She started off by providing basic demographics and where the field of advanced urothelial cancer started to where we are now.
Urothelial carcinoma is the sixth most common cancer in the United States and is responsible for about 164,000 new cases in 2021 and 31,940 deaths. In advanced urothelial carcinoma, disease control occurs in 65-85% with 1st line platinum-based chemotherapy – but PFS and OS are often limited. Since MVAC chemotherapy in 1989, there have been few achievements in the past 30 years aside from the introduction of gem/cis and ddMVAC. Until recently ~25-55% of patients received second-line treatment often with poor results. This has changed drastically with the advent of checkpoint immunotherapy and other novel agents.
The developments of the last 9 years are summarized below:
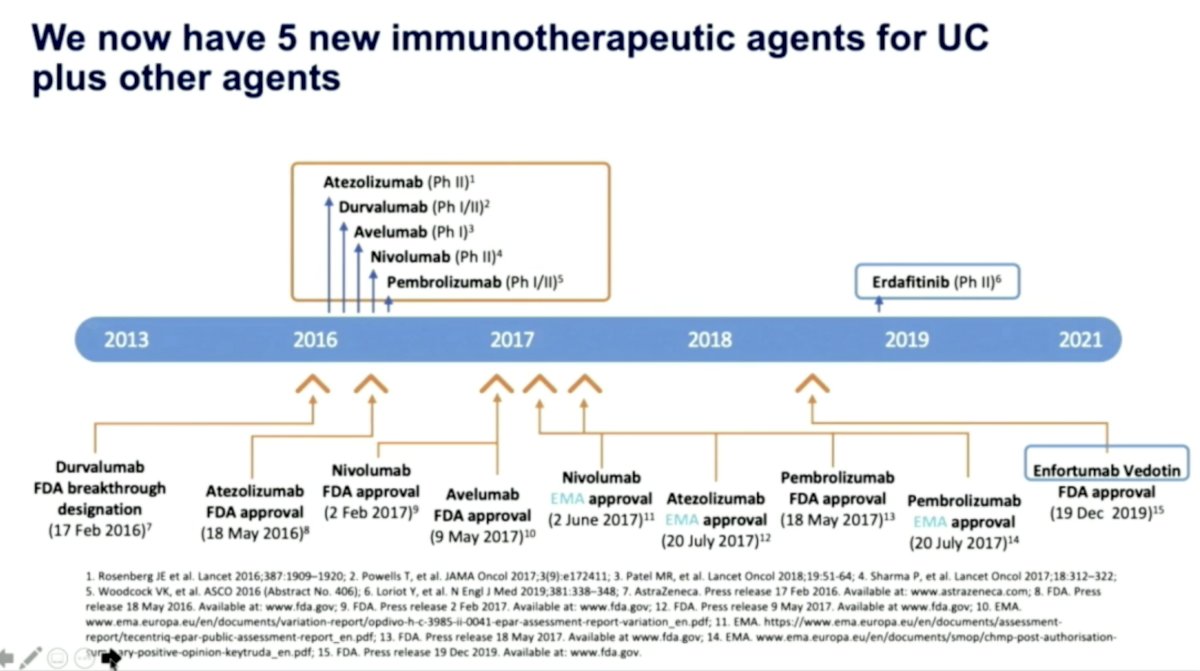
In addition, she highlighted the following facts:
- Urothelial carcinoma has a high number of somatic mutations compared to other tumors, closely modeling lung cancer which is very sensitive to IO therapy.
