(UroToday.com) The 2021 Ramon Guiteras Lecture at the 2021 American Urologic Association (AUA) annual meeting was provided by Dr. Eila Skinner who provided her perspective on three decades of bladder cancer treatment and advances.
Dr. Skinner started by highlighting what bladder cancer was like in 1990, noting that there was an understanding of the basic epidemiology and biology, the importance of stage and grade, and the role of smoking in the etiology. Importantly, in 1994, the role of p53 mutation in invasive bladder cancer was first recognized.1 During the ’90s, all non-muscle invasive bladder cancer was deemed “superficial” bladder cancer, which included a wide range of tumor biology. BCG was the only commonly used intravesical treatment in the United States, however, post-TUR single-dose chemotherapy was becoming routine in Europe, but not widely adopted in the United States. Additionally, early work from the University of Southern California (where Dr. Skinner worked for many years) showed that G3T1 disease was associated with a high rate of upstaging at subsequent cystectomy with behavior similar to clinical stage T2 disease.
With regards to muscle-invasive bladder cancer in the 1990s, it was recognized that clinical staging was inaccurate. As such, cystectomy was the only real option in the United States, however, the importance of surgical technique was already recognized, noting that positive margins were associated with a high rate of metastasis. Also recognized was the value of a thorough pelvic lymph node dissection, with studies from this era suggesting that cure is potentially achievable in 30% of patients with positive lymph nodes. During this time period, the focus at high volume centers was in two main areas:
- Can we improve the cure rate with the addition of chemotherapy?
- Can we make cystectomy more palatable to patients with continent urinary diversions?
Dr. Skinner notes that starting in the early 80’s multiple centers developed continent diversion techniques, with the majority of the initial reconstructions being cutaneous diversions. Neobladders for men started in the mid-1980s, with few centers (including USC) offering this urinary diversion option to most of their patients. Dr. Skinner notes that by the early 1990s at USC almost 80% of men undergoing a cystectomy had a neobladder reconstruction.
Fast-forwarding to 2010, Dr. Skinner highlighted that the SWOG 8507 trial showed a benefit of maintenance BCG for CIS NMIBC. Additionally, there was a shift towards acceptance of the role of post-TURBT therapy, including repeat TURBT for T1 patients decreasing the incidence of NMIBC understaging. Furthermore, there was renewed interest in intravesical chemotherapy, including gemcitabine and docetaxel, as well as new agents being explored with new mechanisms. Additionally, the SWOG 8710 and EORTC randomized trials had recently shown a benefit for neoadjuvant chemotherapy prior to radical cystectomy.
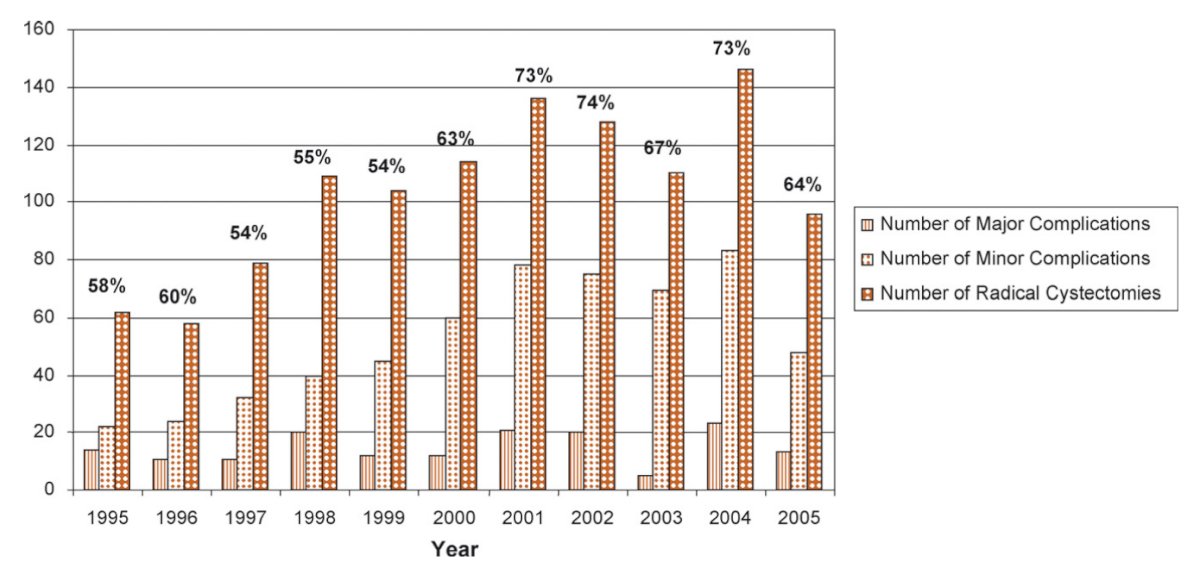
With regards to surgery, large series demonstrated the effectiveness of surgery, but a lack of improved survival over 30 years of follow-up. Additionally, there was initial recognition that hospital and surgeon volume correlated with complex surgical procedures, such as radical cystectomy. A 2009 report from Memorial Sloan Kettering Cancer Center published early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology2 and paving the way for other centers to report their complication data. This study found that 735/1,142 (64%) of patients experienced a complication within 90 days of surgery. The following figure demonstrates the variation in the number of perioperative complications by year of surgery relative to the number of radical cystectomies:
Dr. Skinner then highlighted three key events from the past decade that have changed the landscape of bladder cancer:
- The TCGA and several multi-institutional genomic studies have identified subtypes of high-grade urothelial cancer. These are prognostic and may possibly be predictive of response to specific therapies.
