(UroToday.com) The 2023 GU ASCO annual meeting included a session on trials in progress for prostate cancer, featuring a presentation by Dr. Arun Azad discussing the trial design of LuCAB, a phase I/II trial evaluating cabazitaxel in combination with [177Lu]Lu-PSMA-617 in patients with metastatic castration-resistant prostate cancer (mCRPC). [177Lu]Lu-PSMA-617 was recently FDA-approved for use in the post-taxane, post-antiandrogen setting in patients with mCRPC [1]. Despite conferring a survival benefit, for many the treatment response is short, and progression is inevitable.
One of the likely mechanisms limiting the durability of responses to [177Lu]Lu-PSMA-617 is heterogeneity in tumor PSMA expression. This can be screened for visually using PSMA and FDG PET/CT scans and inspecting for discordant disease, however, micrometastatic disease is unable to be evaluated in this way. In addition, [177Lu]Lu-PSMA-617 may not effectively target micrometastatic disease due to the longer path length of beta emitters. Cabazitaxel has radiosensitizing properties that may enhance the cytotoxic effect of [177Lu]Lu-PSMA-617, while also treating any PSMA-negative disease. Dr. Azad and colleagues hypothesize that the combination of [177Lu]Lu-PSMA-617 and cabazitaxel will be well tolerated and lead to more durable responses.
This single-center, single-arm phase I/II trial will enroll 32-38 patients with mCRPC to evaluate the safety and preliminary efficacy of cabazitaxel and [177Lu]Lu-PSMA-617 in combination. Up to 6 doses of [177Lu]Lu-PSMA-617 (7.4 GBq) will be administered intravenously every 6 weeks. Cabazitaxel will be given concurrently (dose range 12.5mg/m2 - 20mg/m2), on Day 2 and Day 23 of each 6-week cycle:
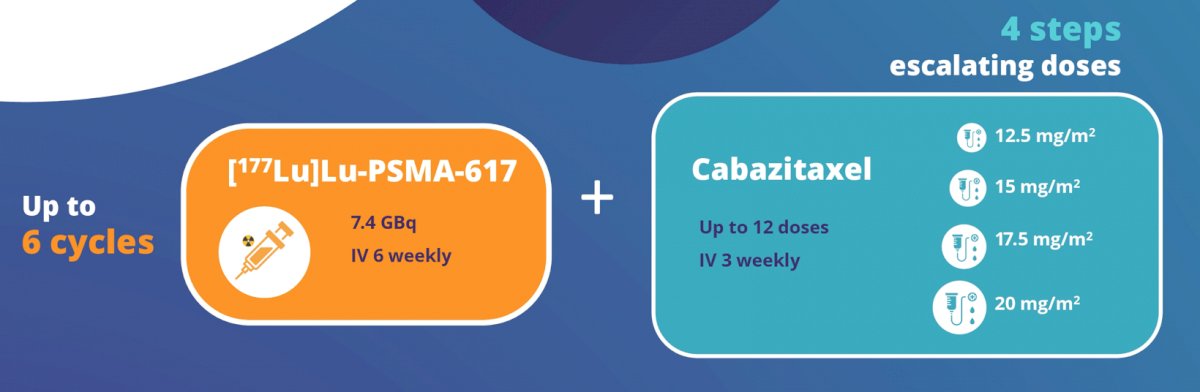
The dose of cabazitaxel will be escalated using a traditional 3+3 design. Key eligibility criteria include a diagnosis of mCRPC with PSMA-positive disease on PSMA PET/CT (SUVmax ≥15), with no evidence of diffuse marrow disease or sites of discordance on FDG PET/CT. Patients must have progressed after prior docetaxel and a second-generation antiandrogen, have adequate bone marrow and organ function and an ECOG performance status of 0-1. The primary objective is to establish the maximum tolerated dose of cabazitaxel and [177Lu]Lu-PSMA-617. Secondary objectives include:
- Measuring the frequency and severity of adverse events
- Assessment of efficacy through PSA 50% response rate
- Radiographic and PSA progression-free survival
- Overall survival
- Objective tumor response rate
- Evaluation of pain and health-related quality of life over the first 12 months

Blood draws will be taken at baseline, during treatment and at progression for ctDNA analysis, the results of which will be correlated with baseline patient and disease characteristics, and response outcomes, to determine biomarkers of treatment response and resistance. Dr. Azad concluded his presentation by noting that patient recruitment commenced in August 2022 and will continue for 18 months. As of January 2023, 5 patients have been enrolled in the study.
Clinical Trial Information: NCT05340374
Presented by: Arun Azad, PhD, MBBS, FRACP, Peter MacCallum Cancer Centre and Sir Peter MacCallum Department of Oncology, University of Melbourne, Melbourne, Australia
Co-Authors: Louise Kathleen Kostos, James Patrick Buteau, Grace Kong, Theresa Yeung, Juliana Di Iulio, Michael T Fahey, Heidi Fettke, Luc Furic, Michael S Hofman
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2023 Genitourinary (GU) American Society of Clinical Oncology (ASCO) Annual Meeting, San Francisco, Thurs, Feb 16 – Sat, Feb 18, 2023.
Reference:
- Sartor O, de Bono J, Chi KN et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2021 Sep 16;385(12):1091-1103.
- Hofman MS, Emmett L, Sandhu S, et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): A randomized, open-label, phase 2 trial. Lancet. 2021 Feb 27;397(10276):797-804.