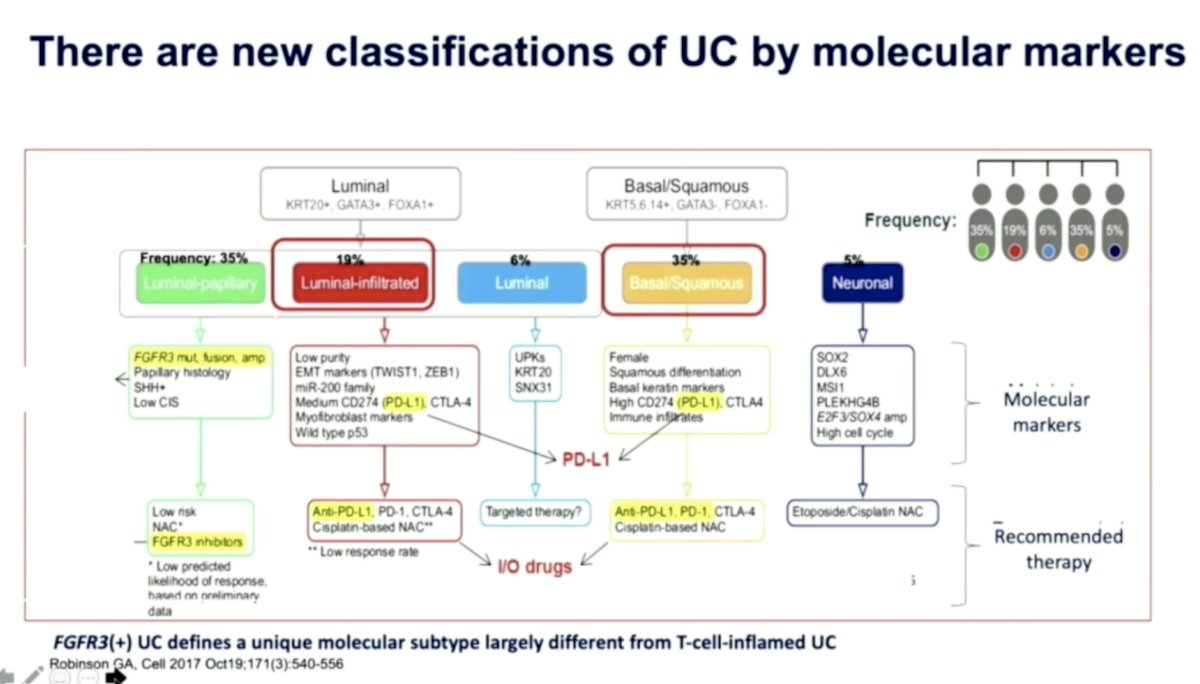
- There are new classification systems of UC using molecular markers – with data supporting the fact that luminal infiltrated and basal-squamous may be IO sensitive.
Second Line Setting (Platinum Refractory)
We have five immune checkpoint inhibitors approved for platinum-refractory advanced UC – with no head-to-head comparisons and often on the strength of phase 2 studies. These are summarized below:
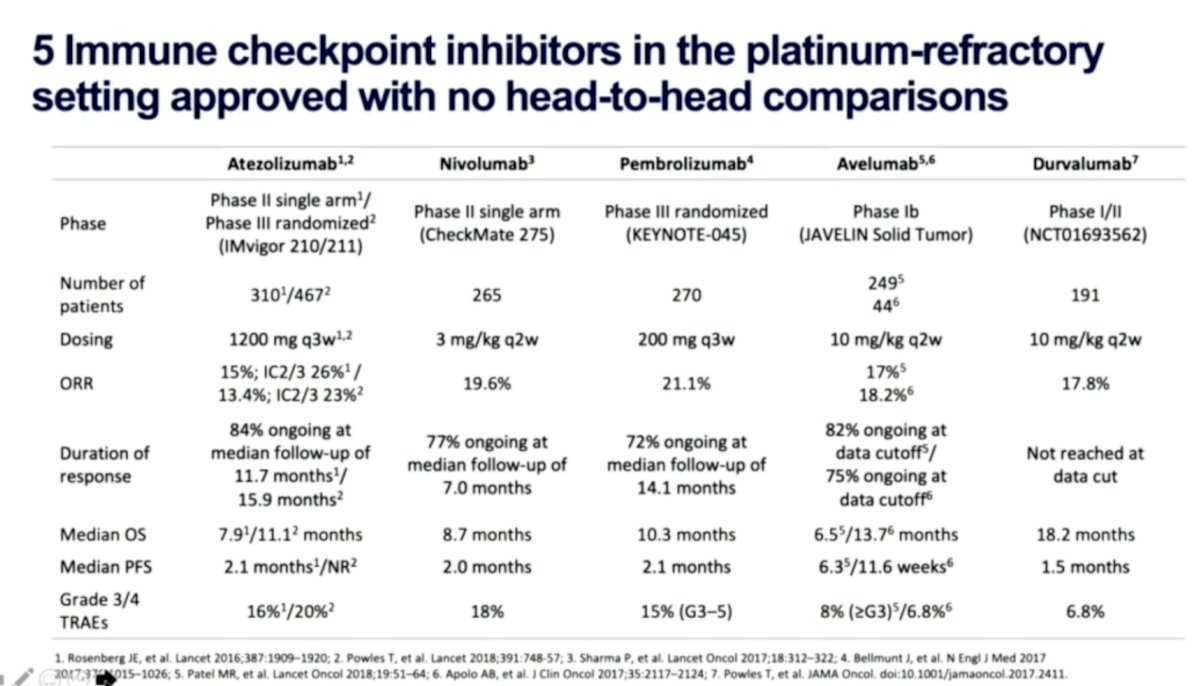
In general, these have an objective response ~20-% and median OS ~10 months.
The only Phase III RCT comparing pembrolizumab to chemotherapy in the second-line setting was the Keynote-045 study.1 In this study, pembro was associated with a 27% risk of death compared to second-line chemotherapy – and effectively solidified the role of IO therapy as second-line following platinum failure.

First Line Setting (Platinum Ineligible)
There are now two IO therapies approved for first-line platinum ineligible advanced urothelial carcinoma – atezolizumab and pembrolizumab. The data supporting their approval is listed below:

Objective response rates were ~25% and median OS ~10-15 months.
Except for the United States, these agents are limited to patients with PD-L1 expressing tumors – in the US, it is PD-L1 agnostic.
In updated OS reporting, patients in Keynote-052 with pembrolizumab who were PD-L1 positive appeared to have a greater benefit to therapy than those who were PD-L1 CPS score < 10.
Dr. Sternberg then highlighted the SAUL study, which she is a part of – this is a real-world study looking at the use of atezolizumab for platinum-ineligible patients in the first-line setting who would have been ineligible for clinical trial (HIV+, autoimmune disease, dialysis patients, CNS mets, etc). The primary outcome was safety, but OS and PFS were assessed.