- The development of anti-PD1/PDL1 treatment: these therapies represent the first new effective systemic treatments in years, which are well tolerated even in older patients. Although the response rate is low, complete responses can be durable even when off of treatment. Additionally, anti-PD1/PDL1 is being investigated in earlier stages, including pembrolizumab being approved for BCG-unresponsive CIS in 2020 based on data from the KEYNOTE-057 trial.3 There are currently over 25 clinical trials in the pre-cystectomy disease space alone with combinations of immunotherapy, targeted agents, and chemotherapy.
- Development of new intravesical therapies for non-muscle invasive bladder cancer: this initiative has been made possible by the FDA accepting modified criteria for potential approval of new agents based on large phase II or phase III single arm trials for BCG-unresponsive disease. There are several new intravesical agents completing/completed phase II/III trials:
- Nadofaragene firadenovec, in the Instillidrin trial,4 showing a 24% complete response rate at one year in 103 patients
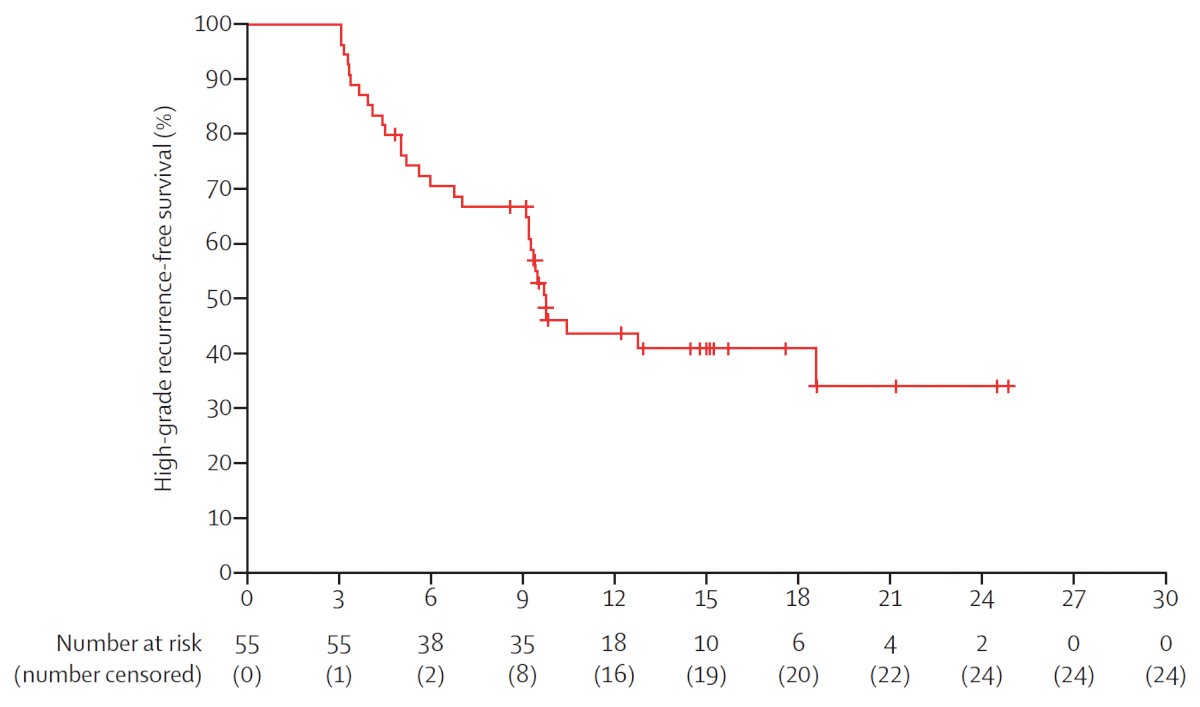
- CG0070 adenovirus-mediated selective oncolytic immunotherapy, showing 35% complete response rate at 18 months in 57 patients
- N803 IL15 superagonist plus BCG, showing a 42% complete response rate at 1 year in 81 patients.
