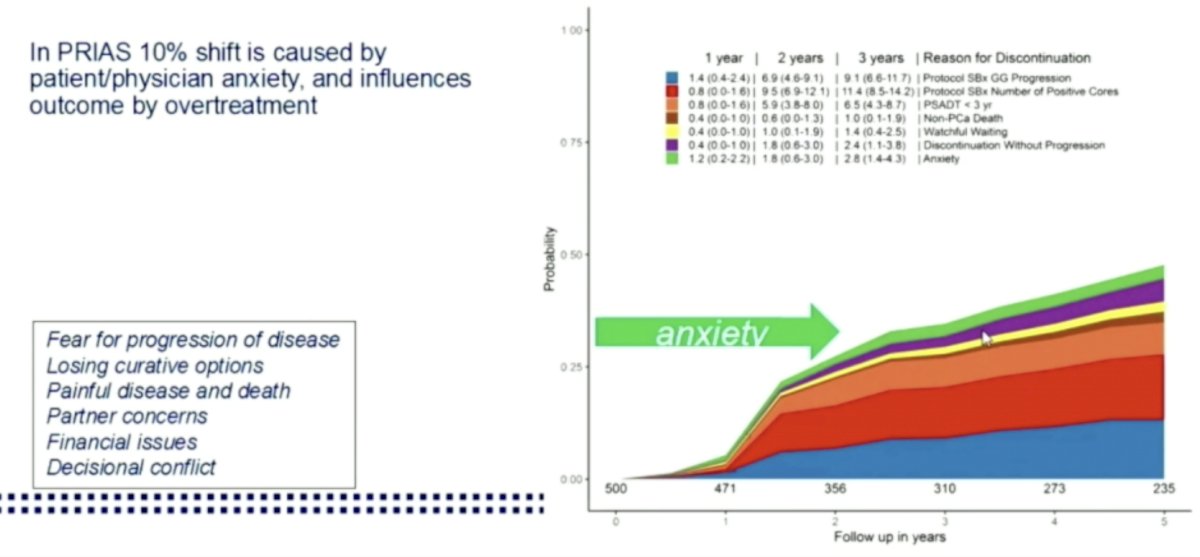
 PRIAS3
PRIAS3PRIAS
In the PRIAS study itself, generic anxiety and fear of disease progression decreased significantly over time. Only 5% of patients discontinued due to AS (of 32% total)
In similar PRIAS evaluation of subpopulations for individual countries, they found:
- Japan PRIAS – Men on AS have better QOL than the matched general population
- Finnish PRIAS – 3.5% of men discontinued AS due to anxiety within the first 3-6 months. No difference in patients on continued AS vs. RALP.
What are some potential methods of identifying anxiety upfront?
- Neuroticism and introversion are associated with increased risk for depression and anxiety in patients
- Symptoms of anxiety and depression are more frequent in individuals with a lower level of education and those living alone
How do patients cope with anxiety?
- Coping mechanisms include fighting spirit, anxious preoccupation, fatalism, helplessness, avoidance
Lastly, what are some potential interventions to reduce anxiety?
- Even a very brief compassionate intervention may help to reduce the patience anxiety
- Use of decision aids to ensure a high standard of patient information or referring someone to an online support group has been shown to effectively reduce anxiety and depression
- The implementation of nurse navigators who can help patients navigate the healthcare system
He concluded with the following remarks:
- In active surveillance anxiety significantly influences decision-making and outcome
- 3.5-7% shift towards invasive treatment maybe due to anxiety
- Emotional distress might be routinely assessed with validated instruments before clinical consultation
- Care providers should recognize patience with less education and those who live alone as the main experience more distress
- Simple interventions and supports that fit coping mechanisms are available and should be offered early on.
Presented by: Chris H. Bangma, MD, PhD, Professor of Urology, Erasmus Medical Center
Written by: Thenappan (Thenu) Chandrasekar, MD – Urologic Oncologist, Assistant Professor of Urology, Sidney Kimmel Cancer Center, Thomas Jefferson University, @tchandra_uromd on Twitter during the 2021 American Urological Association, (AUA) Annual Meeting, Fri, Sep 10, 2021 – Mon, Sep 13, 2021.
References:
- Venderbos LDF, Deschamps A, Dowling J, Carl EG, Remmers S, van Poppel H, Roobol MJ. Europa Uomo Patient Reported Outcome Study (EUPROMS): Descriptive Statistics of a Prostate Cancer Survey from Patients for Patients. Eur Urol Focus. 2020 Dec 3:S2405-4569(20)30297-2. doi: 10.1016/j.euf.2020.11.002. Epub ahead of print. PMID: 33281109.
- Naha U, Freedland SJ, Abern MR, Moreira DM. The association of cancer-specific anxiety with disease aggressiveness in men on active surveillance of prostate cancer. Prostate Cancer Prostatic Dis. 2021 Jun;24(2):335-340. doi: 10.1038/s41391-020-00279-z. Epub 2020 Sep 8. PMID: 32901094.
- Venderbos LD, van den Bergh RC, Roobol MJ, Schröder FH, Essink-Bot ML, Bangma CH, Steyerberg EW, Korfage IJ. A longitudinal study on the impact of active surveillance for prostate cancer on anxiety and distress levels. Psychooncology. 2015 Mar;24(3):348-54. doi: 10.1002/pon.3657. Epub 2014 Aug 20. PMID: 25138075.