As seen below, patients with these conditions had comparable outcomes to patients on trial, indicating they could safely be treated with IO.

First Line Setting, Platinum-Eligible
Lastly, she delved into the platinum-eligible population, looking at the role of IO therapy in this setting. She noted there were 5 key Phase III trials in this setting, summarized below:

Unfortunately, many of these trials were negative and IO therapy has not replaced chemotherapy in the first-line setting.
- IMvigor130 trial2 – demonstrated PFS benefit, but no OS benefit in the 2020 update.
- DANUBE trial3 – demonstrated no OS benefit of durvalumab or durva/treme
- KEYNOTE-361 trial4 – demonstrated no PFS or OS benefit with the addition of pembro
Based on these, there has been no approval of IO therapy for first-line with or without chemotherapy for advanced urothelial carcinoma.
She did touch on the JAVELIN Bladder 100 trial,5 which was slightly different from the rest. As previously summarized on UroToday this study actually assessed the role of avelumab as switch maintenance therapy following initial 1st line chemotherapy. Patients who had stable disease or objective response to first-line therapy were then put on avelumab maintenance therapy vs. best supportive care alone. The addition of avelumab maintenance therapy was associated with a 7.1 month OS benefit (HR 0.69).

In a smaller double-blind Phase II trial of maintenance pembrolizumab,6 there was a similar PFS benefit – but since there was no OS benefit evaluated, pembrolizumab cannot be recommended in this setting.