What our patients need and what we want in the treatment of bladder cancer according to Dr. Skinner is as follows:
- Better diagnosis and surveillance
- More accurate staging
- Predict treatment response
- Minimize the burden of treatment
Whether we can replace cystoscopy with a urine test for surveillance of bladder cancer hinges on having a point of care test, with very high accuracy, and is inexpensive. To date, we do not have this test, although Dr. Skinner is hopeful this is possible in the near future. Indeed, our patients do not want to forego cystoscopy if the biomarker/urine test has poor accuracy. Additionally, histologic grading is still problematic whereby the WHO 2004 grading system lost information compared to the WHO 1973 grading system. Notably, we have had a shift to more high-grade tumors, likely causing over-treatment and obscuring the highest risk patients.
With regards to molecular imaging, standard PET/CT has not particularly improved staging, with high false negatives and occasional false positives. As such, there is a dire need to find urothelial carcinoma-specific PET molecules. Multi-parametric MRI has recently incorporated the utility of VI-RADS scoring system, which may help with assessing the depth of invasion. When utilized to identify MIBC and extravesical extension, a VI-RADS 4-5 lesion was 94% accurate for predicting MIBC.4
In order to move toward predicting treatment response, we need to continue moving towards personalized medicine. However, there are mixed results to date on the ability of genomic classifiers to predict response to specific treatments. Currently, basal subtypes seem to benefit the most from neoadjuvant chemotherapy, and luminal papillary tumors seem to do well regardless of neoadjuvant chemotherapy. Additionally, several subtypes seem to respond to PDL1 inhibitors, however PD-L1 expression status and tumor mutational burden do not correlate with response to checkpoint inhibitors in all studies. Finally, DNA damage repair mutations appear to make tumors quite sensitive to platinum chemotherapy. The remaining challenges for personalized medicine include:
- Tumor sampling
- Tumor heterogeneity
- Tumor plasticity in response to treatment
- Lack of standardization of assays
Dr. Skinner highlighted that there are several important future steps as we aim to continue improving and reshaping bladder cancer care:
- The need to standardize biomarkers and validate in prospective studies
- Prove whether minimizing treatment in specific groups (ie. skipping neoadjuvant chemotherapy in patients with luminal tumors or skipping cystectomy in patients with DNA damage repair mutations) will achieve the same outcomes
- Test whether biomarker-driven treatment is effective
We must keep working to minimize the burden of treatment for our patients, including (i) assessing best to logically sequence new treatments for NMIBC to manage cost, (ii) assess cystectomy, which is still associated with high degree of morbidity, with similar but not superior outcomes utilizing a robotic approach, (iii) assess whether chemoradiation is equivalent to cystectomy, focusing on the appropriate selection of patients and managing post-radiation failures/complications, and (iv) limiting cost of systemic treatments, which are often very high with the burden falling on the patient and their family.
Dr. Skinner concluded her presentation assessing three decades of bladder cancer treatment by highlighting what our patients want in bladder cancer care, noting that we have made immense progress, but still have a way to go:
- How can we prevent recurrence?
- How is my cancer going to behave?
- Will the treatments recommended actually work?
- Do I have to have a urine bag?
- Can you cure me and preserve my quality of life?
- How can I (and our society) manage the cost?
Presented by: Eila C. Skinner, MD, Thomas A. Stamey Research Professor of Urology, Stanford University, Palo Alto, CA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2021 American Urological Association, (AUA) Annual Meeting, Fri, Sep 10, 2021 – Mon, Sep 13, 2021.
References:
- Esrig D, Elmajian D, Groshen S, et al. Accumulation of nuclear p53 and tumor progression in bladder cancer. N Engl J Med. 1994 Nov 10;331(19):1259-1264.
- Shabsigh A, Korets R, Vora KC, et al. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol. 2009 Jan;55(1):164-174.
- Balar AV, Kamat AM, Kulkarni GS, et al. Pembrolizumab monotherapy for the treatment of high-risk non-muscle-invasive bladder cancer unresponsive to BCG (KEYNOTE-057): An open-label, single-arm, multicenter, phase 2 study. Lancet Oncol. 2021 Jul;22(7):919-930.
- Boorjian SA, Alemozaffar M, Konety BR, et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: A single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 2020 Nov 27:S1470-2045(20)30540-4.
- Ueno Y, Takeuchi M, Tamada T, et al. Diagnostic accuracy and interobserver agreement for the Vesical Imaging-Reporting and Data System for Muscle-invasive Bladder Cancer: A Multi-reader Validation Study. Eur Urol. 2019 Jul;76(1):54-56.

