(UroToday.com) On the first day of the American Society for Clinical Oncology (ASCO) Genitourinary Cancer Symposium 2023 focussing on prostate cancer, Dr. Chrisropher Sweeney presented in Poster Session A on an analysis of the IMbassador250 trial the association between ctDNA fraction and androgen receptor (AR) alterations with therapeutic outcomes.
While there has been significant interest in the use of immune checkpoint inhibitors in advanced prostate cancer, one of the prominent trials, IMbassador250, failed to show an overall survival (OS) benefit for adding atezolizumab to enzalutamide for men with mCRPC who had prior progression on abiraterone in a prospective phase III international trial. However, these authors hypothesized that genomic biomarkers in ctDNA may identify patients who have poorer OS with 2nd generation novel hormonal therapy.
To assess this, the authors examined pre-treatment (but post-abiraterone progression) plasma samples from patients on IMbassador250 (NCT03016312). They performed comprehensive genomic profiling using FoundationOne Liquid CDx and detected elevated ctDNA tumor fraction (TF) using a novel algorithm that incorporates aneuploidy as well as tumor-derived short variant signal. A pre-specified TF cutoff of ≥ 2% was defined as high.

A prospective-retrospective biomarker statistical analysis plan was developed in accordance with Simon Criteria, pre-specifying analyses, cutoffs, and power assessments.
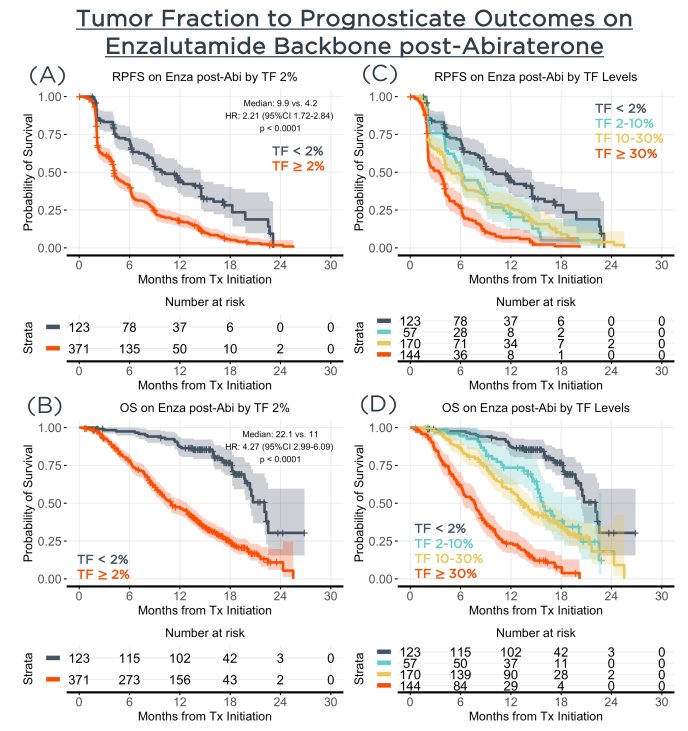
Among patients enrolled on IMbassador250, 494 baseline plasma specimens were evaluable. The TF high group [371 (74%)] had significantly shorter OS than TF low (median 11.0 vs. 22.1 months, HR 4.3, 95% CI 3.0 – 6.1, p < 0.0001) (primary endpoint).
The authors further performed a number of pre-specified supportive analyses which compared the performance to prognosticate OS (concordance [std error]) of baseline PSA alone (0.65 [0.63 – 0.67]) to a model consisting of all evaluable baseline features including PSA, treatment arm, age, race, ECOG score, hemoglobin, alkaline phosphatase, albumin, number of metastatic sites, and sites of metastasis (0.71 [0.70 – 0.73]) to TF alone (0.72 [0.71 – 0.73]) to all features plus TF (0.76 [0.74 – 0.77]).

The authors demonstrated a significant improvement of OS prediction by adding TF to all available clinical features using a likelihood ratio test (p < 0.0001). Focusing on the patients with TF ≥ 2% (n = 371), a further exploratory analysis identifed worse outcomes with detection of AR amplifications (n = 203 [55%], HR: 1.4, 95%CI: 1.1 – 1.8), while individual AR mutations were not as strongly associated with OS: L702 ([16%], HR: 1.2, 95%CI: 0.88 – 1.7), W742 ([4.3%], HR: 0.59, 95%CI: 0.30 – 1.2), H875 ([9.4%], HR: 0.71, 95%CI: 0.46 – 1.1), T878 ([25.3%], HR: 0.86, 95%CI: 0.65 – 1.1).
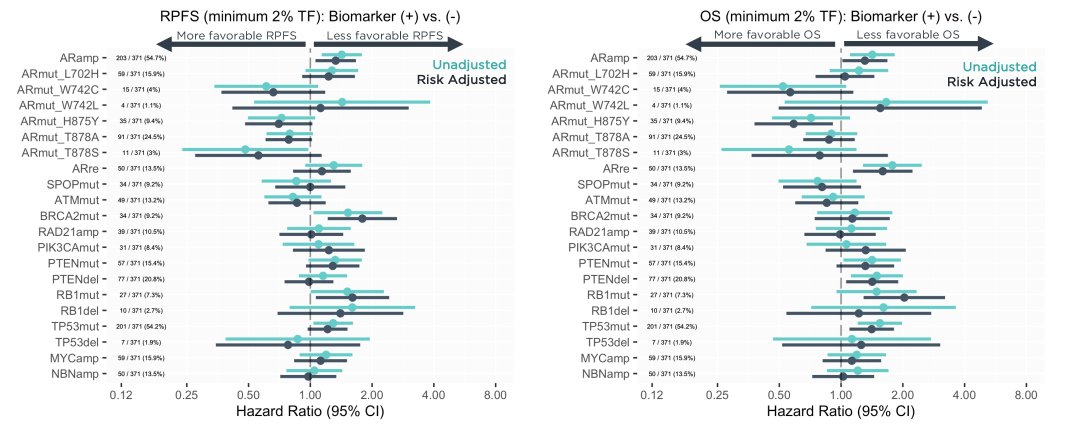
Thus, the authors conclude that this cohort of patients from the IMbassador250 trial demonstrated that a TF < 2% at baseline is strongly associated with favorable OS on enzalutamide after progression on abiraterone compared to TF ≥ 2%. Additionally, TF has comparable discriminatory ability to anticipate overall survival compared to all available clinical features combined, and adds significant prognostic power when combined with clinical features in this setting.
Presented by: Christopher Sweeney, MBBS Medical Oncologist at the Dana-Farber Cancer Institute