She does note that avelumab switch maintenance should be considered a standard of care for these patients at this time.
Antibody Drug Conjugates
The next section of her talk focused on ADCs (antibody-drug conjugates), which are a new approach to metastatic urothelial carcinoma. These agents link a monoclonal antibody to a cytotoxic drug or radionucleotide, thereby enabling the potency and effectiveness a monoclonal antibodies while also minimizing nonspecific systemic toxicity.
a) Enfortumab vedotin (EV) – this agent links monomethyl auristatin E (payload) to an antibody against Nectin-4.
- In EV-201,7 a phase II trial of EV after platinum-based chemotherapy and IO, there was a confirmed objective response rate of 44%
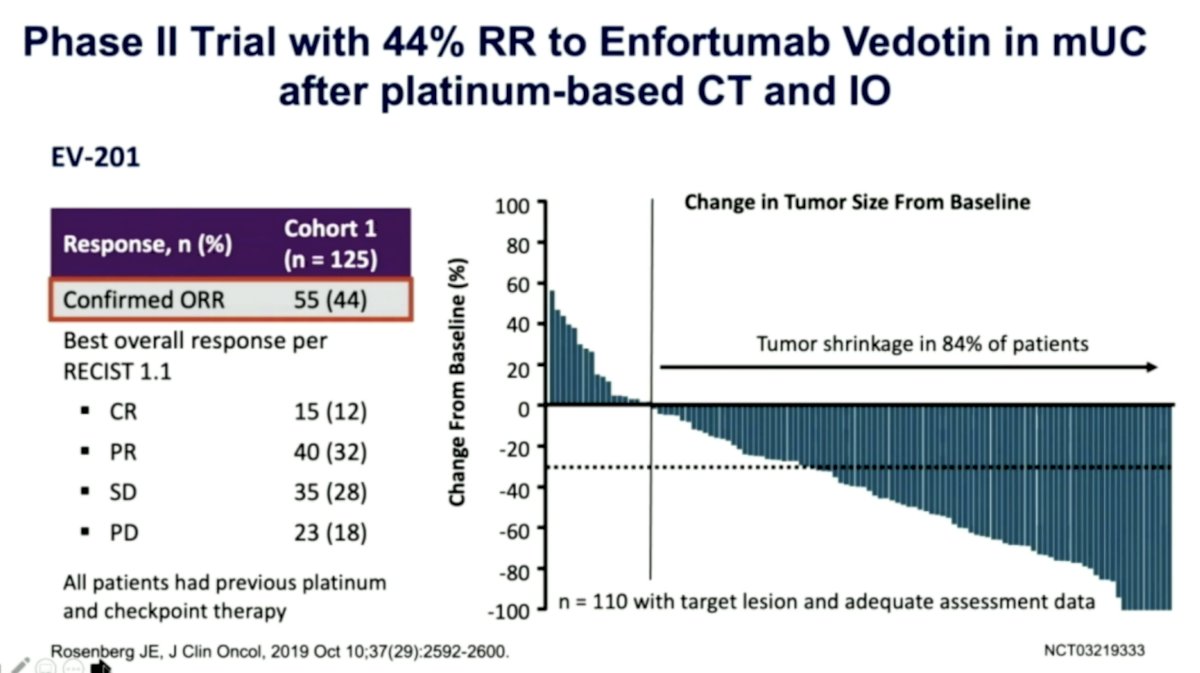
- In EV-301,8 this was further validated in a phase III RCT, with EV improving OS by 30% in the interim analysis. PFS benefit is seen below:

b) Sacituzumab Govitecan (SG) – this agent links SN-38 (payload) to an antibody against Trop-2.
- In TROPHY-U-01 Cohort 1,9 a phase II registrational study, SG was associated with a 27% ORR after platinum-based chemo and IO.
- There is an ongoing phase III trial enrolling currently to assess its efficacy in a phase III RCT, entitled TROPiCS-04
Fibroblast Growth Factor (FGFR) Pathway
The last section of her talk touched on FGFR inhibitors. FGFR3 mutations are commonly found in urothelial carcinoma. This makes this an appetizing target for targeted therapy.
Erdafinitib is an established FGFR3 inhibitor that has demonstrated success in the treatment of mUC.
- BLC2001 trial,10 a phase 2 trial of 99 patients with mUC with known FGFR3 mutation or FGFR2/3 fusion, erdafinitib demonstrated an ORR of 40% in all patients – and 59% in patients who had received prior IO therapy.
- An ongoing Phase III trial (THOR) will further evaluate this agent in patients with advanced urothelial carcinoma, either as a second-line therapy or 3rd line therapy.
Conclusions
She concluded with the following statements:
- Chemotherapy is here to stay! It is unlikely to be replaced as a first-line therapy
- Maintenance therapy with Avelumab has become a standard of care
- ADCs and FGFR targeted therapy are excellent options
- We have made progress!
Presented by: Cora Sternberg, MD, FACP, Professor of Medicine, Weill Cornell Medicine, New-York Presbyterian, New York
Written by: Thenappan (Thenu) Chandrasekar, MD – Urologic Oncologist, Assistant Professor of Urology, Sidney Kimmel Cancer Center, Thomas Jefferson University, @tchandra_uromd on Twitter during the 2021 American Urological Association, (AUA) Annual Meeting, Fri, Sep 10, 2021 – Mon, Sep 13, 2021.
References:
- Bellmunt J, de Wit R, Vaughn DJ, Fradet Y, Lee JL, Fong L, Vogelzang NJ, Climent MA, Petrylak DP, Choueiri TK, Necchi A, Gerritsen W, Gurney H, Quinn DI, Culine S, Sternberg CN, Mai Y, Poehlein CH, Perini RF, Bajorin DF; KEYNOTE-045 Investigators. Pembrolizumab as Second-Line Therapy for Advanced Urothelial Carcinoma. N Engl J Med. 2017 Mar 16;376(11):1015-1026. doi: 10.1056/NEJMoa1613683. Epub 2017 Feb 17. PMID: 28212060; PMCID: PMC5635424.
- Galsky MD, Arija JÁA, Bamias A, Davis ID, De Santis M, Kikuchi E, Garcia-Del-Muro X, De Giorgi U, Mencinger M, Izumi K, Panni S, Gumus M, Özgüroğlu M, Kalebasty AR, Park SH, Alekseev B, Schutz FA, Li JR, Ye D, Vogelzang NJ, Bernhard S, Tayama D, Mariathasan S, Mecke A, Thåström A, Grande E; IMvigor130 Study Group. Atezolizumab with or without chemotherapy in metastatic urothelial cancer (IMvigor130): a multicentre, randomised, placebo-controlled phase 3 trial. Lancet. 2020 May 16;395(10236):1547-1557. doi: 10.1016/S0140-6736(20)30230-0. PMID: 32416780.
- Powles T, van der Heijden MS, Castellano D, Galsky MD, Loriot Y, Petrylak DP, Ogawa O, Park SH, Lee JL, De Giorgi U, Bögemann M, Bamias A, Eigl BJ, Gurney H, Mukherjee SD, Fradet Y, Skoneczna I, Tsiatas M, Novikov A, Suárez C, Fay AP, Duran I, Necchi A, Wildsmith S, He P, Angra N, Gupta AK, Levin W, Bellmunt J; DANUBE study investigators. Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2020 Dec;21(12):1574-1588. doi: 10.1016/S1470-2045(20)30541-6. Epub 2020 Sep 21. Erratum in: Lancet Oncol. 2021 Jan;22(1):e5. PMID: 32971005.
- Powles T, Csőszi T, Özgüroğlu M, Matsubara N, Géczi L, Cheng SY, Fradet Y, Oudard S, Vulsteke C, Morales Barrera R, Fléchon A, Gunduz S, Loriot Y, Rodriguez-Vida A, Mamtani R, Yu EY, Nam K, Imai K, Homet Moreno B, Alva A; KEYNOTE-361 Investigators. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. Lancet Oncol. 2021 Jul;22(7):931-945. doi: 10.1016/S1470-2045(21)00152-2. Epub 2021 May 26. PMID: 34051178.
- Powles T, Park SH, Voog E, et al. Avelumab Maintenance Therapy for Advanced or Metastatic Urothelial Carcinoma. N Engl J Med 2020 Sept 24;383(13):1218-1230.
- Galsky MD, Mortazavi A, Milowsky MI, George S, Gupta S, Fleming MT, Dang LH, Geynisman DM, Walling R, Alter RS, Kassar M, Wang J, Gupta S, Davis N, Picus J, Philips G, Quinn DI, Haines GK 3rd, Hahn NM, Zhao Q, Yu M, Pal SK. Randomized Double-Blind Phase II Study of Maintenance Pembrolizumab Versus Placebo After First-Line Chemotherapy in Patients With Metastatic Urothelial Cancer. J Clin Oncol. 2020 Jun 1;38(16):1797-1806. doi: 10.1200/JCO.19.03091. Epub 2020 Apr 9. PMID: 32271672; PMCID: PMC7255983.
- Yu EY, Petrylak DP, O'Donnell PH, Lee JL, van der Heijden MS, Loriot Y, Stein MN, Necchi A, Kojima T, Harrison MR, Hoon Park S, Quinn DI, Heath EI, Rosenberg JE, Steinberg J, Liang SY, Trowbridge J, Campbell M, McGregor B, Balar AV. Enfortumab vedotin after PD-1 or PD-L1 inhibitors in cisplatin-ineligible patients with advanced urothelial carcinoma (EV‑201): a multicentre, single-arm, phase 2 trial. Lancet Oncol. 2021 Jun;22(6):872-882. doi: 10.1016/S1470-2045(21)00094-2. Epub 2021 May 12. Erratum in: Lancet Oncol. 2021 Jun;22(6):e239. PMID: 33991512.
- Powles T, Rosenberg JE, Sonpavde GP, Loriot Y, Durán I, Lee JL, Matsubara N, Vulsteke C, Castellano D, Wu C, Campbell M, Matsangou M, Petrylak DP. Enfortumab Vedotin in Previously Treated Advanced Urothelial Carcinoma. N Engl J Med. 2021 Mar 25;384(12):1125-1135. doi: 10.1056/NEJMoa2035807. Epub 2021 Feb 12. PMID: 33577729.
- Tagawa ST, Balar AV, Petrylak DP, Kalebasty AR, Loriot Y, Fléchon A, Jain RK, Agarwal N, Bupathi M, Barthelemy P, Beuzeboc P, Palmbos P, Kyriakopoulos CE, Pouessel D, Sternberg CN, Hong Q, Goswami T, Itri LM, Grivas P. TROPHY-U-01: A Phase II Open-Label Study of Sacituzumab Govitecan in Patients With Metastatic Urothelial Carcinoma Progressing After Platinum-Based Chemotherapy and Checkpoint Inhibitors. J Clin Oncol. 2021 Aug 1;39(22):2474-2485. doi: 10.1200/JCO.20.03489. Epub 2021 Apr 30. PMID: 33929895; PMCID: PMC8315301.
- Loriot Y, Necchi A, Park SH, Garcia-Donas J, Huddart R, Burgess E, Fleming M, Rezazadeh A, Mellado B, Varlamov S, Joshi M, Duran I, Tagawa ST, Zakharia Y, Zhong B, Stuyckens K, Santiago-Walker A, De Porre P, O'Hagan A, Avadhani A, Siefker-Radtke AO; BLC2001 Study Group. Erdafitinib in Locally Advanced or Metastatic Urothelial Carcinoma. N Engl J Med. 2019 Jul 25;381(4):338-348. doi: 10.1056/NEJMoa1817323. PMID: 31340094.

